Advanced Asymmetric Hydrogenation for High-Purity Chiral Dihydroisoflavone Intermediates
Introduction to Breakthrough Chiral Synthesis Technology
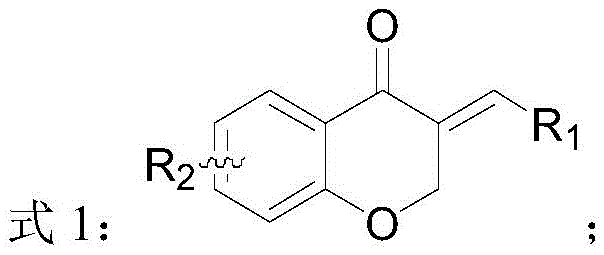
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access biologically active chiral scaffolds, particularly those found in natural products like isoflavones. A significant advancement in this domain is detailed in patent CN111574487A, which discloses a novel preparation method for chiral dihydroisoflavone compounds. This technology addresses a critical gap in synthetic chemistry by enabling the direct asymmetric hydrogenation of isoflavonoids possessing an exocyclic conjugated structure. Unlike previous methods that struggled with selectivity or required complex multi-step sequences, this approach utilizes a specialized chiral catalyst system to achieve high enantioselectivity and yield. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates. The ability to precisely control stereochemistry at the C-3 position of the dihydroisoflavone core opens new avenues for developing potent therapeutic agents with improved biological profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral dihydroisoflavones has been fraught with challenges that hinder efficient commercial production. Traditional strategies often relied on the resolution of racemic mixtures, a process that inherently limits the maximum theoretical yield to 50% and generates substantial chemical waste. Other methods, such as transfer hydrogenation or intramolecular asymmetric hydroacylation, while effective in specific contexts, often suffer from narrow substrate scope or require harsh reaction conditions that are difficult to maintain on a large scale. Furthermore, many existing protocols involve the use of stoichiometric amounts of chiral auxiliaries or expensive reagents, driving up the cost of goods significantly. The lack of a general method for reducing the exocyclic carbon-carbon double bond with high stereocontrol has been a persistent bottleneck, forcing manufacturers to rely on less efficient routes that compromise both purity and throughput.
The Novel Approach
The methodology outlined in the patent introduces a transformative solution by employing direct asymmetric hydrogenation using molecular hydrogen. This approach leverages a sophisticated catalytic system comprising a transition metal, specifically iridium or rhodium, coordinated with a chiral P,N-ligand. By targeting the exocyclic conjugated double bond directly, the process achieves exceptional atom economy, as hydrogen is the only reagent added to the substrate. The reaction conditions are remarkably tunable, operating effectively across a wide temperature range from -40 °C to 120 °C and pressures between 2 and 100 bar. This flexibility allows for optimization based on specific substrate requirements, ensuring high conversion rates and minimizing side reactions. The result is a streamlined synthetic route that not only improves yield but also simplifies downstream purification, making it highly attractive for industrial application.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the unique design of the chiral catalyst, which orchestrates the stereoselective addition of hydrogen to the substrate. The catalyst is formed by the complexation of an iridium precursor, such as [Ir(COD)Cl]2, with a chiral ligand featuring both phosphine and oxazoline moieties. This P,N-ligand architecture creates a rigid chiral environment around the metal center, effectively differentiating the two faces of the prochiral double bond. The steric bulk provided by substituents on the ligand, such as tert-butyl or sec-butyl groups, plays a crucial role in directing the approach of the substrate. Electronic effects are also finely balanced; the nitrogen atom in the oxazoline ring enhances the bonding strength with the metal, stabilizing the active catalytic species throughout the reaction cycle.
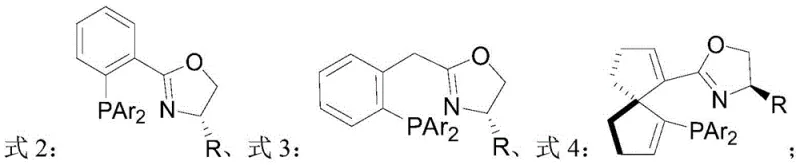
During the catalytic cycle, the substrate coordinates to the metal center, positioning the exocyclic double bond for hydride transfer. The chiral pocket formed by the ligand ensures that hydrogen delivery occurs exclusively from one specific spatial orientation, leading to the formation of a single enantiomer with high fidelity. This mechanism effectively suppresses the formation of unwanted diastereomers or racemic byproducts, which are common impurities in less selective processes. The robustness of the iridium complex allows it to withstand the reaction conditions without significant decomposition, maintaining high turnover numbers. Understanding this mechanistic nuance is vital for process chemists aiming to replicate these results, as slight variations in ligand structure or metal loading can impact the final enantiomeric excess.
How to Synthesize Chiral Dihydroisoflavone Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction parameters to ensure optimal performance. The process begins with the in situ generation or pre-formation of the chiral iridium complex under an inert atmosphere to prevent oxidation of the sensitive metal center. Once the catalyst is ready, the exocyclic conjugated substrate is introduced along with a suitable solvent such as toluene or dichloromethane. The reaction mixture is then subjected to hydrogen pressure in a sealed vessel, where temperature control is critical for balancing reaction rate and selectivity. Monitoring the reaction progress via thin-layer chromatography allows for precise determination of the endpoint, preventing over-reduction or degradation of the product. Detailed standardized synthesis steps for this specific transformation are provided in the guide below.
- Prepare the chiral catalyst by reacting a specific P,N-ligand (such as oxazoline derivatives) with an iridium or rhodium metal precursor in a solvent like dichloromethane under inert gas protection.
- Charge the reaction vessel with the exocyclic conjugated substrate (Formula 1 derivative), the prepared chiral catalyst, and a suitable solvent such as toluene or tetrahydrofuran.
- Perform the hydrogenation reduction reaction under hydrogen pressure (2-100 bar) at controlled temperatures (-40 to 120 °C), monitoring progress via TLC until completion, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of modern supply chain management and cost control. By shifting from resolution-based methods to direct asymmetric synthesis, manufacturers can theoretically double their yield from the same amount of starting material, drastically reducing the raw material burden. The elimination of stoichiometric chiral resolving agents not only lowers material costs but also simplifies waste disposal protocols, contributing to a greener manufacturing footprint. Furthermore, the use of molecular hydrogen as the reducing agent is significantly cheaper and cleaner compared to alternative hydride sources, offering substantial cost savings in reagent procurement. These factors combine to create a more resilient and economical supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the dramatic improvement in atom economy and yield. By avoiding the 50% yield ceiling inherent in racemic resolution, the overall consumption of precious starting materials is significantly reduced. Additionally, the catalyst loading can be kept relatively low, typically around 2.5 wt% of iridium content relative to the substrate, which minimizes the expense associated with noble metals. The simplified workup procedure, often requiring only standard column chromatography, reduces labor hours and solvent usage compared to complex crystallization or extraction sequences needed for other methods. These cumulative efficiencies translate into a lower cost of goods sold, enhancing profit margins for the final API or intermediate.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system contributes to greater supply chain stability by reducing the risk of batch failures. The tolerance of the reaction to various functional groups means that a single catalytic protocol can be applied to a wide range of substrate derivatives, reducing the need for multiple specialized production lines. Moreover, the starting materials, such as substituted chromones and benzaldehydes, are generally commercially available and stable, ensuring a consistent feedstock supply. The ability to operate under moderate pressures and temperatures also means that the process can be executed in standard stainless steel reactors without requiring exotic high-pressure equipment, facilitating easier technology transfer between sites.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the homogeneous nature of the catalytic system and the use of common organic solvents. The reaction generates minimal hazardous byproducts, primarily relying on hydrogen gas which converts to harmless protons upon workup, aligning well with strict environmental regulations. The high enantioselectivity achieved (often exceeding 96% ee) reduces the need for energy-intensive recrystallization steps to upgrade optical purity, further lowering the environmental impact. This compliance with green chemistry principles not only mitigates regulatory risks but also enhances the marketability of the final product to eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific product portfolios. The answers reflect the current state of the art as described in the intellectual property, highlighting both capabilities and operational parameters.
Q: What distinguishes this asymmetric hydrogenation method from traditional isoflavone reduction techniques?
A: Unlike traditional methods that often rely on transfer hydrogenation or resolution of racemic mixtures, this patented approach utilizes direct asymmetric hydrogenation of exocyclic conjugated double bonds. This offers superior atom economy, eliminates the need for stoichiometric reducing agents, and achieves high enantioselectivity (up to 99% ee) using specific iridium-based chiral catalysts.
Q: Can this synthesis method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It operates under relatively mild conditions (temperatures ranging from -40 °C to 120 °C and pressures from 2 to 100 bar) and uses robust catalysts that can be prepared from commercially available ligands and metal salts. The workup involves standard column chromatography, making it feasible for large-scale manufacturing.
Q: What types of substituents are tolerated on the isoflavone substrate?
A: The method demonstrates broad substrate scope, tolerating various substituents at the R1 and R2 positions. These include hydrogen, alkyl, aryl, alkoxy, ester groups, nitro, halogens, cyano, and heterocyclic substituents. Specific examples in the patent include phenyl, methoxyphenyl, fluorophenyl, and tolyl groups, indicating versatility for diverse drug candidate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Dihydroisoflavone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN111574487A for the production of high-value chiral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements. We are committed to leveraging such innovative synthetic routes to deliver cost-effective and high-quality solutions for our global clientele.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for chiral dihydroisoflavone derivatives. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this catalytic hydrogenation route. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your target molecules, ensuring that your project moves forward with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
