Scalable Manufacturing of L-Erythro-Biopterin via Odorless Thiol Technology
The pharmaceutical industry continuously seeks robust and environmentally benign pathways for the synthesis of critical active pharmaceutical ingredients (APIs) and their intermediates. A significant advancement in this domain is detailed in patent CN101792445B, which discloses a novel preparation method for L-erythro-biopterin. This compound serves as a pivotal intermediate for the clinical drug (R)-2-amino-6-[(1R,2S)-1,2-dihydroxypropyl]-5,6,7,8-tetrahydro-4(3H)-pteridone hydrochloride, commonly known as Sapropterin hydrochloride, used effectively in treating atypical hyperphenylalaninemia. The disclosed technology represents a paradigm shift from traditional methodologies by addressing long-standing issues related to odor, safety, and operational complexity. By leveraging a unique thioacetal protection strategy using long-chain alkyl mercaptans, this process not only optimizes the chemical workflow but also aligns with modern green chemistry principles, making it an attractive option for reliable pharmaceutical intermediates supplier networks aiming for sustainable growth.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of L-erythro-biopterin has relied heavily on routes originating from L-rhamnose, which involves conversion to 5-deoxy-L-arabinose through a series of cumbersome steps. A critical bottleneck in these conventional processes, as illustrated in prior art such as the method reported by Taylor E.C., is the utilization of ethanethiol for thioacetal formation. Ethanethiol is notorious for its unbearable, pervasive stench, which necessitates the installation of specialized and costly deodorization equipment in manufacturing facilities. Furthermore, its low boiling point introduces significant stability challenges during reaction and workup phases, complicating the engineering controls required for safe handling. These factors collectively extend the production cycle and inflate the capital expenditure associated with facility maintenance, creating a substantial barrier for cost reduction in pharmaceutical intermediates manufacturing.
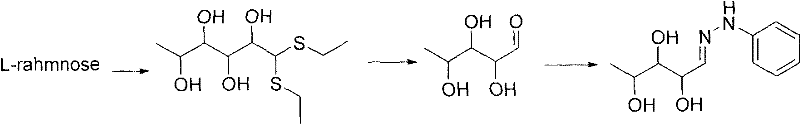
The Novel Approach
In stark contrast, the innovative methodology presented in the patent data circumvents these drawbacks by employing alkyl mercaptans with a carbon chain length of nine or greater. This strategic substitution fundamentally alters the physical properties of the reaction system; the higher molecular weight thiols are effectively odorless, thereby eliminating the immediate need for complex scrubbing systems and drastically improving the occupational health and safety profile of the plant. The process initiates with the direct acetalization of L-(+)-arabinose, bypassing the degradation steps required for L-rhamnose. This streamlined approach ensures milder reaction conditions and a shorter overall cycle time. By integrating sulfonylation and reductive deoxygenation steps that are highly selective, the new route achieves high purity profiles essential for downstream API synthesis, positioning it as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
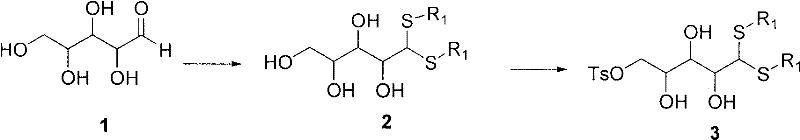
Mechanistic Insights into Thioacetal-Mediated Side Chain Construction
The core chemical ingenuity of this process lies in the manipulation of the carbohydrate side chain using robust protecting group chemistry. The initial reaction involves the condensation of L-(+)-arabinose with a long-chain alkyl mercaptan in the presence of concentrated hydrochloric acid. This forms a dithioacetal intermediate which serves a dual purpose: it protects the aldehyde functionality from unwanted oxidation and activates the adjacent positions for subsequent functionalization. The use of a long hydrophobic chain (CnH2n+1, n≥9) not only mitigates odor but also enhances the solubility characteristics of the intermediate in organic solvents like ethyl acetate, facilitating efficient extraction and purification. This solubility advantage is critical for removing inorganic salts and polar impurities early in the synthesis, ensuring a cleaner substrate for the subsequent cyclization steps.
Following the formation of the thioacetal, the process employs a sulfonylation-reduction sequence to precisely install the required stereochemistry and oxygenation pattern. The introduction of a sulfonyl group, typically using p-toluenesulfonyl chloride, activates a specific hydroxyl group for reductive removal. Subsequent treatment with reducing agents such as lithium aluminum hydride or sodium borohydride selectively cleaves the sulfonate ester without disturbing the sensitive thioacetal moiety. This chemoselectivity is paramount for maintaining the integrity of the chiral centers derived from the natural sugar pool. Finally, the deprotection of the thioacetal under acidic conditions regenerates the aldehyde, which is then condensed with phenylhydrazine to form a hydrazone. This hydrazone acts as a nucleophilic partner in the final condensation with 2,4,5-triaminopyrimidinone, closing the pteridine ring system to yield the target biopterin scaffold with high fidelity.
How to Synthesize L-Erythro-Biopterin Efficiently
The synthesis of L-erythro-biopterin via this patented route involves a carefully orchestrated sequence of nine distinct chemical transformations, beginning with the readily available chiral pool material L-(+)-arabinose. The process is designed to maximize atom economy while minimizing environmental impact through the use of non-volatile reagents. Operators must pay close attention to the stoichiometry of the alkyl mercaptan and the temperature control during the acetalization phase to ensure complete conversion. The subsequent sulfonylation and reduction steps require anhydrous conditions and precise pH control during workup to prevent hydrolysis of sensitive intermediates. For a comprehensive understanding of the specific reaction parameters, solvent choices, and purification techniques required to replicate this high-efficiency pathway, please refer to the standardized technical guide below.
- React L-(+)-arabinose with long-chain alkyl mercaptan (n≥9) in concentrated hydrochloric acid to form the thioacetal intermediate.
- Perform sulfonylation with sulfonyl chloride followed by reductive removal of the sulfonate group using lithium aluminum hydride or borohydride.
- Deprotect the thioacetal to obtain 5-deoxy-L-arabinose, react with phenylhydrazine, acylate, and finally cyclize with 2,4,5-triaminopyrimidinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemical yield. The primary value driver is the significant simplification of the manufacturing infrastructure. By eliminating the requirement for specialized deodorization units associated with ethanethiol handling, facilities can reduce both capital investment and ongoing operational maintenance costs. This reduction in engineering complexity translates directly into lower overheads per kilogram of produced intermediate. Furthermore, the use of stable, high-boiling-point thiols reduces the risk of raw material loss due to evaporation, ensuring better mass balance and more predictable inventory management. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without the bottlenecks typical of hazardous chemical processing.
- Cost Reduction in Manufacturing: The economic impact of switching to odorless long-chain thiols is profound. Traditional methods incur hidden costs related to environmental compliance, specifically the energy and maintenance required to scrub noxious gases from the exhaust stream. By removing this necessity, the process inherently lowers the utility burden on the production site. Additionally, the streamlined reaction sequence reduces the total number of unit operations, which minimizes labor hours and solvent consumption. The ability to perform extractions and filtrations more efficiently due to improved solubility profiles further drives down the cost of goods sold (COGS), allowing for more competitive pricing structures in the global marketplace for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the regulatory hurdles associated with malodorous chemicals. This new method utilizes alkyl mercaptans that are easier to store and transport in bulk quantities without triggering stringent hazardous material protocols applicable to volatile sulfur compounds. Moreover, the starting material, L-(+)-arabinose, is a commodity chemical with a stable global supply, unlike some specialized sugar derivatives that may face sourcing volatility. This robustness in raw material sourcing ensures that production schedules remain uninterrupted, providing downstream API manufacturers with the confidence of consistent delivery timelines and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden inefficiencies, particularly regarding heat transfer and waste management. The mild reaction conditions described in this patent, operating generally between 0°C and 100°C, are highly amenable to standard stainless steel reactor setups without the need for exotic alloys or cryogenic cooling. The waste stream is also significantly cleaner, lacking the heavy sulfur contamination typical of ethanethiol processes, which simplifies wastewater treatment and disposal. This environmental compatibility not only facilitates faster regulatory approvals for new manufacturing sites but also aligns with the increasingly strict sustainability mandates of multinational pharmaceutical corporations, ensuring long-term viability of the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-erythro-biopterin synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or for procurement specialists assessing the risk profile of this supply chain. The answers reflect the practical realities of running this chemistry at an industrial scale, focusing on safety, efficiency, and product quality assurance.
Q: Why is long-chain alkyl mercaptan preferred over ethanethiol in this synthesis?
A: Long-chain alkyl mercaptans (carbon number ≥ 9) are odorless compared to the unbearable stench of ethanethiol. This eliminates the need for expensive deodorization equipment and significantly improves the working environment and operational safety.
Q: What is the primary starting material for this novel L-erythro-biopterin process?
A: The process utilizes L-(+)-arabinose as the primary chiral pool starting material, which undergoes acetalization and subsequent functional group transformations to build the side chain required for the pteridine ring closure.
Q: Does this method offer advantages for commercial scale-up?
A: Yes, the reaction conditions are mild, the cycle time is short, and the operation is easy. The elimination of volatile, malodorous reagents simplifies the engineering controls required for large-scale production, enhancing overall supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Erythro-Biopterin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this odorless thiol technology are fully realized in practice. Our state-of-the-art facilities are equipped to handle complex multi-step syntheses with stringent purity specifications, supported by rigorous QC labs that employ advanced analytical techniques to verify identity and potency. We are committed to delivering L-erythro-biopterin that meets the exacting standards required for the synthesis of life-saving medications like Sapropterin hydrochloride.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and reliability in your pharmaceutical manufacturing operations. Let us collaborate to bring high-quality, cost-effective intermediates to the market faster.
