Advanced Catalytic Oxidation for Scalable Quinoline API Intermediate Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust, environmentally benign methodologies for constructing privileged heterocyclic scaffolds, among which the quinoline nucleus stands out as a cornerstone structure. As detailed in patent CN111848508A, a groundbreaking approach has been established for the efficient preparation of quinoline compounds through the oxidative dehydrogenation of tetrahydroquinoline precursors. This technology represents a paradigm shift from classical stoichiometric oxidations, leveraging a synergistic catalytic system composed of inexpensive copper salts and N-hydroxyphthalimide (NHPI) under an oxygen atmosphere. For R&D directors and procurement strategists, this innovation offers a compelling value proposition: it transforms a traditionally waste-intensive transformation into a streamlined, atom-economical process that aligns perfectly with modern green chemistry principles while maintaining high yields and exceptional purity profiles suitable for API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline ring has relied heavily on classical condensation reactions such as the Skraup synthesis, which dates back to the 19th century. While effective for simple substrates, these traditional pathways are plagued by severe operational and environmental drawbacks that hinder modern commercial scalability. The Skraup synthesis, for instance, necessitates the use of concentrated sulfuric acid as both a catalyst and dehydrating agent, often requiring nitrobenzene as a harsh oxidant. 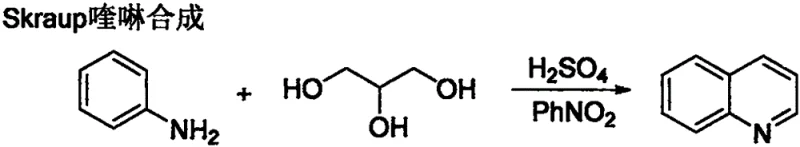 As illustrated in the reaction scheme above, the reliance on strong mineral acids leads to significant equipment corrosion, necessitating expensive specialized reactors and rigorous safety protocols. Furthermore, the generation of large volumes of acidic waste streams creates a substantial burden on wastewater treatment facilities, driving up the total cost of ownership and complicating regulatory compliance. Other methods like the Friedländer or Combes syntheses often suffer from limited substrate tolerance or require high temperatures that can degrade sensitive functional groups, thereby restricting the chemical diversity accessible to medicinal chemists.
As illustrated in the reaction scheme above, the reliance on strong mineral acids leads to significant equipment corrosion, necessitating expensive specialized reactors and rigorous safety protocols. Furthermore, the generation of large volumes of acidic waste streams creates a substantial burden on wastewater treatment facilities, driving up the total cost of ownership and complicating regulatory compliance. Other methods like the Friedländer or Combes syntheses often suffer from limited substrate tolerance or require high temperatures that can degrade sensitive functional groups, thereby restricting the chemical diversity accessible to medicinal chemists.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN111848508A introduces a mild, catalytic oxidative dehydrogenation strategy that circumvents the need for harsh acidic conditions. The core innovation lies in the utilization of molecular oxygen (O2) as the terminal oxidant, coupled with a dual-catalyst system comprising a copper salt and N-hydroxyphthalimide.  As depicted in the general reaction scheme, this approach converts readily available tetrahydroquinolines directly into their aromatic quinoline counterparts with remarkable efficiency. The reaction operates under relatively mild thermal conditions (80-120°C) in common organic solvents like acetonitrile, avoiding the extreme environments that typically lead to byproduct formation. This not only simplifies the reaction setup but also significantly enhances the safety profile of the manufacturing process by eliminating explosive nitrating agents and corrosive acid baths, making it an ideal candidate for reliable quinoline intermediate supplier operations seeking to modernize their production lines.
As depicted in the general reaction scheme, this approach converts readily available tetrahydroquinolines directly into their aromatic quinoline counterparts with remarkable efficiency. The reaction operates under relatively mild thermal conditions (80-120°C) in common organic solvents like acetonitrile, avoiding the extreme environments that typically lead to byproduct formation. This not only simplifies the reaction setup but also significantly enhances the safety profile of the manufacturing process by eliminating explosive nitrating agents and corrosive acid baths, making it an ideal candidate for reliable quinoline intermediate supplier operations seeking to modernize their production lines.
Mechanistic Insights into Copper-Catalyzed Aerobic Oxidation
The mechanistic elegance of this transformation is rooted in the cooperative interaction between the copper catalyst and the organocatalyst NHPI, which facilitates a radical-mediated hydrogen abstraction pathway. In this catalytic cycle, the copper species, preferably cuprous oxide (Cu2O), acts to activate molecular oxygen, generating reactive oxygen species that regenerate the phthalimide-N-oxyl (PINO) radical from NHPI. This PINO radical is the key active species responsible for abstracting hydrogen atoms from the benzylic positions of the tetrahydroquinoline substrate. This radical mechanism is highly selective for the dehydrogenation process, minimizing over-oxidation or ring-opening side reactions that often plague non-selective oxidants. The presence of 4-dimethylaminopyridine (DMAP) further modulates the reaction environment, likely acting as a base to facilitate proton transfer steps or stabilizing intermediate species, thereby accelerating the overall turnover frequency of the catalyst system.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity quinoline derivatives. Because the reaction proceeds via a controlled radical pathway rather than uncontrolled combustion or harsh acid-catalyzed polymerization, the formation of tarry byproducts is significantly suppressed. The mild conditions ensure that sensitive substituents on the aromatic ring, such as halogens or methoxy groups, remain intact throughout the transformation. This chemoselectivity is crucial for pharmaceutical applications where downstream purification can be costly and yield-limiting. By minimizing the generation of complex impurity profiles, this method reduces the burden on analytical QC labs and streamlines the isolation process, ensuring that the final product meets stringent purity specifications required for clinical-grade materials without the need for extensive recrystallization or chromatographic polishing.
How to Synthesize Quinoline Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it highly attractive for process chemistry teams aiming to scale up production. The protocol involves a straightforward one-pot procedure where the tetrahydroquinoline substrate is combined with the catalytic system in a standard reactor. Detailed optimization data indicates that the reaction is robust across a variety of substituted substrates, delivering consistent results. For those interested in the specific operational parameters, molar ratios, and workup procedures validated in the patent examples, the detailed standardized synthesis steps are provided in the guide below.
- Combine tetrahydroquinoline substrate, N-hydroxyphthalimide (NHPI), copper salt catalyst (preferably Cu2O), and 4-dimethylaminopyridine (DMAP) in an organic solvent such as acetonitrile.
- Stir the reaction mixture under an oxygen atmosphere at elevated temperatures ranging from 80°C to 120°C for approximately 12 hours to ensure complete dehydrogenation.
- Filter the reaction mixture, concentrate under reduced pressure, and purify the resulting residue via column chromatography to isolate the high-purity quinoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic oxidation technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from stoichiometric oxidants to catalytic aerobic oxidation fundamentally alters the cost structure of quinoline manufacturing. By replacing expensive and hazardous chemical oxidants with atmospheric oxygen, the process eliminates a major cost driver and reduces the logistical complexity associated with handling dangerous reagents. Furthermore, the use of earth-abundant copper salts instead of precious metals like palladium or platinum ensures that raw material costs remain low and stable, shielding the supply chain from the volatility often seen in the precious metals market.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the drastic simplification of the reaction workflow and waste management. Since the process avoids the use of strong mineral acids and generates minimal toxic waste, the costs associated with neutralization, waste disposal, and equipment maintenance are significantly reduced. The high atom economy of using oxygen as the oxidant means that fewer raw materials are wasted, leading to a more efficient utilization of inputs. Additionally, the high yields reported across diverse substrates minimize the loss of valuable starting materials, directly improving the overall cost-of-goods-sold (COGS) for the final quinoline intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as copper oxide, NHPI, and acetonitrile ensures a robust and resilient supply chain. Unlike specialized reagents that may have single-source suppliers or long lead times, the catalysts and solvents used in this process are widely available from multiple global vendors. This redundancy mitigates the risk of supply disruptions and allows for flexible sourcing strategies. Moreover, the operational simplicity of the reaction reduces the dependency on highly specialized operator skills, facilitating easier technology transfer between manufacturing sites and ensuring consistent production continuity even during periods of high demand.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the inherent safety and mildness of the reaction conditions. The absence of exothermic runaway risks associated with strong oxidants allows for safer operation in large-scale reactors. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding volatile organic compounds (VOCs) and heavy metal discharge. The low metal loading and the ability to potentially recover and recycle the copper catalyst further enhance the sustainability profile, making it easier to obtain necessary environmental permits and maintain a positive corporate social responsibility standing in the eyes of stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's capabilities and limitations for potential partners.
Q: What are the primary advantages of this copper-catalyzed oxidation method over traditional Skraup synthesis?
A: Unlike the traditional Skraup synthesis which requires harsh acidic conditions and generates significant corrosive waste, this method utilizes molecular oxygen as a green oxidant and mild copper catalysis, drastically reducing environmental impact and equipment corrosion risks.
Q: Is this method suitable for large-scale commercial production of quinoline intermediates?
A: Yes, the process is highly scalable due to the use of inexpensive and readily available catalysts (Cu2O) and oxygen gas, eliminating the need for expensive stoichiometric oxidants and simplifying downstream purification processes.
Q: What types of substituents are tolerated on the tetrahydroquinoline ring?
A: The method demonstrates broad substrate scope, successfully tolerating various functional groups including alkyl (methyl), halogens (fluoro, chloro, bromo), and alkoxy (methoxy) groups at multiple positions on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the catalytic oxidation technology described in CN111848508A and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of quinoline intermediate delivered meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Whether you require custom synthesis of novel quinoline derivatives or scale-up of existing routes, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this green oxidation platform can optimize your supply chain and accelerate your time to market.
