Advanced Asymmetric Hydrogenation for High-Purity Chiral Dihydroisoflavone Manufacturing
Introduction to Patent CN111574487B
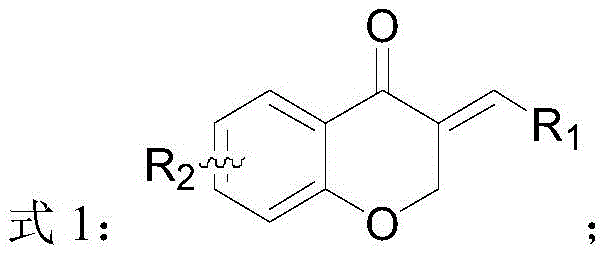
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access bioactive chiral scaffolds, particularly those found in natural products like isoflavones. Patent CN111574487B, published in early 2023, introduces a groundbreaking preparation method for chiral dihydroisoflavone compounds, addressing a long-standing challenge in synthetic organic chemistry. This technology utilizes a highly specific chiral catalyst system to perform asymmetric hydrogenation on exocyclic conjugated compounds, transforming them into valuable chiral isoflavanones with exceptional stereocontrol. The significance of this invention lies in its ability to bypass traditional, inefficient synthetic routes, offering a direct and atom-economical pathway to compounds known for their potent biological activities, including angiogenesis inhibition and estrogen receptor modulation.
The core innovation described in the patent involves the use of transition metal complexes, specifically based on iridium or rhodium, coordinated with novel chiral ligands containing phosphorus and nitrogen donor atoms. This catalytic system enables the precise delivery of hydrogen to the prochiral double bond of the substrate, establishing a new stereogenic center with high fidelity. For R&D directors and process chemists, this represents a significant leap forward, as it provides a robust method to access enantiomerically pure intermediates that were previously difficult to synthesize without resorting to tedious resolution steps. The versatility of the method is demonstrated across a wide range of substrates with varying electronic and steric properties, suggesting broad applicability in the synthesis of diverse pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral dihydroisoflavones has been plagued by inefficiencies inherent in classical resolution techniques and complex multi-step sequences. Traditional approaches often involve the synthesis of racemic mixtures followed by chiral resolution, a process that is fundamentally flawed from a manufacturing perspective because it discards at least 50% of the produced material, leading to poor atom economy and inflated raw material costs. Furthermore, alternative asymmetric methods reported in literature, such as intramolecular asymmetric hydroacylation or asymmetric alkylation, frequently require harsh reaction conditions, expensive stoichiometric chiral auxiliaries, or substrates that are difficult to prepare on a large scale. These limitations create significant bottlenecks for procurement managers and supply chain heads, as the reliance on low-yielding steps increases the overall cost of goods sold (COGS) and extends lead times for critical API intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in CN111574487B leverages direct asymmetric hydrogenation, a reaction type renowned for its cleanliness and high atom economy. By utilizing a tailored iridium-based catalyst system, the method directly reduces the exocyclic carbon-carbon double bond of readily available isoflavone precursors. This eliminates the need for wasteful resolution steps and avoids the use of stoichiometric chiral reagents. The process operates under mild conditions, typically using common solvents like toluene or dichloromethane and moderate hydrogen pressures. This shift from resolution to direct asymmetric synthesis not only simplifies the operational workflow but also drastically improves the theoretical yield ceiling from 50% to nearly 100%. For manufacturing teams, this translates to a streamlined process that requires fewer unit operations, less solvent consumption, and reduced waste generation, aligning perfectly with modern green chemistry principles and cost-reduction mandates.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this transformation hinges on the sophisticated design of the chiral catalyst, which acts as a molecular machine to dictate the stereochemical outcome of the reaction. The catalyst is formed in situ or pre-prepared by coordinating a chiral P,N-ligand (such as those shown in Formulas 2, 3, and 4) with an iridium precursor like [Ir(COD)Cl]2. The ligand architecture is critical; it features a rigid oxazoline ring coupled with a phosphine group, creating a well-defined chiral pocket around the metal center. When the substrate coordinates to the iridium, the bulky substituents on the ligand (such as tert-butyl or sec-butyl groups) exert strong steric influence, blocking one face of the prochiral double bond. This forces the oxidative addition of hydrogen and subsequent migratory insertion to occur exclusively from the less hindered face, thereby ensuring high enantioselectivity.
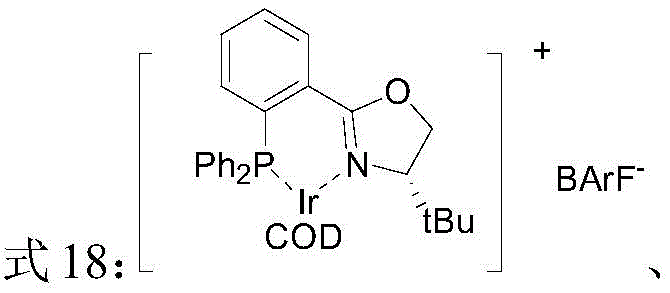
Beyond stereocontrol, the catalyst system exhibits remarkable chemoselectivity, a crucial factor for maintaining product purity. The isoflavone substrate contains both a reducible exocyclic alkene and a ketone carbonyl group. Under the specific conditions optimized in this patent, the iridium catalyst preferentially activates and reduces the C=C bond while leaving the C=O bond intact. This selectivity prevents the formation of over-reduced alcohol byproducts, which would otherwise complicate downstream purification and lower the overall yield. The mechanism likely involves the formation of a cationic iridium-hydride species, stabilized by the weakly coordinating anion (such as BARF-), which facilitates the heterolytic cleavage of hydrogen. This precise control over reactivity ensures that the final product profile is clean, with minimal impurities, reducing the burden on quality control laboratories and ensuring consistent batch-to-batch reproducibility essential for regulatory compliance.
How to Synthesize Chiral Dihydroisoflavone Efficiently
The practical implementation of this technology involves a straightforward protocol that balances reaction efficiency with operational safety. The synthesis begins with the preparation of the active catalyst species, typically involving the mixing of the chiral ligand and the iridium salt in an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. Once the catalyst is activated, the exocyclic conjugated substrate is introduced along with a suitable solvent. The reaction mixture is then subjected to a hydrogen atmosphere in a sealed vessel. Monitoring the reaction progress is easily achieved via thin-layer chromatography (TLC), allowing operators to determine the precise endpoint and prevent over-reaction. Following the completion of the hydrogenation, the crude product is isolated and purified, often using standard silica gel column chromatography, to afford the target chiral dihydroisoflavone in high optical purity.
- Prepare the chiral catalyst by reacting a specific P,N-chiral ligand with an iridium precursor like [Ir(COD)Cl]2 and a non-coordinating anion source in an inert atmosphere.
- Combine the exocyclic conjugated isoflavone substrate with the prepared catalyst in a suitable solvent such as toluene or dichloromethane within a pressure vessel.
- Conduct the hydrogenation reaction under mild hydrogen pressure (2-10 bar) and controlled temperature, followed by purification via column chromatography to isolate the high-enantiopurity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this catalytic technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift from resolution-based methods to direct asymmetric hydrogenation fundamentally alters the cost structure of producing these high-value intermediates. By eliminating the inherent 50% material loss associated with racemic resolution, the effective throughput of the manufacturing plant is doubled without requiring additional capital investment in reactors or infrastructure. This dramatic improvement in material efficiency directly correlates to a significant reduction in the cost of raw materials, which is often the largest component of COGS in fine chemical manufacturing. Furthermore, the use of earth-abundant solvents and the potential for catalyst recycling further contribute to lowering the overall production expenses.
- Cost Reduction in Manufacturing: The elimination of resolution steps removes the need for expensive chiral resolving agents and the associated processing costs for separating and recovering the unwanted enantiomer. Additionally, the mild reaction conditions (low pressure and temperature) reduce energy consumption compared to high-pressure hydrogenation or high-temperature cyclization methods. The high selectivity of the catalyst minimizes the formation of side products, which reduces the complexity and cost of downstream purification processes, leading to substantial overall cost savings in the manufacturing of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically the exocyclic conjugated isoflavones, are generally more accessible and easier to synthesize than the specialized substrates required for other asymmetric methods. This availability ensures a stable supply of feedstock, reducing the risk of production delays due to raw material shortages. Moreover, the robustness of the iridium catalyst system allows for consistent performance across different batches, ensuring reliable delivery schedules to downstream customers. The simplified process flow also reduces the number of potential failure points in the supply chain, enhancing overall operational resilience.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at low hydrogen pressures (2-10 bar), which allows it to be run in standard stainless steel reactors commonly available in multipurpose chemical plants, avoiding the need for specialized high-pressure equipment. From an environmental perspective, the high atom economy and reduced solvent usage align with strict regulatory standards for waste disposal and emissions. The cleaner reaction profile results in less hazardous waste generation, simplifying compliance with environmental regulations and reducing the costs associated with waste treatment and disposal, making it a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and licensees.
Q: What distinguishes this asymmetric hydrogenation method from traditional resolution techniques?
A: Unlike traditional resolution which theoretically limits yield to 50%, this catalytic asymmetric hydrogenation converts the prochiral exocyclic alkene directly into the chiral product with high enantioselectivity (up to 99% ee) and near-quantitative yields, effectively doubling the material efficiency.
Q: Does this catalyst system exhibit chemoselectivity between the C=C double bond and the carbonyl group?
A: Yes, the specific iridium-P,N-ligand complex described in the patent demonstrates excellent chemoselectivity, preferentially reducing the exocyclic carbon-carbon double bond while leaving the ketone carbonyl group intact, which is critical for maintaining the isoflavanone scaffold.
Q: Are the reaction conditions suitable for large-scale industrial production?
A: The process operates under relatively mild conditions, typically requiring low hydrogen pressures (2-10 bar) and moderate temperatures (25-40°C), which significantly reduces safety risks and equipment costs compared to high-pressure hydrogenation processes, facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Dihydroisoflavone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in CN111574487B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly translate such innovative laboratory methods into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage supply is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of chiral dihydroisoflavone meets the highest quality standards required by global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis route for your specific pipeline needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this technology can optimize your supply chain. We encourage you to contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and cost-effective supply of these critical chiral building blocks.
