Advanced Enzymatic Manufacturing of Posaconazole Intermediate for Global Pharma Supply Chains
Advanced Enzymatic Manufacturing of Posaconazole Intermediate for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of critical antifungal agents, particularly second-generation triazoles like posaconazole. Patent CN115448912A, published in December 2022, introduces a significant technological breakthrough in the preparation of the key posaconazole intermediate, structurally defined as Compound A. This document details a sophisticated multi-step synthesis that transitions from a diol precursor through enzymatic resolution and cyclization to the final tosylated product. For R&D directors and procurement specialists, this patent represents a pivotal shift away from traditional, low-yielding Lewis acid catalysis toward a more efficient, enzyme-mediated process. The innovation lies not merely in the chemical transformation but in the holistic optimization of solvent systems and purification protocols, addressing long-standing pain points regarding toxicity, yield loss, and operational complexity in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing this class of intermediates have historically relied on harsh chemical conditions that compromise both economic efficiency and environmental safety. For instance, earlier patents such as CN201310063736.9 described processes utilizing Lewis acids like zinc chloride or aluminum chloride in aprotic solvents at cryogenic temperatures ranging from -40°C to 0°C. These methods suffered from inherent inefficiencies, primarily due to the necessity of recrystallization to achieve acceptable purity, which resulted in substantial product loss and overall yields rarely exceeding 70%. Furthermore, alternative enzymatic approaches disclosed in patents like CN201180024633.0 attempted to mitigate some chemical hazards but introduced new logistical burdens, such as the requirement for pure acetonitrile as a reaction solvent. Pure acetonitrile is not only highly toxic but also complicates waste stream management, creating significant disposal costs and regulatory hurdles for large-scale facilities. The reliance on single-enzyme systems in these older routes often led to suboptimal stereoselectivity, necessitating additional downstream purification steps that further eroded the final mass balance.
The Novel Approach
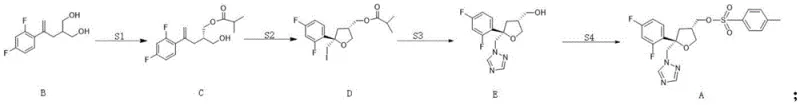
The methodology outlined in CN115448912A fundamentally reengineers the synthetic landscape by introducing a composite lipase system coupled with a non-ionic surfactant, Tween 80. This approach facilitates a highly selective kinetic resolution in a mixed solvent system of ethyl acetate and acetonitrile, drastically reducing the reliance on toxic pure acetonitrile. The strategic design of this route allows for a telescoped operation where the crude product from the initial enzymatic step can be directly utilized in the subsequent iodocyclization without intermediate isolation or solvent swapping. This continuity is made possible because ethyl acetate serves as a compatible medium for both the enzymatic esterification and the subsequent chemical cyclization. By eliminating the need for intermediate recrystallization and solvent exchanges, the new process minimizes material handling losses and significantly shortens the production cycle time. The result is a streamlined workflow that enhances the overall throughput of the facility while maintaining rigorous quality standards for the final intermediate.
Mechanistic Insights into Composite Lipase-Catalyzed Resolution and Cyclization
The core of this technological advancement resides in the synergistic interaction between the composite lipase blend and the surfactant additive during the initial esterification phase. The patent specifies a precise formulation comprising Novozym435 lipase and Chiralzyme IM-100, optimized to function within a specific temperature window of -10°C to -15°C. The inclusion of Tween 80 is not merely a solubilizing agent but acts as a critical promoter that enhances the interfacial activity of the enzymes, leading to an exceptional enantiomeric excess (ee) value of 99.8%. This high degree of stereocontrol is paramount for the biological efficacy of the final antifungal drug, as the wrong enantiomer can be inactive or even toxic. The mechanism likely involves the surfactant modifying the micro-environment around the enzyme active sites, facilitating better substrate access and transition state stabilization. This level of precision ensures that the downstream cyclization step proceeds with a homogenous chiral pool, preventing the formation of difficult-to-separate diastereomeric impurities that often plague triazole synthesis.
Following the enzymatic resolution, the process employs an iodocyclization mechanism to construct the critical tetrahydrofuran ring found in Compound D. In this step, the mono-esterified intermediate reacts with iodine and sodium bicarbonate. A key mechanistic advantage of this specific route is the tolerance of the reaction system to residual solvents and minor impurities from the previous step. The patent data indicates that diester impurities, which may form in trace amounts during S1, are inert under the iodocyclization conditions and do not interfere with the ring closure. This chemoselectivity allows the manufacturer to bypass the purification of Compound C entirely, feeding the crude reaction mixture directly into the cyclization reactor. This "one-pot" philosophy reduces the number of unit operations, lowers solvent consumption, and minimizes the exposure of reactive intermediates to potentially degrading conditions during isolation. The subsequent substitution with sodium triazole and final tosylation are executed with standard high-efficiency protocols, ensuring that the stereochemical integrity established in the first step is preserved throughout the entire synthetic sequence.
How to Synthesize Posaconazole Intermediate Efficiently
The synthesis of this high-value intermediate requires precise control over reaction parameters to maximize the benefits of the enzymatic cascade. The process begins with the dissolution of the starting diol in the optimized solvent blend, followed by the careful addition of the biocatalyst system. Maintaining the temperature strictly between -10°C and -15°C is crucial for achieving the reported 99.8% ee value. Following the enzymatic conversion, the reaction mixture undergoes a simple filtration to remove the immobilized enzyme, and the filtrate is washed with water to remove the surfactant before proceeding immediately to the iodocyclization. The detailed standardized operating procedures for each stage, including specific molar ratios and workup techniques, are essential for replicating the high yields observed in the patent examples. For a comprehensive guide on executing this synthesis with industrial precision, please refer to the technical protocol below.
- Step S1: Enzymatic esterification of Compound B using a composite lipase (Novozym435 and Chiralzyme IM-100) and Tween 80 in an ethyl acetate/acetonitrile solvent system.
- Step S2: Iodocyclization of the crude Compound C solution using iodine and sodium bicarbonate to form the tetrahydrofuran ring structure of Compound D.
- Step S3: Substitution with sodium triazole followed by hydrolysis and acid-base extraction to isolate high-purity Compound E.
- Step S4: Tosylation of Compound E using p-toluenesulfonyl chloride to yield the final Posaconazole Intermediate (Compound A).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply reliability. By eliminating the recrystallization step required in conventional Lewis acid methods, the process avoids the significant mass loss typically associated with such purification techniques. This efficiency gain means that less raw material is required to produce the same amount of final active pharmaceutical ingredient (API) intermediate, effectively lowering the cost of goods sold without compromising on quality. Furthermore, the replacement of pure acetonitrile with a mixed solvent system containing ethyl acetate reduces the procurement burden associated with hazardous, high-cost solvents and simplifies the regulatory compliance landscape regarding volatile organic compound (VOC) emissions.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of redundant purification steps and the optimization of solvent usage. In traditional routes, the need to isolate and recrystallize intermediate compounds consumes vast amounts of energy and solvent, while simultaneously discarding a portion of the product in the mother liquor. The new method's ability to telescope the first two steps without isolation removes these cost centers entirely. Additionally, the high catalytic efficiency of the composite lipase system ensures that expensive enzymatic reagents are utilized to their maximum potential, reducing the catalyst loading required per kilogram of product. This lean manufacturing approach results in substantial cost savings in posaconazole intermediate manufacturing by minimizing waste generation and maximizing resource utilization throughout the production line.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that are sensitive to minor variations in raw material quality or environmental conditions. The robustness of this enzymatic route, demonstrated by its successful scale-up from gram to hectogram scales in the patent examples, suggests a high degree of process stability. The use of commercially available and stable enzymes, combined with common industrial solvents like ethyl acetate, mitigates the risk of supply disruptions caused by specialty reagent shortages. Moreover, the simplified workup procedure reduces the dependency on specialized purification equipment, allowing for greater flexibility in manufacturing site selection. This resilience ensures reducing lead time for high-purity pharmaceutical intermediates, enabling suppliers to respond more agilely to fluctuating market demands for antifungal medications.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial waste intensify, the environmental profile of a chemical process becomes a critical factor in vendor selection. This synthesis route significantly lowers the environmental footprint by avoiding the use of heavy metal Lewis acids and reducing the volume of toxic acetonitrile waste. The aqueous workups described in the patent are straightforward and generate waste streams that are easier to treat compared to those containing heavy metal residues. The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, as evidenced by the consistent yields observed during the twenty-fold expansion experiments. This scalability ensures that manufacturers can meet growing global demand for posaconazole without encountering the bottlenecks typically associated with transitioning from laboratory discovery to full-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing production frameworks.
Q: How does the new enzymatic method improve yield compared to traditional Lewis acid catalysis?
A: The novel method utilizes a composite lipase system with Tween 80, achieving an ee value of 99.8% and significantly reducing product loss associated with recrystallization steps found in older Lewis acid methods, which typically yielded less than 70%.
Q: What are the solvent advantages in this synthesis route?
A: The process replaces highly toxic pure acetonitrile with a mixed solvent system of ethyl acetate and acetonitrile. Furthermore, the compatibility of ethyl acetate allows the crude product from Step S1 to proceed directly to Step S2 without solvent exchange, streamlining the workflow.
Q: Is this synthesis route scalable for industrial production?
A: Yes, experimental data demonstrates stable yields when scaling from 5g to 100g batches. The process avoids complex purification between early steps, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Posaconazole Intermediate Supplier
The technological advancements detailed in patent CN115448912A underscore the evolving complexity of modern pharmaceutical synthesis, requiring partners who possess both deep scientific expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is reflected in our stringent purity specifications and rigorous QC labs, which ensure that every batch of posaconazole intermediate meets the exacting standards required by global regulatory bodies. We understand that the transition from a patented laboratory method to a commercial reality involves nuanced engineering challenges, and our team is equipped to navigate these complexities to deliver consistent, high-quality supply.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized enzymatic processes, we can help you achieve significant efficiencies in your supply chain. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the antifungal market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
