Advanced Normal-Pressure Catalytic Reduction for High-Purity Serinol Production
The pharmaceutical industry continuously seeks robust synthetic pathways for critical diagnostic agents, and the preparation of serinol stands as a pivotal challenge in the supply chain for iodine-based contrast media. Patent CN102731324A introduces a transformative methodology for synthesizing serinol through normal-pressure catalytic reduction, effectively addressing the safety and cost bottlenecks inherent in legacy technologies. This technical breakthrough utilizes a sophisticated reduction system comprising hydrazine hydrate and a recyclable catalyst, specifically targeting the conversion of 5-nitro-2,2-dimethyl-1,3-dioxane into high-purity amino diols. By shifting away from hazardous high-pressure hydrogenation, this process not only enhances operational safety but also aligns with modern green chemistry principles required by top-tier regulatory bodies. For R&D directors and procurement strategists, understanding this shift is crucial for securing a reliable serinol supplier capable of delivering consistent quality without the volatility associated with high-energy inputs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of serinol has been plagued by severe engineering constraints and safety hazards that inflate operational expenditures. Traditional routes, such as those described in earlier literature by Schmidt and Den Otter, rely heavily on the reduction of nitro compounds using precious metal catalysts like palladium or ruthenium under high-pressure hydrogen atmospheres. As illustrated in the reaction scheme below, these methods necessitate specialized autoclaves capable of withstanding pressures between 3 MPa and 6 MPa, creating a significant barrier to entry for many manufacturers due to the capital intensity of such equipment.
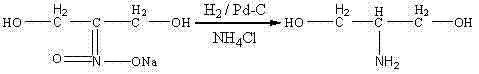
Furthermore, alternative pathways involving sodium metal reductions, while avoiding high-pressure gas, introduce extreme pyrophoric risks and generate substantial quantities of hazardous waste that complicate downstream purification. The reliance on stoichiometric amounts of sodium metal often leads to exothermic runaways if not meticulously controlled, posing a direct threat to plant personnel and continuity of supply. Additionally, the catalysts used in these conventional hydrogenation processes, typically palladium on carbon, suffer from rapid deactivation and are often discarded after a single use, driving up the raw material costs significantly. These cumulative factors render traditional serinol manufacturing economically inefficient and operationally fragile in a modern, safety-conscious production environment.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data leverages a transfer hydrogenation strategy using hydrazine hydrate as the hydrogen donor, operating effectively under normal atmospheric pressure. This method cleverly employs 5-nitro-2,2-dimethyl-1,3-dioxane as the starting material, where the acetone moiety serves as a temporary protecting group for the hydroxyl functions, preventing unwanted side reactions during the reduction phase. The reaction proceeds smoothly at moderate temperatures ranging from 40°C to 85°C, eliminating the need for complex high-pressure infrastructure and allowing for the use of standard glass-lined or stainless steel reactors. By utilizing Raney-Ni or Pd-C in conjunction with hydrazine, the process achieves complete reduction of the nitro group to the amino group with exceptional selectivity, thereby simplifying the impurity profile of the crude product. This paradigm shift represents a major leap forward for cost reduction in pharmaceutical intermediates manufacturing, offering a scalable solution that balances yield, safety, and economic viability.
Mechanistic Insights into Hydrazine-Mediated Catalytic Reduction
The core chemical transformation in this patented process involves the catalytic decomposition of hydrazine hydrate on the surface of the metal catalyst to generate active hydrogen species in situ. Unlike molecular hydrogen which requires high pressure to achieve sufficient concentration on the catalyst surface, hydrazine acts as a liquid hydrogen carrier that decomposes to release nitrogen gas and reactive hydrogen atoms directly at the active sites of the Raney-Ni or Pd-C. This mechanism ensures a steady and controlled supply of reducing equivalents to the nitro group of the dioxane derivative, facilitating a stepwise reduction through nitroso and hydroxylamine intermediates before finally yielding the primary amine. The presence of the acetonide protecting group is critical here, as it stabilizes the molecule against potential over-reduction or cleavage of the carbon-oxygen bonds under the reducing conditions, ensuring the structural integrity of the carbon backbone is maintained throughout the reaction cycle.
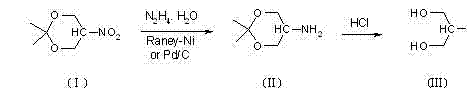
Following the reduction step, the process incorporates a strategic hydrolysis phase using hydrochloric acid to remove the acetone protecting group, revealing the free hydroxyl groups essential for the biological activity of serinol. The choice of acid concentration, typically between 15% and 36%, is optimized to ensure complete deprotection without inducing degradation of the sensitive amino-diol structure. This two-stage sequence—reduction followed by hydrolysis—allows for precise control over the final impurity spectrum, as the intermediate amine can be isolated or processed directly with minimal formation of polymeric by-products often seen in direct nitro-alcohol reductions. The ability to recycle the catalyst for 3 to 4 batches further underscores the efficiency of the catalytic cycle, as the metal surface remains active and resistant to poisoning by the nitrogenous by-products generated during hydrazine decomposition. This mechanistic robustness is key to achieving the reported GC purity levels of 99.5% to 99.8% after simple recrystallization.
How to Synthesize Serinol Efficiently
The synthesis of serinol via this normal-pressure catalytic reduction method offers a streamlined pathway that is highly amenable to industrial scale-up while maintaining rigorous quality standards. The procedure begins with the charging of the reactor with the protected nitro precursor, a suitable alcoholic solvent such as methanol or ethanol, and the selected catalyst, followed by the controlled addition of hydrazine hydrate. Detailed operational parameters, including specific temperature ramps and addition rates, are critical to managing the exotherm associated with hydrazine decomposition and ensuring maximum conversion. For a comprehensive understanding of the exact stoichiometry and workup procedures required to replicate these results in a GMP environment, please refer to the standardized synthesis guide below.
- Charge the reactor with 5-nitro-2,2-dimethyl-1,3-dioxane, solvent (methanol/ethanol), and Raney-Ni catalyst, then heat to 40-85°C.
- Slowly drip hydrazine hydrate (75-85% concentration) into the mixture and maintain reaction temperature for 3-8 hours to reduce the nitro group.
- Filter the catalyst for reuse, then hydrolyze the intermediate with 15-36% hydrochloric acid to remove the protecting group and obtain serinol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this normal-pressure catalytic reduction technology translates into tangible strategic benefits that extend far beyond simple unit cost savings. By eliminating the requirement for high-pressure hydrogenation equipment, manufacturers can significantly reduce capital expenditure (CAPEX) and lower the barrier for expanding production capacity to meet surging demand for contrast agent intermediates. The ability to operate at atmospheric pressure also drastically reduces the regulatory burden and insurance costs associated with handling high-pressure flammable gases, thereby enhancing the overall resilience and continuity of the supply chain against unforeseen operational disruptions. Furthermore, the use of hydrazine hydrate as a liquid reductant simplifies logistics and storage compared to compressed hydrogen gas, allowing for more flexible inventory management and just-in-time delivery models.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the elimination of expensive high-pressure infrastructure and the ability to recycle the catalyst multiple times. Since the catalyst, whether Raney-Ni or Pd-C, can be recovered and reused for 3 to 4 cycles without significant loss of activity, the effective cost per kilogram of catalyst consumption is drastically lowered compared to single-use systems. Additionally, the avoidance of high-pressure hydrogen removes the need for costly compression energy and specialized maintenance protocols, leading to substantial operational expenditure (OPEX) savings over the lifecycle of the production facility. These efficiencies allow suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a distinct advantage in price-sensitive tender processes for bulk pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures that production schedules are less susceptible to delays caused by equipment failure or safety shutdowns common in high-risk hydrogenation units. The starting material, 5-nitro-2,2-dimethyl-1,3-dioxane, is stable and readily available, and the use of common solvents like methanol and ethanol ensures that raw material sourcing is not constrained by niche supply chains. This stability is crucial for long-term supply agreements, as it minimizes the risk of force majeure events related to specialized reagent shortages or equipment downtime, guaranteeing a steady flow of high-purity serinol to downstream API manufacturers. Consequently, partners can rely on consistent lead times and volume availability, which is essential for planning their own production cycles for iopamidol and other diagnostic agents.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers a cleaner profile with reduced hazardous waste generation compared to sodium metal reduction routes. The primary by-product of the reduction is nitrogen gas, which is environmentally benign, and the aqueous waste streams from the hydrolysis step are easier to treat than the complex organic sludges generated by alternative methods. The process is inherently scalable from pilot plant quantities to multi-ton commercial production without the need for re-engineering reaction vessels for higher pressure ratings, facilitating rapid capacity expansion. This alignment with green chemistry principles not only satisfies increasingly strict environmental regulations but also enhances the corporate sustainability profile of the supply chain, a factor of growing importance to global pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this serinol synthesis technology, derived directly from the patent specifications and experimental data. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the safety advantages of this serinol synthesis method compared to traditional hydrogenation?
A: Unlike conventional methods requiring high-pressure hydrogen gas (3-6 MPa), this process operates at normal pressure using hydrazine hydrate, significantly eliminating explosion risks and reducing equipment investment costs.
Q: Can the catalyst be reused in this production process?
A: Yes, the patent explicitly states that the Raney-Ni or Pd/C catalyst can be filtered and recycled for subsequent batches, with optimal performance observed over 3 to 4 reuse cycles.
Q: What purity levels can be achieved with this normal-pressure reduction technique?
A: Through recrystallization of the hydrolyzed product, the method consistently achieves GC purity levels between 99.5% and 99.8%, meeting stringent requirements for diagnostic agent intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Serinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving diagnostic agents, and we are fully equipped to leverage this advanced normal-pressure reduction technology for our clients. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and compliant with international standards. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of serinol meets the exacting requirements for iopamidol synthesis, providing our partners with the confidence they need to secure their supply chains against market volatility.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this safer, more efficient production method. By contacting our technical procurement team, you can request specific COA data from our recent pilot runs and receive detailed route feasibility assessments tailored to your specific volume requirements. Let us demonstrate how our commitment to innovation and quality can drive value for your organization, ensuring a reliable source of high-purity serinol that supports your long-term strategic goals in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
