Advanced Manufacturing of Citalopram Intermediates via Novel Grignard Cyclization
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable routes for the production of high-volume antidepressants like Citalopram. Patent CN1339436A introduces a transformative methodology that fundamentally alters the synthetic landscape for this critical active pharmaceutical ingredient (API). By shifting the synthetic strategy from traditional halogenated precursors to a carboxylic acid-based approach, this technology addresses long-standing challenges regarding toxicity, waste management, and raw material availability. The core innovation lies in the utilization of 5-carboxy-2-benzo[c]furanone as a pivotal starting scaffold, which undergoes sequential Grignard additions followed by a sophisticated dehydration sequence to yield the target nitrile functionality without the need for hazardous cyanide salts. This report analyzes the technical depth of this patent to provide R&D directors, procurement managers, and supply chain heads with a clear understanding of its commercial viability and strategic value.
As a leading entity in the fine chemical sector, understanding the nuances of such proprietary processes is essential for maintaining a competitive edge. The disclosed method not only simplifies the molecular construction of the phthalane core but also offers distinct advantages in impurity profiling, which is a critical parameter for regulatory compliance in global markets. For organizations aiming to secure a reliable API intermediate supplier partnership, mastering this chemistry represents a significant opportunity to diversify supply sources while adhering to stricter environmental, health, and safety (EHS) standards. The following sections will dissect the mechanistic advantages and commercial implications of adopting this novel pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Citalopram has relied heavily on pathways described in earlier patents such as US4136193 and DE2657013, which typically involve the introduction of the nitrile group via a Rosenmund-von Braun type reaction using cuprous cyanide (CuCN). This traditional approach presents severe drawbacks for modern large-scale manufacturing, primarily due to the extreme toxicity and handling difficulties associated with cyanide salts. The use of stoichiometric amounts of copper cyanide generates substantial quantities of heavy metal waste, necessitating complex and costly purification steps to ensure the final API meets stringent residual metal limits set by pharmacopeias. Furthermore, the precursor materials often involve 5-bromo derivatives, which can be more expensive and less atom-economical compared to carboxylic acid analogues. The reliance on concentrated sulfuric acid for cyclization in some older methods also poses significant corrosion and safety risks, limiting the throughput capacity of standard glass-lined reactors and increasing maintenance costs for production facilities.
The Novel Approach
In stark contrast, the methodology outlined in CN1339436A circumvents these issues by employing a 5-carboxy intermediate that is converted to the nitrile through a mild dehydration protocol involving sulfonamides and dehydrating agents like thionyl chloride or phosphorus oxychloride. This strategic shift eliminates the requirement for cuprous cyanide entirely, thereby removing a major bottleneck related to hazardous waste disposal and operator safety. The process leverages the inherent reactivity of the carboxylic acid group, which is activated in situ to form an intermediate species capable of undergoing dehydration to the nitrile under controlled thermal conditions. By avoiding the harsh conditions of traditional cyanation, this new route significantly reduces the formation of undesired by-products and tar, leading to cleaner reaction profiles and higher crude purity. This advancement is particularly relevant for cost reduction in pharmaceutical manufacturing, as it simplifies the downstream processing train and reduces the load on wastewater treatment systems.
Mechanistic Insights into Grignard Addition and Sulfonamide-Mediated Dehydration
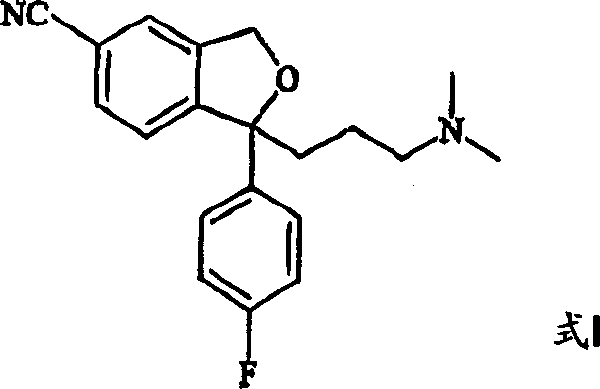
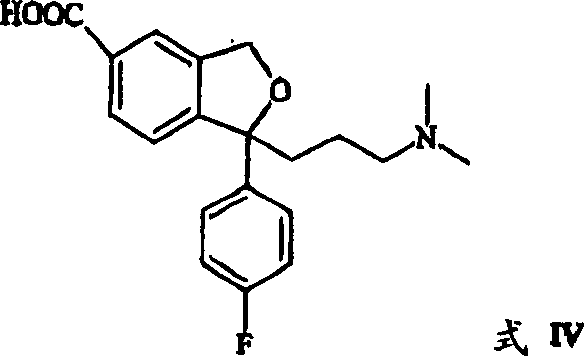
The heart of this synthetic innovation is the dual Grignard addition to the lactone carbonyl of 5-carboxy-2-benzo[c]furanone. The first equivalent of Grignard reagent, typically 4-fluorophenylmagnesium bromide, attacks the lactone to open the ring and form a ketone intermediate, which is not isolated. Subsequently, a second Grignard reagent, 3-(N,N-dimethylamino)propylmagnesium chloride, adds to the ketone to generate a tertiary alcohol diol species (Formula XI). This one-pot telescoping of two Grignard reactions is highly efficient, minimizing solvent usage and exposure of reactive intermediates to the atmosphere. The subsequent acid-catalyzed cyclization closes the phthalane ring to yield the 5-carboxy-Citalopram intermediate (Formula IV), a stable and isolable solid that serves as a crucial quality control checkpoint in the synthesis.
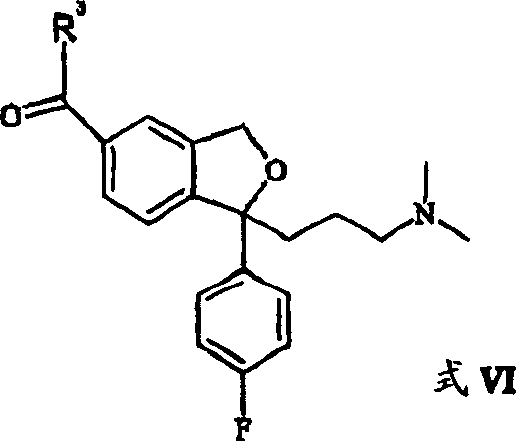
The conversion of this carboxylic acid intermediate to the final nitrile is achieved through a mechanistically elegant dehydration sequence. The patent describes activating the carboxylic acid (Formula IV) using a dehydrating agent to form an activated derivative (Formula VI), such as an acid chloride or mixed anhydride. This activated species then reacts with a sulfonamide, preferably sulfamide (H2N-SO2-NH2), to form a transient intermediate that readily eliminates sulfur dioxide and ammonia upon heating to yield the nitrile group. This mechanism avoids the direct use of cyanide ions, relying instead on the elimination of small gaseous molecules to drive the equilibrium forward. The use of solvents like sulfolane or acetonitrile facilitates heat transfer during this exothermic step, ensuring uniform reaction kinetics and preventing localized hot spots that could degrade the product. This level of mechanistic control is vital for producing high-purity pharmaceutical intermediates consistently.
How to Synthesize 5-Carboxycitalopram Efficiently
The synthesis of the key 5-carboxy intermediate and its subsequent conversion to Citalopram requires precise control over stoichiometry, temperature, and addition rates to maximize yield and minimize impurities. The process begins with the suspension of 5-carboxy-2-benzo[c]furanone in anhydrous THF under an inert nitrogen atmosphere, followed by the careful addition of ligands like TMEDA to enhance the reactivity of the Grignard reagents. The sequential addition of the aryl and alkyl Grignard reagents must be monitored closely, typically via HPLC or TLC, to ensure complete consumption of the starting lactone before proceeding to the cyclization step. Once the diol is formed, acidification triggers the ring closure to Formua IV, which can be isolated or carried forward directly. The final dehydration step involves dissolving the carboxy-intermediate in a high-boiling solvent like sulfolane, adding sulfamide and thionyl chloride, and heating the mixture to approximately 130°C to effect the transformation. Detailed standardized operating procedures for these steps are critical for technology transfer.
- React 5-carboxy-2-benzo[c]furanone sequentially with 4-halofluorophenyl Grignard reagent and 3-halo-N,N-dimethylpropylamine Grignard reagent.
- Perform ring-closure of the resulting diol intermediate (Formula XI) to form the 5-carboxy derivative (Formula IV) using acid catalysis.
- Convert the 5-carboxy compound to Citalopram (Formula I) via activation with a dehydrating agent and reaction with a sulfonamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the process described in CN1339436A offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the decoupling of the supply chain from volatile cyanide markets and the regulatory burdens associated with transporting and storing highly toxic cyanide salts. By utilizing 5-carboxy-2-benzo[c]furanone, which can be derived from abundant feedstocks like terephthalic acid, manufacturers can secure a more stable and predictable raw material base. This stability translates directly into enhanced supply chain reliability, as the risk of production stoppages due to hazardous material shortages or regulatory inspections is significantly mitigated. Furthermore, the elimination of copper cyanide removes the need for expensive heavy metal scavenging resins and complex filtration units, leading to substantial capital expenditure (CAPEX) savings for new plant installations or retrofits.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the purification workflow. Traditional cyanation routes often require multiple recrystallizations or chromatographic steps to remove trace copper and cyanide residues to meet ICH Q3D guidelines. In contrast, the sulfonamide-mediated dehydration produces volatile by-products (SO2, NH3) that are easily removed, resulting in a crude product of significantly higher purity. This reduction in downstream processing steps lowers utility consumption, solvent recovery costs, and labor hours per kilogram of API produced. Additionally, the potential for a "one-pot" synthesis, where the intermediate Formula IV is not isolated but converted directly to Citalopram, further compresses the manufacturing cycle time and reduces solvent inventory requirements, driving down the overall cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The starting material, 5-carboxy-2-benzo[c]furanone, is accessible via established industrial chemistry involving terephthalic acid and formaldehyde, commodities that are produced on a massive global scale with robust supply networks. This contrasts with specialized halogenated precursors that may have limited suppliers. By anchoring the synthesis on widely available carboxylic acid building blocks, the risk of supply disruption is minimized. Moreover, the process conditions (using THF, sulfolane, standard Grignard reagents) are compatible with multi-purpose pharmaceutical manufacturing suites, allowing for flexible production scheduling. This flexibility ensures that commercial scale-up of complex pharmaceutical intermediates can be executed rapidly to meet fluctuating market demand without the need for dedicated, single-use infrastructure.
- Scalability and Environmental Compliance: From an EHS perspective, this route is vastly superior. The absence of cyanide eliminates the generation of cyanide-containing wastewater, which requires specialized oxidation treatment before discharge. The gaseous by-products of the dehydration step can be scrubbed using standard caustic scrubbers, simplifying waste gas management. The process inherently generates less hazardous solid waste, aligning with green chemistry principles and reducing disposal costs. Scalability is further supported by the exothermic nature of the Grignard and dehydration steps, which can be managed effectively in large-scale reactors using jacketed cooling and controlled dosing strategies. This makes the technology ideal for reducing lead time for high-purity pharmaceutical intermediates while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Citalopram synthesis route. These insights are derived directly from the experimental data and claims within patent CN1339436A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into their existing portfolios or sourcing strategies. The answers reflect the specific advantages of the carboxy-intermediate pathway over legacy technologies.
Q: What is the primary advantage of using 5-carboxy-2-benzo[c]furanone over 5-bromo derivatives?
A: Using 5-carboxy-2-benzo[c]furanone eliminates the need for toxic cuprous cyanide (CuCN) in the final cyanation step, significantly improving the environmental profile and reducing heavy metal contamination risks in the final API.
Q: How does the novel dehydration method improve process safety?
A: The patent describes using sulfonamides (like sulfamide) with dehydrating agents like thionyl chloride or POCl3, which offers better control over the exothermic reaction compared to traditional concentrated sulfuric acid methods, enhancing operational safety.
Q: Can this process be performed as a one-pot synthesis?
A: Yes, the patent explicitly discloses embodiments where the preparation of the Formula IV intermediate and its subsequent conversion to Citalopram are carried out without isolation, streamlining the workflow and reducing solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Supplier
The technological advancements detailed in this report underscore the complexity and sophistication required to manufacture high-quality antidepressant intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly transitioned to industrial manufacturing. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles in accordance with global regulatory standards.
We invite potential partners to engage with us to explore how this optimized synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic gains of switching to this cyanide-free process. We encourage you to contact our technical procurement team to obtain specific COA data for our Citalopram intermediates and to discuss route feasibility assessments tailored to your specific production requirements. Let us collaborate to build a more sustainable and efficient future for pharmaceutical manufacturing.
