Advanced Rhodium-Catalyzed Asymmetric Hydrogenation for Scalable Brivaracetam Intermediate Production
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds, particularly for antiepileptic agents like Brivaracetam. Patent CN116120138A introduces a transformative approach to synthesizing optically active gamma-butyrolactone derivatives, which serve as critical precursors in this therapeutic class. This technology leverages a sophisticated rhodium(I) catalyst system paired with novel ferrocenyl-based chiral ligands, specifically the Zhaophos family, to overcome historical limitations in asymmetric hydrogenation. By shifting away from conventional ruthenium or iridium-based protocols, this method delivers exceptional enantioselectivity and conversion rates under remarkably mild conditions. For R&D directors and process chemists, this represents a significant leap forward in accessing high-purity intermediates with reduced process complexity. The ability to tune the ligand structure allows for precise control over the stereochemical outcome, ensuring that the resulting gamma-butyrolactone cores meet the stringent purity specifications required for modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of gamma-butenolactones has been plagued by significant technical hurdles that impede efficient commercial production. Early methodologies, such as those utilizing Ru/BINAP catalysts, often necessitated harsh reaction conditions including elevated temperatures and extreme pressures, yet frequently resulted in disappointing enantiomeric excess values. 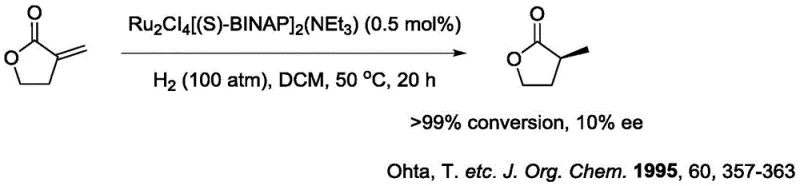 Furthermore, alternative approaches employing iridium complexes, while utilizing cheaper metals, suffered from intrinsically poor catalytic activity, leading to low conversion rates and prolonged reaction times that are economically unviable for large-scale operations.
Furthermore, alternative approaches employing iridium complexes, while utilizing cheaper metals, suffered from intrinsically poor catalytic activity, leading to low conversion rates and prolonged reaction times that are economically unviable for large-scale operations. 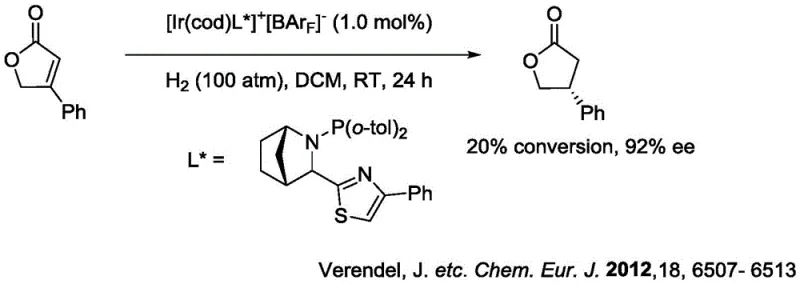 These legacy processes often require extensive downstream purification to remove metal residues and separate racemic byproducts, thereby inflating production costs and extending lead times. The inability to consistently achieve high optical purity without sacrificing yield has long been a bottleneck for supply chain managers seeking reliable sources of chiral building blocks.
These legacy processes often require extensive downstream purification to remove metal residues and separate racemic byproducts, thereby inflating production costs and extending lead times. The inability to consistently achieve high optical purity without sacrificing yield has long been a bottleneck for supply chain managers seeking reliable sources of chiral building blocks.
The Novel Approach
The methodology disclosed in CN116120138A fundamentally redefines the efficiency landscape for gamma-butyrolactone synthesis through the strategic application of a rhodium-Zhaophos catalyst system. This novel approach operates effectively at ambient temperatures ranging from 10°C to 60°C, drastically reducing energy consumption compared to thermal-intensive legacy methods. The system demonstrates remarkable versatility, accommodating a wide array of substituents on the lactone ring while maintaining conversion rates that frequently exceed 99%. By optimizing the electronic and steric properties of the ferrocenyl-thiourea ligand backbone, the process achieves enantiomeric excess values up to 98%, effectively eliminating the need for costly chiral resolution steps. This breakthrough not only simplifies the synthetic route but also enhances the overall atom economy, providing a sustainable and commercially attractive pathway for generating high-value pharmaceutical intermediates with superior quality profiles.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the unique coordination chemistry between the rhodium(I) metal center and the specialized Zhaophos ligands. The catalyst is typically generated in situ or pre-formed by mixing a rhodium precursor, such as Rh(NBD)2BF4, with the chiral ligand in a suitable organic solvent like dichloromethane. 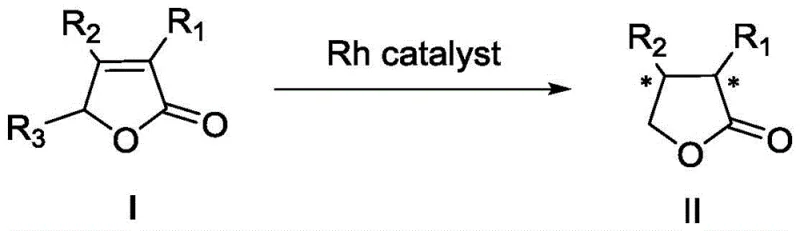 The ligand structure, featuring a ferrocenyl scaffold coupled with a thiourea moiety and phosphine groups, creates a highly defined chiral pocket around the metal center. This specific geometry dictates the facial selectivity of hydrogen addition to the carbon-carbon double bond of the substrate. The thiourea component likely plays a crucial role in substrate activation through hydrogen bonding interactions, particularly when hydroxyl-substituted substrates are employed, thereby enhancing both reactivity and stereocontrol. This dual-activation mechanism ensures that the hydrogenation proceeds with high fidelity, minimizing the formation of unwanted diastereomers or regioisomers that could complicate downstream processing.
The ligand structure, featuring a ferrocenyl scaffold coupled with a thiourea moiety and phosphine groups, creates a highly defined chiral pocket around the metal center. This specific geometry dictates the facial selectivity of hydrogen addition to the carbon-carbon double bond of the substrate. The thiourea component likely plays a crucial role in substrate activation through hydrogen bonding interactions, particularly when hydroxyl-substituted substrates are employed, thereby enhancing both reactivity and stereocontrol. This dual-activation mechanism ensures that the hydrogenation proceeds with high fidelity, minimizing the formation of unwanted diastereomers or regioisomers that could complicate downstream processing.
From an impurity control perspective, this catalytic system offers distinct advantages by suppressing side reactions commonly associated with lactone hydrogenation. The mild reaction conditions prevent thermal degradation of the sensitive lactone ring, while the high chemoselectivity of the rhodium complex avoids over-reduction or hydrogenolysis of other functional groups present on the molecule. The patent data indicates that even with substrates bearing diverse aryl, alkyl, or heteroaryl groups, the reaction maintains consistent performance, yielding products with minimal impurity profiles. This robustness is critical for regulatory compliance, as it reduces the burden on analytical teams to identify and quantify trace impurities. Furthermore, the ability to fine-tune the ligand substituents allows process chemists to optimize the catalyst for specific substrate classes, ensuring that the final API intermediate meets the rigorous purity standards demanded by global health authorities without requiring extensive recrystallization or chromatographic purification.
How to Synthesize Chiral Gamma-Butyrolactone Derivatives Efficiently
The implementation of this asymmetric hydrogenation protocol is designed for operational simplicity, making it highly accessible for both laboratory-scale optimization and pilot plant execution. The process begins with the preparation of the active catalyst species, typically involving the mixing of the rhodium precursor and the Zhaophos ligand in anhydrous dichloromethane under an inert argon atmosphere to prevent oxidation. Once the catalyst is activated, the gamma-butenolactone substrate is introduced, optionally along with an acidic additive like trifluoroacetic acid to boost performance for specific hydroxylated derivatives. The reaction mixture is then subjected to hydrogen pressure, generally between 4 MPa and 6 MPa, and stirred at room temperature for approximately 24 hours. Detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Prepare the catalyst by mixing Rh(NBD)2BF4 precursor with Zhaophos ligand in anhydrous DCM under inert atmosphere.
- Add the gamma-butenolactone substrate and optional acidic additive (e.g., CF3COOH) to the catalyst solution.
- Pressurize the reactor with hydrogen gas (4-6 MPa) and stir at room temperature for 24 hours to achieve high conversion and enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology translates into tangible strategic benefits regarding cost stability and supply reliability. The primary economic driver is the substantial reduction in processing steps; because the reaction achieves such high enantioselectivity and conversion directly, the need for expensive chiral separation technologies or multiple recrystallization cycles is effectively eliminated. This streamlining of the manufacturing workflow significantly lowers the overall cost of goods sold (COGS) by reducing solvent usage, energy consumption, and labor hours associated with purification. Additionally, the catalyst loading is remarkably low, often ranging from 0.01% to 1%, which mitigates the financial impact of using precious metal catalysts. The ability to recover and potentially recycle the rhodium species further enhances the economic viability of the process, ensuring that raw material costs remain predictable and manageable even in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps and the high efficiency of the catalyst directly contribute to a leaner manufacturing process. By avoiding the waste associated with discarding unwanted enantiomers, the overall material throughput is maximized, leading to significant cost savings in raw material procurement. The mild operating conditions also reduce the capital expenditure required for specialized high-temperature or high-pressure reactors, allowing for production in standard stainless steel equipment. Furthermore, the high yield minimizes the volume of waste streams requiring treatment, lowering environmental compliance costs and disposal fees associated with hazardous chemical waste.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream API manufacturers. The use of commercially available and stable starting materials, such as substituted gamma-butenolactones, reduces the risk of supply disruptions caused by obscure or custom-synthesized precursors. The short reaction time of approximately 24 hours at room temperature allows for faster turnaround times compared to multi-day thermal processes, enabling more responsive production scheduling. This agility helps mitigate inventory risks and ensures that just-in-time delivery commitments to pharmaceutical partners can be met with greater confidence and precision.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common solvents like dichloromethane that are well-understood in industrial settings and can be efficiently recovered and recycled. The low catalyst loading reduces the heavy metal burden in the final product, simplifying the purification process to meet strict residual metal limits set by regulatory agencies. This aligns with green chemistry principles by improving atom economy and reducing the E-factor of the synthesis. The mild conditions also enhance operational safety, reducing the risks associated with high-temperature exotherms, which facilitates smoother technology transfer from R&D to commercial manufacturing sites without extensive re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the advantages of this Rh-catalyzed method over traditional Ru/BINAP systems?
A: Unlike traditional Ru/BINAP systems which often suffer from poor enantioselectivity and harsh reaction conditions, this novel Rh-Zhaophos protocol operates under milder temperatures with significantly improved stereocontrol, achieving up to 98% ee.
Q: Can this process handle substrates with hydroxyl groups?
A: Yes, the process is highly robust. By incorporating specific acidic additives like trifluoroacetic acid, the system maintains high conversion and enantioselectivity even for challenging hydroxyl-substituted gamma-butenolactone substrates.
Q: Is this method suitable for large-scale industrial production?
A: Absolutely. The reaction utilizes low catalyst loadings (0.01%-1%), operates at moderate pressures, and employs common solvents like DCM, making it economically viable and technically feasible for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
The synthesis pathway outlined in CN116120138A demonstrates immense potential for the commercial production of Brivaracetam and related analogues, offering a blend of high stereocontrol and operational efficiency. 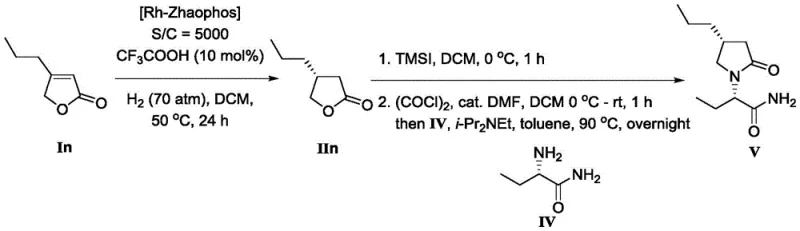 NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of chiral gamma-butyrolactone intermediate meets the exacting standards required for epilepsy medication manufacturing. We understand the critical nature of supply continuity in the pharmaceutical sector and have optimized our processes to deliver consistent quality at scale.
NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of chiral gamma-butyrolactone intermediate meets the exacting standards required for epilepsy medication manufacturing. We understand the critical nature of supply continuity in the pharmaceutical sector and have optimized our processes to deliver consistent quality at scale.
We invite you to engage with our technical procurement team to discuss how this innovative rhodium-catalyzed route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the potential economic benefits of switching to this high-efficiency method. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Our team is prepared to collaborate closely with your R&D and supply chain divisions to ensure a seamless transition to this superior manufacturing technology, securing a competitive advantage in the global market for antiepileptic therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
