Advanced Manufacturing of Levetiracetam via Mild Boc-Activation Cyclization Technology
Introduction to Next-Generation Levetiracetam Manufacturing
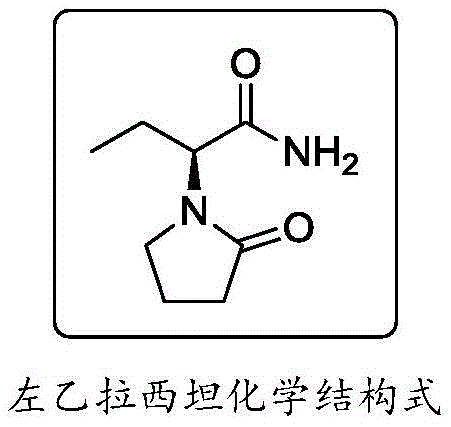
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the production of critical antiepileptic agents, and the synthesis technique disclosed in patent CN109053528B represents a significant leap forward in this domain. This innovative methodology addresses the longstanding challenges associated with traditional Levetiracetam production by introducing a mild, environmentally benign route that utilizes di-tert-butyl dicarbonate ((Boc)2O) as a key activating agent. Unlike conventional processes that rely on hazardous reagents, this approach ensures high finished product quality while drastically simplifying the operational workflow for large-scale manufacturing. By leveraging (S)-2-(4-chlorobutanamide) butyric acid as a chiral starting material, the process inherently bypasses the need for inefficient chemical resolution steps, thereby maximizing atomic economy and overall yield potential. The strategic implementation of pyridine as a base and ammonium salts for amidation creates a robust reaction system that is both cost-effective and compliant with modern green chemistry principles. For global supply chain leaders, this technology offers a compelling value proposition by mitigating regulatory risks associated with toxic solvents like benzene and corrosive acid chlorides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
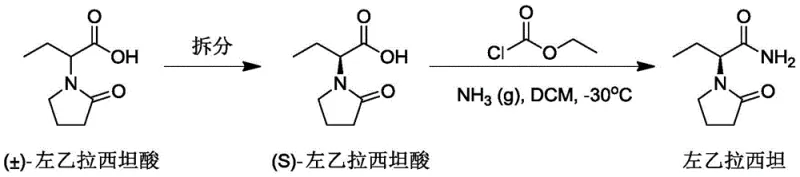
Historically, the industrial synthesis of Levetiracetam has been plagued by significant safety and efficiency bottlenecks that hinder optimal commercial scale-up of complex pharmaceutical intermediates. Early methodologies, such as the resolution route disclosed by UCB, relied heavily on benzene as a solvent, a Class 1 carcinogen that is strictly prohibited under current ICH Q3 guidelines for residual solvents. Furthermore, these legacy processes often necessitated the use of ethyl chloroformate to generate mixed anhydrides, a reagent known for its high toxicity and classification as a controlled tube product in many jurisdictions. Alternative routes involving thionyl chloride or oxalyl chloride for esterification introduce severe corrosion risks to reactor vessels and generate substantial quantities of acidic waste liquid, imposing a heavy burden on environmental compliance teams. The reliance on chemical resolution in some older pathways also inherently caps the maximum theoretical yield at 50%, creating a fundamental economic inefficiency that drives up the cost of goods sold. Additionally, the use of strong acids and harsh conditions in these traditional methods can lead to racemization, compromising the optical purity of the final API and requiring costly downstream purification steps. These cumulative factors create a fragile supply chain vulnerable to regulatory crackdowns and fluctuating raw material costs associated with hazardous chemical handling.
The Novel Approach
In stark contrast to these legacy limitations, the novel synthesis technique presented in CN109053528B offers a streamlined, two-step pathway that fundamentally redefines the manufacturing landscape for this critical antiepileptic drug. The core innovation lies in the activation of the carboxylic acid group using (Boc)2O in the presence of pyridine, which facilitates a gentle yet highly efficient conversion to the corresponding amide without generating toxic byproducts. This method completely eliminates the need for corrosive acid chlorides or toxic chloroformates, thereby removing the requirement for specialized corrosion-resistant equipment and reducing the safety training burden for plant personnel. The subsequent cyclization step is performed under mild alkaline conditions at low temperatures (0-5°C), which preserves the stereochemical integrity of the chiral center and ensures an optical purity of 99.9%. By avoiding the use of benzene and other restricted solvents, the process aligns perfectly with global regulatory standards, facilitating smoother market entry and reducing the risk of batch rejection. The operational simplicity of this route, characterized by straightforward filtration and crystallization workups, translates directly into reduced cycle times and higher throughput capabilities for manufacturing facilities. Ultimately, this approach provides a reliable Levetiracetam supplier with a distinct competitive advantage through superior process safety and environmental sustainability.
Mechanistic Insights into Boc-Mediated Amide Activation and Cyclization
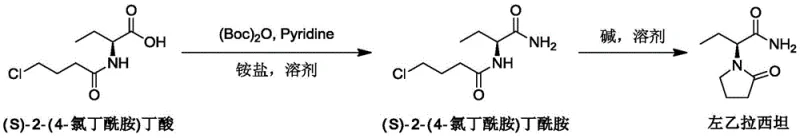
The chemical elegance of this process is rooted in the specific mechanistic pathway utilized for amide bond formation, which avoids the high-energy intermediates typical of acid chloride chemistry. In the first stage, (Boc)2O reacts with the carboxylic acid moiety of the starting material in the presence of pyridine to form a reactive mixed anhydride or activated ester species in situ. This activation step is crucial as it renders the carbonyl carbon sufficiently electrophilic to undergo nucleophilic attack by the ammonia derived from the ammonium salt, yet mild enough to prevent degradation of the sensitive chloroalkyl chain. The use of pyridine serves a dual purpose, acting both as a nucleophilic catalyst to accelerate the formation of the active species and as a base to scavenge the tert-butanol byproduct, driving the equilibrium towards the desired amide. This mechanism stands in sharp contrast to the violent exotherms observed when using thionyl chloride, allowing for precise temperature control between 20-30°C which is vital for maintaining process safety. The resulting (S)-2-(4-chlorobutanamide) butanamide intermediate is formed with high fidelity, setting the stage for the final ring-closing transformation without the introduction of extraneous impurities.
Following the amide formation, the cyclization mechanism proceeds via an intramolecular nucleophilic substitution that is meticulously controlled to preserve chirality. Upon the addition of a strong base such as potassium tert-butoxide at cryogenic temperatures (0-5°C), the amide nitrogen is deprotonated to form a nucleophilic anion. This anion then attacks the terminal carbon bearing the chlorine atom in an SN2-type displacement, closing the five-membered pyrrolidone ring characteristic of Levetiracetam. The low temperature is a critical parameter here, as it suppresses competing elimination reactions or epimerization at the alpha-carbon, which are common failure modes in less optimized processes. The choice of solvent, typically tetrahydrofuran or acetonitrile, plays a pivotal role in solvating the ionic intermediates while maintaining the solubility of the organic substrate. This precise orchestration of reaction conditions ensures that the final product meets stringent purity specifications with minimal need for chromatographic purification, relying instead on simple recrystallization techniques. Such mechanistic control is essential for producing high-purity pharmaceutical intermediates that meet the rigorous demands of global regulatory bodies.
How to Synthesize Levetiracetam Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing multipurpose chemical reactors, requiring only standard agitation and temperature control capabilities. Operators begin by dissolving the chiral starting acid in a suitable solvent like acetonitrile, followed by the controlled addition of the activating agent and base to generate the reactive intermediate. The subsequent addition of ammonium salts triggers the amidation, which proceeds to completion over several hours under mild stirring conditions, after which the intermediate is isolated via a straightforward crystallization from acetone. For the final cyclization, the purified intermediate is redissolved and cooled to near-freezing temperatures before the base is introduced in batches to manage the reaction exotherm effectively. Detailed standardized synthetic steps see the guide below for specific molar ratios and workup procedures that guarantee reproducible high-yield results.
- Activate (S)-2-(4-chlorobutanamide) butyric acid with (Boc)2O and pyridine in acetonitrile at 20-30°C, then react with ammonium bicarbonate to form the diamide intermediate.
- Purify the intermediate by dissolving in acetone at 60-70°C and crystallizing at 10-20°C to obtain high-purity (S)-2-(4-chlorobutanamide) butanamide.
- Perform cyclization by treating the diamide with potassium tert-butoxide in THF at 0-5°C, followed by pH adjustment and recrystallization to yield Levetiracetam.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this manufacturing technology offers profound benefits that extend far beyond simple yield improvements, fundamentally altering the cost structure of Levetiracetam production. By eliminating the reliance on controlled and highly toxic reagents such as ethyl chloroformate and thionyl chloride, manufacturers can significantly reduce the overhead costs associated with hazardous material storage, specialized waste disposal, and regulatory compliance reporting. The absence of benzene and other Class 1 solvents removes a major barrier to market entry in regions with strict environmental laws, ensuring uninterrupted supply continuity for global pharmaceutical partners. Furthermore, the use of inexpensive and readily available ammonium salts as the nitrogen source replaces costly amines or ammonia gas handling systems, contributing to substantial cost savings in raw material procurement. The mild reaction conditions also translate to lower energy consumption for heating and cooling, as the process operates efficiently near ambient temperatures for the majority of the synthesis duration. These factors combine to create a resilient supply chain capable of withstanding market volatility while delivering consistent quality to downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous activating agents like ethyl chloroformate directly lowers the bill of materials, while the simplified workup reduces solvent consumption and waste treatment expenses. By avoiding the need for corrosion-resistant reactors lined with exotic alloys, capital expenditure for new production lines is drastically minimized, allowing for faster ROI on manufacturing assets. The high yield and purity achieved in the crystallization steps mean that less material is lost to mother liquors or reprocessing, further enhancing the overall economic efficiency of the plant. Additionally, the reduced safety risk profile lowers insurance premiums and operational downtime related to safety audits, contributing to a leaner cost structure. This holistic approach to cost reduction ensures that the final API remains competitive in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key reagents, including (Boc)2O and ammonium salts, are commodity chemicals available from multiple global suppliers, reducing single-source dependency risks. The robustness of the process against minor variations in raw material quality ensures consistent batch-to-batch performance, minimizing the risk of production delays due to out-of-spec inputs. Since the process does not generate large volumes of acidic waste, there is less reliance on third-party waste management vendors who may face capacity constraints or regulatory shutdowns. The scalability of the route from kilogram to multi-ton scales has been proven, providing confidence to supply chain heads that volume demands can be met without lengthy process re-validation. This reliability is critical for maintaining just-in-time inventory levels and meeting the tight delivery schedules of major pharmaceutical clients.
- Scalability and Environmental Compliance: The green chemistry credentials of this route facilitate easier permitting and environmental impact assessments, accelerating the timeline for bringing new capacity online. The absence of heavy metals or persistent organic pollutants in the waste stream simplifies effluent treatment, allowing facilities to operate within stricter discharge limits without additional investment in remediation technology. The mild thermal profile of the reactions reduces the load on utility systems, making it easier to scale up production in facilities with limited steam or chilled water capacity. Moreover, the high optical purity achieved without chiral chromatography reduces the solvent intensity of the purification process, aligning with corporate sustainability goals and reducing the carbon footprint of manufacturing. This alignment with environmental, social, and governance (ESG) criteria makes the supply chain more attractive to investors and partners focused on sustainable development.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this advanced synthesis pathway for Levetiracetam. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on process robustness and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for commercial production. The responses highlight the specific advantages in terms of safety, purity, and operational simplicity that distinguish this technology from legacy methods.
Q: Why is the Boc-activation method superior to ethyl chloroformate for Levetiracetam synthesis?
A: The Boc-activation method eliminates the use of highly toxic and corrosive ethyl chloroformate, significantly reducing safety hazards and waste treatment costs while maintaining high optical purity.
Q: Does this process require chiral resolution?
A: No, the process starts directly from chiral (S)-2-(4-chlorobutanamide) butyric acid, avoiding the theoretical 50% yield loss associated with chemical resolution methods.
Q: What are the critical temperature controls for the cyclization step?
A: The cyclization reaction must be strictly controlled between 0-5°C to prevent side reactions and ensure the final optical purity exceeds 99.9%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levetiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to maintain a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against international pharmacopoeia standards. Our expertise in handling chiral intermediates and executing mild cyclization reactions positions us as an ideal partner for companies seeking to optimize their Levetiracetam supply chain. By leveraging our deep technical knowledge, we can help you navigate the complexities of regulatory filings and process validation with confidence.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits of switching to this greener, more efficient manufacturing method. Our experts are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us collaborate to build a more sustainable and profitable future for your antiepileptic drug portfolio.
