Optimizing Meropenem Intermediate Production: A Technical Analysis of Promoter-Assisted Rhodium Catalysis
Optimizing Meropenem Intermediate Production: A Technical Analysis of Promoter-Assisted Rhodium Catalysis
The synthesis of high-value carbapenem antibiotics, particularly meropenem, represents a critical challenge in modern pharmaceutical manufacturing due to the complexity of the beta-lactam scaffold and the stringent purity requirements for clinical applications. Patent CN101891743B introduces a refined methodology for producing key meropenem intermediates, specifically focusing on the transformation of diazo precursors into the final bicyclic structure through a highly efficient, telescoped process. This technical disclosure is of paramount interest to R&D directors and process chemists seeking to optimize the production of 1-beta-methyl carbapenem antibiotics, which are renowned for their stability against beta-lactamases and broad-spectrum activity. By leveraging a dual-catalyst system involving noble metals and specific promoters, the patented route addresses longstanding issues regarding catalyst consumption and process throughput.
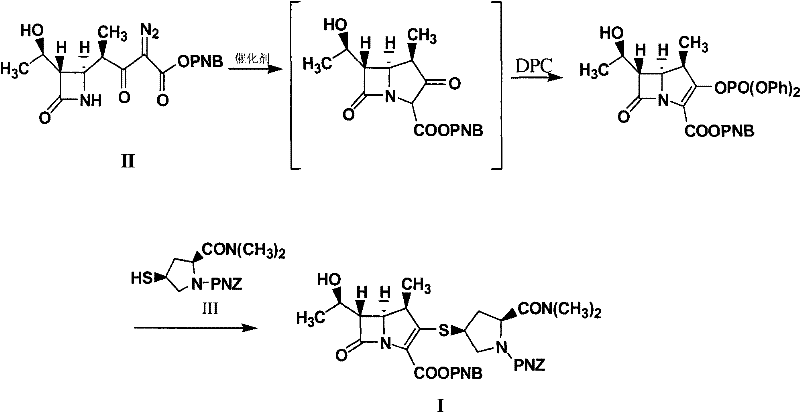
The core innovation lies in the strategic modification of the reaction environment to enhance the efficiency of the rhodium-catalyzed cyclization step. Traditional methods often suffer from incomplete conversions and the necessity for extensive purification between steps, which drives up both operational costs and environmental waste. In contrast, the approach detailed in CN101891743B utilizes a synergistic combination of rhodium catalysts—such as rhodium acetate or rhodium caprylate—alongside promoters like zinc bromide, copper powder, or DMAP. This combination not only accelerates the formation of the critical intermediate (Compound IV) but also ensures a cleaner reaction profile, thereby reducing the burden on downstream purification units. For supply chain leaders, this translates to a more robust and predictable manufacturing timeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques for synthesizing meropenem intermediates typically rely on a stepwise approach where each chemical transformation is treated as a discrete unit operation requiring isolation. In these conventional workflows, the cyclization of the diazo compound is followed by a complete workup, including solvent removal, purification, and recrystallization, before the material is redissolved for the subsequent phosphorylation and coupling reactions. This fragmented approach is inherently inefficient, as it demands significant equipment occupancy time and increases the risk of product degradation during handling. Furthermore, the reliance on high loadings of expensive noble metal catalysts to drive these reactions to completion results in substantial raw material costs and complicates the removal of trace metal impurities, which is a critical quality attribute for pharmaceutical ingredients.
Additionally, the accumulation of impurities in multi-step batch processes often necessitates complex chromatographic separations or repeated crystallizations, which drastically reduce the overall mass yield. The generation of 'three wastes'—wastewater, waste residue, and waste gas—is also exacerbated by the excessive use of solvents required for multiple isolation steps. From a commercial perspective, these inefficiencies create bottlenecks in production capacity and inflate the cost of goods sold (COGS), making it difficult for manufacturers to remain competitive in the generic antibiotic market. The instability of certain intermediates further compounds these issues, as prolonged exposure to ambient conditions during isolation can lead to decomposition.
The Novel Approach
The methodology presented in the patent data offers a paradigm shift by implementing a telescoped synthesis strategy that eliminates intermediate isolation. By maintaining the reaction mixture in solution, the process seamlessly transitions from the rhodium-catalyzed cyclization of Compound II to the formation of the enol phosphate (Compound V) and finally to the coupling with the thiol component (Compound III). This continuous flow of chemistry minimizes the exposure of sensitive intermediates to potentially degrading conditions and significantly reduces the total volume of solvents required. The introduction of promoters plays a pivotal role here, as they activate the catalyst system to operate effectively at lower loadings, ensuring that the reaction proceeds to high conversion without the need for excess reagents.
Moreover, the novel approach incorporates a specific quenching and washing protocol using acidic aqueous solutions, such as phosphoric acid or formic acid, which effectively removes metal residues and basic impurities in a single operation. This streamlined workup procedure not only simplifies the operational workflow but also enhances the purity profile of the final crude product, often allowing for direct crystallization without the need for column chromatography. The result is a process that is not only chemically superior in terms of yield and purity but also operationally leaner, aligning perfectly with the principles of green chemistry and cost-effective manufacturing. The ability to achieve mass yields exceeding 130% (due to the molecular weight increase from coupling) demonstrates the high efficiency of material utilization in this route.
Mechanistic Insights into Rhodium-Catalyzed Cyclization and Coupling
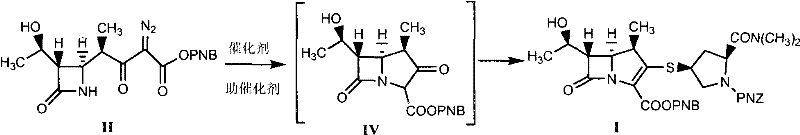
The heart of this synthetic route is the rhodium-catalyzed decomposition of the diazo group in Compound II, which generates a reactive metal-carbene species. This electrophilic intermediate undergoes an intramolecular C-H insertion or cyclization to form the bicyclic beta-lactam core, designated as Compound IV. The presence of promoters, such as zinc halides or copper salts, is hypothesized to coordinate with the carbonyl oxygen or the diazo functionality, thereby stabilizing the transition state and lowering the activation energy for the cyclization. This cooperative catalysis allows the reaction to proceed rapidly even when the concentration of the expensive rhodium species is kept minimal, typically in the range of 0.1‰ to 20‰ molar ratio relative to the substrate. Such precise control over catalyst loading is essential for meeting strict residual metal specifications in the final API.
Following the formation of the ketone intermediate (Compound IV), the reaction mixture is treated with diphenyl phosphate chloride (DPC) in the presence of an organic base. This step converts the ketone into an enol phosphate (Compound V), which serves as an excellent leaving group for the subsequent nucleophilic substitution. The final step involves the addition of Compound III, a thiol-containing pyrrolidine derivative protected with a PNZ group. The thiol attacks the enol phosphate, displacing the phosphate group and establishing the critical carbon-sulfur bond found in meropenem. The entire sequence is conducted under inert atmosphere to prevent oxidation of the sensitive thiol and the beta-lactam ring, ensuring high stereochemical integrity and minimizing the formation of diastereomeric impurities.
How to Synthesize Meropenem Intermediate Efficiently
The synthesis of this complex beta-lactam intermediate requires precise control over temperature and reagent addition to ensure safety and reproducibility. The process begins with the activation of the diazo precursor under reflux conditions, followed by a carefully controlled cooling phase to manage the exothermic phosphorylation and coupling reactions. Operators must monitor the reaction progress using HPLC or TLC to determine the exact endpoints for each stage, ensuring that no unreacted starting material carries over into the final product. The detailed standard operating procedures for scaling this reaction from laboratory to commercial production are outlined below, providing a roadmap for process engineers to implement this technology.
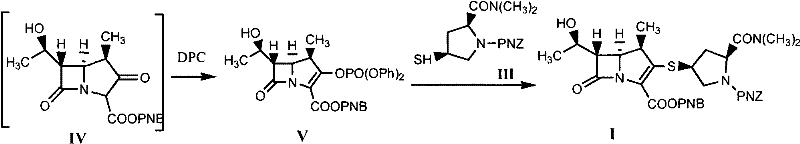
- React Compound II with a noble metal catalyst (e.g., rhodium acetate) and a promoter (e.g., zinc bromide) in an organic solvent under inert gas to form Compound IV.
- Without isolation, add diphenyl phosphate chloride (DPC) and an organic base to the reaction mixture to convert Compound IV into the phosphate intermediate (Compound V).
- Add Compound III and additional organic base to the mixture at controlled low temperatures to finalize the coupling, followed by aqueous workup and crystallization to obtain Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this promoter-assisted synthesis route offers tangible benefits that extend beyond mere chemical yield. The primary advantage is the drastic reduction in the consumption of noble metal catalysts. Rhodium is a precious metal with volatile pricing and supply constraints; by optimizing the catalyst system with inexpensive promoters like zinc bromide, manufacturers can decouple their production costs from the fluctuations of the precious metals market. This strategic adjustment leads to significant cost savings in raw material procurement without compromising the reaction kinetics or product quality, thereby stabilizing the long-term cost structure of the supply chain.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps fundamentally alters the cost equation of the manufacturing process. By removing the need for filtration, drying, and redissolving of intermediates, the facility saves on labor hours, energy consumption for heating and cooling, and solvent usage. Furthermore, the reduced catalyst loading directly lowers the bill of materials. These operational efficiencies compound to deliver a substantially lower cost per kilogram of the final intermediate, enhancing the margin potential for downstream API production. The simplified workflow also reduces the wear and tear on equipment, extending asset life and lowering maintenance overheads.
- Enhanced Supply Chain Reliability: A telescoped process is inherently faster than a stepwise one, as it removes the logistical delays associated with quarantine and testing of intermediates. This acceleration in cycle time allows for quicker turnaround on production batches, enabling suppliers to respond more agilely to market demand spikes. Additionally, the robustness of the reaction, evidenced by the high conversion rates and tolerance to varying conditions, reduces the risk of batch failures. A more reliable production schedule ensures consistent availability of the meropenem intermediate, mitigating the risk of stockouts that could disrupt the broader antibiotic supply network.
- Scalability and Environmental Compliance: The reduction in solvent volume and waste generation aligns with increasingly stringent environmental regulations. Less wastewater and solid waste mean lower disposal costs and a smaller environmental footprint, which is a key metric for sustainable sourcing initiatives. The process is designed to be scalable, with reaction parameters that can be safely translated from pilot plants to multi-ton reactors. The use of common organic solvents like acetonitrile and methylene dichloride, which are readily available in the global chemical market, further secures the supply chain against raw material shortages, ensuring uninterrupted commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific operational parameters and advantages highlighted in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is crucial for assessing the feasibility of integrating this technology into existing manufacturing lines.
Q: How does the addition of promoters affect the rhodium catalyst loading?
A: According to patent CN101891743B, the addition of promoters such as zinc bromide or DMAP allows for a significant reduction in the usage of expensive noble metal catalysts while maintaining or improving reaction conversion rates.
Q: Is intermediate isolation required between the cyclization and coupling steps?
A: No, the process is designed as a telescoped sequence. The reaction solution containing the intermediate (Compound IV) proceeds directly to the phosphorylation and coupling steps without the need for separation, purification, or crystallization, which streamlines the workflow.
Q: What are the typical reaction conditions for this synthesis?
A: The initial cyclization typically occurs under reflux in solvents like methylene dichloride. Subsequent steps require cooling to temperatures ranging from -50°C to 35°C, utilizing organic bases like diisopropylethylamine to facilitate the reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficient production of carbapenem intermediates is vital for the global supply of life-saving antibiotics. Our technical team has extensively analyzed advanced synthetic routes, including the promoter-assisted rhodium catalysis described in CN101891743B, to ensure our manufacturing capabilities are at the forefront of industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity materials. Our rigorous QC labs and stringent purity specifications guarantee that every batch of meropenem intermediate meets the demanding requirements of regulatory agencies worldwide.
We invite pharmaceutical partners to collaborate with us to leverage these advanced synthetic technologies for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized manufacturing processes can enhance your project's economic viability and speed to market. Let us be your strategic partner in delivering high-quality pharmaceutical intermediates with unmatched reliability.
