Advancing Diagnostic Agent Intermediates: Scalable Serinol Production via Safe Catalytic Reduction
Advancing Diagnostic Agent Intermediates: Scalable Serinol Production via Safe Catalytic Reduction
The pharmaceutical industry's relentless pursuit of safer, more efficient synthetic routes for critical diagnostic intermediates has led to significant innovations in process chemistry. A pivotal development in this domain is detailed in patent CN102731324B, which discloses a robust preparation method for serinol (2-amino-1,3-propanediol) utilizing normal-pressure catalytic reduction. Serinol serves as a fundamental building block for the synthesis of iopamidol, a non-ionic X-ray contrast agent widely used globally for urinary tract, vascular, and CT imaging. As the demand for high-quality diagnostic agents continues to surge, the ability to produce key intermediates like serinol with high purity and operational safety becomes a strategic imperative for supply chain stability. This patent introduces a transformative approach that replaces hazardous high-pressure hydrogenation with a milder, hydrazine-based reduction system, offering a compelling value proposition for both R&D teams focused on impurity control and procurement managers seeking cost optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of serinol has been plagued by significant safety hazards and economic inefficiencies associated with traditional reduction technologies. Early methodologies, such as those reported by Schmidt and Den Otter, relied on the condensation of nitromethane and paraformaldehyde followed by reduction using hydrogen gas over precious metal catalysts like palladium-carbon or ruthenium-carbon. These processes inherently require high-pressure equipment to facilitate the hydrogenation of the nitro group, creating substantial capital expenditure burdens and ongoing maintenance costs for specialized high-pressure reactors. Furthermore, the reliance on expensive noble metals increases the variable cost of production, while the handling of high-pressure hydrogen gas introduces severe safety risks, including potential leaks and explosions, which necessitate rigorous and costly safety protocols. Another conventional route described in US4448999 utilizes the sodium salt of 2-nitro-1,3-propanediol under ammonium chloride conditions, yet it still suffers from the requirement of high hydrogenation pressures ranging from 3 to 6 MPa.
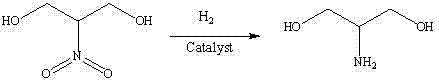
Beyond the safety and equipment constraints, these legacy methods often struggle with catalyst deactivation and difficult purification steps. For instance, alternative routes involving diethyl malonate and metallic sodium reduction, while chemically feasible, present operability challenges due to the large quantities of reactive sodium metal required, which poses fire hazards and complicates waste treatment. The accumulation of by-products in these older synthetic pathways frequently compromises the purity profile of the final serinol, making it difficult to meet the stringent quality specifications required for pharmaceutical-grade iopamidol synthesis. Consequently, there has been a persistent industry need for a manufacturing process that eliminates high-pressure operations, reduces reliance on scarce precious metals, and delivers a cleaner impurity profile without sacrificing yield.
The Novel Approach
The methodology outlined in patent CN102731324B represents a paradigm shift towards safer and more economical serinol manufacturing by leveraging a clever protection-reduction-deprotection strategy. Instead of starting with unprotected polyols that are prone to side reactions, this novel route begins with 5-nitro-2,2-dimethyl-1,3-dioxane, where the hydroxyl groups are masked as an acetonide protecting group. This structural modification allows the subsequent reduction of the nitro group to proceed with high selectivity under normal atmospheric pressure, completely bypassing the need for high-pressure hydrogenation equipment. The reduction is achieved using hydrazine hydrate as the reducing agent in the presence of a catalyst such as Raney-Nickel or Palladium-Carbon. This switch from gaseous hydrogen to liquid hydrazine not only mitigates explosion risks but also simplifies the reactor design, allowing for the use of standard glass-lined or stainless steel vessels commonly found in fine chemical facilities.
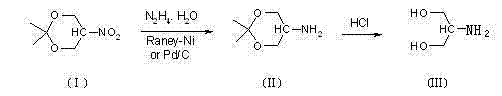
Following the reduction step, the process incorporates a straightforward hydrolysis stage using hydrochloric acid to remove the acetonide protecting group, yielding the target serinol molecule. This two-step sequence (reduction followed by hydrolysis) is highly efficient and generates minimal by-products, ensuring that the final product achieves a GC purity of approximately 99.5% to 99.8% after simple recrystallization. The elimination of high-pressure constraints and the use of readily available, lower-cost reagents position this method as a superior alternative for commercial scale-up. By addressing the core pain points of safety, cost, and purity simultaneously, this technology offers a reliable pathway for establishing a robust supply chain for diagnostic agent intermediates, aligning perfectly with the goals of modern green chemistry and sustainable manufacturing practices.
Mechanistic Insights into Normal-Pressure Hydrazine Reduction
The core chemical transformation in this innovative process is the catalytic reduction of the nitro group (-NO2) to an amino group (-NH2) using hydrazine hydrate (N2H4·H2O). Mechanistically, this reaction proceeds through a transfer hydrogenation pathway where the hydrazine serves as the hydrogen donor. In the presence of the metal catalyst surface (either Raney-Nickel or Pd/C), hydrazine decomposes to generate active hydrogen species which are then transferred to the nitrogen atom of the nitro group. The presence of the acetonide ring in the starting material, 5-nitro-2,2-dimethyl-1,3-dioxane, plays a critical mechanistic role by electronically and sterically shielding the adjacent carbon centers. This protection prevents the reduction of other functional groups or the occurrence of intermolecular condensation reactions that often plague unprotected amino-alcohol syntheses. The reaction conditions are optimized to operate between 40°C and 85°C, a temperature window that balances reaction kinetics with thermal safety, ensuring complete conversion of the nitro substrate without degrading the sensitive dioxane ring prematurely.
Impurity control is intrinsically built into this mechanism through the selectivity of the hydrazine reduction system. Unlike catalytic hydrogenation with H2 gas, which can sometimes lead to over-reduction or hydrogenolysis of C-O bonds under harsh conditions, the hydrazine-mediated process is remarkably chemoselective for the nitro functionality. The subsequent hydrolysis step is equally critical; by using dilute hydrochloric acid (15% to 36% concentration), the acetonide group is cleaved efficiently to regenerate the free hydroxyl groups. This acid-catalyzed hydrolysis is highly specific, minimizing the formation of chlorinated by-products or dehydration products that could arise under more aggressive acidic conditions. The result is a clean reaction profile where the primary impurities are easily removed via recrystallization, ensuring that the final serinol meets the rigorous quality standards necessary for downstream coupling reactions in iopamidol synthesis. This mechanistic elegance translates directly into process reliability, reducing the burden on downstream purification units.
How to Synthesize Serinol Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and catalyst life. The process begins by charging a reactor with the protected nitro precursor, a suitable solvent such as methanol or ethanol, and the chosen catalyst. The mixture is heated to the optimal range of 60°C to 75°C, at which point hydrazine hydrate is added slowly via dropping to control the exotherm and gas evolution (nitrogen). Maintaining the temperature within this narrow window is essential to drive the reaction to completion within 3 to 8 hours while preventing catalyst sintering. Once the reduction is complete, the solid catalyst is filtered off for regeneration and reuse, a key step for economic viability. The filtrate is then concentrated, and the residue is treated with aqueous hydrochloric acid to effect hydrolysis, followed by neutralization and recrystallization to isolate the high-purity serinol product.
- Charge the reactor with 5-nitro-2,2-dimethyl-1,3-dioxane, solvent (methanol/ethanol), and catalyst (Raney-Ni or Pd/C), then heat to 40-85°C.
- Slowly dropwise add hydrazine hydrate (75%-85% concentration) and maintain reaction temperature for 3-8 hours to reduce the nitro group.
- Filter the reusable catalyst, evaporate solvent, and hydrolyze the intermediate with dilute hydrochloric acid to obtain crude serinol, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this normal-pressure catalytic reduction technology offers profound strategic advantages that extend beyond simple unit cost savings. The most immediate impact is the drastic simplification of the manufacturing infrastructure. By eliminating the requirement for high-pressure hydrogenation reactors (which typically operate at 3-6 MPa), facilities can utilize standard atmospheric pressure vessels. This significantly reduces capital expenditure (CapEx) for new production lines and lowers the insurance and maintenance costs associated with high-pressure equipment. Furthermore, the shift away from gaseous hydrogen removes the logistical complexities and safety hazards of storing and handling bulk hydrogen on-site, thereby enhancing overall site safety and reducing regulatory compliance burdens related to hazardous gas management.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the recyclability of the catalyst. The patent explicitly demonstrates that catalysts like Raney-Nickel can be recovered and reused for multiple batches (3 to 4 times) without significant loss of activity. This stands in stark contrast to processes requiring stoichiometric amounts of reducing agents or single-use precious metal catalysts. Additionally, the use of hydrazine hydrate as a liquid reducing agent is generally more cost-effective and easier to handle than high-purity hydrogen gas, leading to substantial reductions in raw material costs. The high yield and purity achieved also minimize waste generation and solvent consumption per kilogram of product, further driving down the total cost of ownership.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically 5-nitro-2,2-dimethyl-1,3-dioxane and hydrazine hydrate, are commodity chemicals with stable global supply chains. Unlike specialized high-pressure catalysts or gases that may be subject to supply disruptions, these inputs are readily available from multiple vendors. The robustness of the reaction conditions—operating at normal pressure and moderate temperatures—also means that production is less susceptible to unplanned shutdowns caused by equipment failure or safety interlocks. This reliability ensures a consistent flow of serinol to downstream manufacturers of iopamidol, securing the supply chain for critical diagnostic imaging agents.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally favorable. The absence of heavy metal waste streams associated with stoichiometric metal reductions (like sodium metal methods) simplifies wastewater treatment. The ability to recycle the catalyst reduces the volume of solid hazardous waste generated. Moreover, the process is inherently scalable; moving from pilot scale to multi-ton commercial production does not require complex engineering changes related to pressure containment. This ease of scale-up allows manufacturers to respond quickly to market demand surges for diagnostic agents, ensuring that supply can match the growing global need for advanced medical imaging capabilities without lengthy lead times for facility upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this serinol synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is critical for R&D teams planning technology transfers and for procurement teams negotiating supply agreements.
Q: Why is the normal-pressure hydrazine reduction method superior to traditional hydrogenation for serinol production?
A: Traditional methods often require high-pressure hydrogenation (3-6 MPa) using expensive precious metal catalysts like Palladium or Ruthenium, posing significant safety risks and equipment costs. The novel normal-pressure method utilizes hydrazine hydrate with reusable Raney-Nickel or Pd/C, eliminating the need for high-pressure vessels and drastically reducing operational hazards while maintaining high yield and purity.
Q: How does the acetonide protecting group strategy improve product quality in this synthesis?
A: The use of 5-nitro-2,2-dimethyl-1,3-dioxane as the starting material effectively protects the two hydroxyl groups during the reduction phase. This prevents unwanted side reactions and ensures that the nitro group is selectively reduced to an amino group without affecting the alcohol functionalities, resulting in a cleaner reaction profile and higher purity final product suitable for diagnostic agents like iopamidol.
Q: Is the catalyst used in this process recyclable, and what represents the cost benefit?
A: Yes, the catalyst (specifically Raney-Nickel or Pd/C) can be filtered and reused for 3 to 4 cycles without significant loss of activity. This recyclability significantly lowers the raw material cost per batch compared to single-use catalytic systems, contributing to substantial overall cost reduction in manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Serinol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes like the normal-pressure catalytic reduction of serinol is vital for the future of the diagnostic agent industry. As a leading CDMO and supplier, we possess the technical expertise to not only supply high-purity serinol but also to assist partners in optimizing their own manufacturing processes based on these advanced methodologies. Our facilities are equipped to handle diverse synthetic pathways, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of serinol meets the exacting standards required for the synthesis of iopamidol and other critical pharmaceutical intermediates.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Whether you are looking to secure a long-term supply of high-purity serinol or need assistance in evaluating the feasibility of this specific reduction route for your internal production, our team is ready to support you. Please contact our technical procurement team to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and detailed route feasibility assessments to help you make informed decisions that drive efficiency and reduce costs in your diagnostic agent manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
