Advanced Citalopram Synthesis: Leveraging Novel Ketone Intermediates for Commercial Scalability
Introduction to Advanced Citalopram Manufacturing Technologies
The pharmaceutical landscape for antidepressant manufacturing is constantly evolving, driven by the need for safer, more efficient, and environmentally sustainable processes. A pivotal advancement in this domain is detailed in patent CN1550497A, which outlines a novel preparation method for Citalopram, a widely prescribed selective serotonin reuptake inhibitor (SSRI). This technology represents a significant departure from legacy synthetic routes that often rely on hazardous reagents and multi-step sequences with lower atom economy. By focusing on the strategic use of protected ketone intermediates, specifically compounds of Formula IV, this methodology enables a streamlined single-step Grignard reaction that spontaneously cyclizes to form the critical isobenzofuran core. For global procurement leaders and R&D directors, understanding this shift is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials with reduced supply chain risk. The implications of this patent extend beyond mere chemical curiosity; they offer a tangible pathway for cost reduction in API manufacturing through simplified processing and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Citalopram has been plagued by significant chemical and operational challenges that impact both cost and environmental compliance. Traditional methods, such as those disclosed in early patents like DE 2,657,271, often necessitate the use of cuprous cyanide to introduce the essential cyano group at the 5-position of the isobenzofuran ring. The handling of cuprous cyanide presents severe safety hazards due to its high toxicity and the complex waste treatment protocols required to manage cyanide-containing effluents. Furthermore, alternative legacy routes involving the reaction of 5-bromo-2-benzo[c]furanone with Grignard reagents typically require multiple distinct steps, including separate alkylation and dehydration phases. These multi-stage processes inherently accumulate yield losses at each transition, increase solvent consumption, and complicate the purification of the final active pharmaceutical ingredient (API). The reliance on harsh dehydrating agents like strong sulfuric acid in older cyclization methods also introduces risks of side reactions and degradation, leading to difficult-to-remove impurities that compromise the quality of the high-purity pharmaceutical intermediates required for regulatory approval.
The Novel Approach
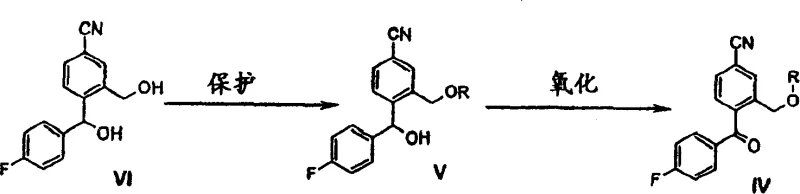
In stark contrast, the methodology described in CN1550497A introduces a robust and elegant solution by utilizing a protected ketone intermediate, designated as Formula IV, where R represents a versatile protecting group such as an acyl or sulfonyl moiety. This innovative route bypasses the need for toxic cyanide exchanges entirely by constructing the carbon skeleton prior to the final cyclization event. The core of this novelty lies in the reaction of Formula IV with 3-(N,N-dimethylamino)propyl magnesium halide. Unlike previous methods that required isolated dehydration steps, this reaction proceeds with a spontaneous ring closure, directly yielding the Citalopram structure in a single operational phase. This consolidation of steps not only accelerates the production timeline but also drastically reduces the volume of organic solvents and reagents needed. The versatility of the protecting group R allows manufacturers to optimize reaction conditions for specific scale-up requirements, ensuring consistent quality. By adopting this approach, a reliable pharmaceutical intermediates supplier can offer a product with a superior impurity profile, free from heavy metal contaminants associated with copper-catalyzed cyanation.
Mechanistic Insights into Grignard Addition and Spontaneous Cyclization
The chemical elegance of this process is rooted in the specific reactivity of the protected ketone intermediate towards organomagnesium reagents. When the compound of Formula IV is treated with 3-(N,N-dimethylamino)propyl magnesium halide, the Grignard reagent attacks the carbonyl carbon to form a tertiary alcohol intermediate. In conventional syntheses, this alcohol would require a separate, energy-intensive dehydration step using acidic catalysts to induce ring closure. However, in this novel system, the presence of the ortho-hydroxymethyl group (protected as an ester or ether) facilitates an intramolecular nucleophilic attack immediately following the Grignard addition or during the workup phase. This spontaneous cyclization mechanism effectively locks the molecular conformation into the desired 1,3-dihydroisobenzofuran structure without the need for external dehydrating agents. This mechanistic feature is critical for R&D teams focused on commercial scale-up of complex pharmaceutical intermediates, as it removes a potential bottleneck where yield losses and impurity formation are most common. The stability of the intermediate during this transformation ensures that the reaction can be run at controlled temperatures, typically ranging from 0°C to ambient conditions, further enhancing process safety and reproducibility.
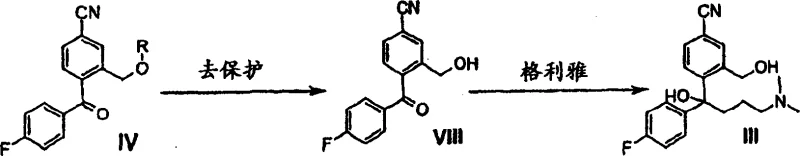
Furthermore, the patent elucidates a sophisticated strategy for managing chirality, which is increasingly relevant given the market demand for enantiomerically pure drugs like Escitalopram. The process allows for the conversion of specific enantiomers of Formula III back into the achiral ketone Formula IV, which can then be recycled into the racemic pool or directed towards specific enantiomeric synthesis. This capability is underpinned by oxidative cleavage methods using agents such as permanganate or ozone, which cleanly convert the chiral alcohol back to the ketone without destroying the sensitive cyano functionality. For quality assurance professionals, this means the impurity spectrum is tightly controlled, as the pathway avoids the formation of regio-isomers often seen in direct alkylation of the phthalide ring. The ability to toggle between chiral and achiral forms using this ketone intermediate provides a flexible platform for manufacturing both racemic Citalopram and its active S-enantiomer, maximizing the utility of the production asset.
How to Synthesize Citalopram Efficiently
Implementing this synthesis route requires precise control over the protection and oxidation steps to generate the high-quality Formula IV intermediate. The process begins with the protection of the hydroxymethyl group on the benzonitrile precursor, followed by oxidation of the benzylic alcohol to a ketone. Once the key intermediate is secured, the final assembly involves the addition of the amine-containing Grignard reagent. The following guide outlines the standardized operational framework derived from the patent examples, ensuring that technical teams can replicate the high yields and purity levels demonstrated in the literature. Detailed standard operating procedures for each unit operation are critical for maintaining batch-to-batch consistency.
- Protect the hydroxymethyl group of the benzonitrile precursor using acyl chlorides or sulfonyl chlorides to form the protected intermediate.
- Oxidize the secondary alcohol to a ketone using catalytic sodium tungstate and hydrogen peroxide or other suitable oxidizing agents.
- React the protected ketone with 3-(N,N-dimethylamino)propyl magnesium halide, allowing spontaneous ring closure to form the isobenzofuran core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend well beyond the laboratory bench. The primary advantage lies in the substantial simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and improved throughput. By eliminating the need for cuprous cyanide, facilities can avoid the高昂 costs associated with hazardous waste disposal and specialized containment infrastructure. This removal of a major environmental liability streamlines regulatory compliance and reduces the risk of production stoppages due to safety audits. Additionally, the convergence of the Grignard addition and cyclization into a single step reduces the total number of reactor turnovers required, effectively increasing the capacity of existing manufacturing assets without capital investment. This efficiency gain is crucial for reducing lead time for high-purity pharmaceutical intermediates in a volatile global market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and toxic reagents like cuprous cyanide, which require costly neutralization and disposal protocols. Furthermore, the spontaneous cyclization mechanism removes the need for separate dehydration units and the associated consumption of strong acids and solvents. This consolidation of steps leads to a significant decrease in raw material consumption and energy usage per kilogram of product. The high yields reported in the patent examples, often exceeding 90% for the oxidation and protection steps, ensure that material throughput is maximized, minimizing the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as substituted benzonitriles and standard acyl chlorides, are commodity chemicals with robust global supply chains, reducing the risk of raw material shortages. The process tolerance for various protecting groups (R = alkyl, acyl, sulfonyl) provides procurement teams with flexibility to source the most cost-effective reagents available at any given time. This adaptability ensures continuous production even if specific reagent prices fluctuate. Moreover, the simplified purification requirements mean that production batches are less likely to fail quality control specifications, guaranteeing a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The avoidance of heavy metal catalysts simplifies the wastewater treatment profile, making it easier for manufacturing sites to meet stringent environmental regulations. The exothermic nature of the Grignard reaction is manageable at large scales due to the single-step design, allowing for better thermal control in large reactors. This scalability ensures that the process can be seamlessly transferred from pilot plant quantities of 100 kgs to commercial production scales of 100 MT annually without losing efficiency or safety margins.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Citalopram synthesis technology. These insights are derived directly from the experimental data and claims within patent CN1550497A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing supply networks.
Q: How does the novel ketone route improve safety compared to conventional methods?
A: The novel route eliminates the need for cuprous cyanide, a highly toxic reagent used in traditional methods for introducing the cyano group, thereby significantly reducing hazardous waste handling requirements.
Q: What are the advantages of the spontaneous cyclization step?
A: The spontaneous ring closure during the Grignard reaction reduces the number of unit operations, minimizing solvent usage and processing time while improving overall yield consistency.
Q: Can this method be adapted for producing S-Citalopram?
A: Yes, the process allows for the recycling of the R-enantiomer back into the racemic mixture or conversion pathways, facilitating the efficient production of the active S-enantiomer with high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the ketone intermediate route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Citalopram intermediate meets the highest global pharmacopeial standards. Our commitment to process optimization allows us to deliver cost-effective solutions without compromising on the quality essential for patient safety.
We invite you to collaborate with us to optimize your supply chain for antidepressant manufacturing. By leveraging our technical proficiency, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of Citalopram intermediates that supports your long-term commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
