Advanced Chiral Tertiary Phosphine Thiourea Catalysts for Efficient Asymmetric Synthesis
The landscape of asymmetric organocatalysis has been significantly advanced by the innovations detailed in patent CN103421044A, which introduces a novel class of chiral tertiary phosphine thiourea compounds incorporating a dehydroabietylamine structural unit. This technological breakthrough addresses long-standing challenges in the efficient synthesis of complex chiral molecules, particularly through the Morita-Baylis-Hillman (MBH) reaction. By leveraging the rigid, naturally occurring chiral scaffold of dehydroabietylamine, these new catalysts offer a robust platform for generating high-value pharmaceutical intermediates with exceptional stereochemical fidelity. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and highly selective catalytic systems that eliminate the need for toxic heavy metals while maintaining rigorous purity standards required for drug substance manufacturing.
Furthermore, the commercial implications of this technology extend far beyond the laboratory bench, offering substantial value to procurement and supply chain stakeholders. The utilization of dehydroabietylamine, a derivative of abietic acid found in abundant pine rosin, ensures a stable and cost-effective supply of the chiral source material. This reliance on renewable natural products mitigates the volatility associated with synthetic chiral pools and reduces the overall cost of goods sold (COGS) for downstream fine chemical manufacturing. As a reliable pharmaceutical intermediate supplier, understanding the mechanistic superiority and economic viability of such organocatalysts is essential for optimizing production pipelines and securing a competitive edge in the global market for high-purity chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric Morita-Baylis-Hillman reaction has posed significant hurdles for process chemists, primarily due to the sluggish reaction kinetics and poor enantioselectivity observed with traditional monofunctional catalysts. Simple chiral tertiary phosphines, while effective as Lewis bases for activating electron-deficient olefins, often fail to exert sufficient stereocontrol over the incoming electrophile, leading to racemic mixtures or low enantiomeric excess (ee) values that necessitate costly and wasteful recrystallization steps. Moreover, many existing catalytic systems require harsh reaction conditions, cryogenic temperatures, or stoichiometric amounts of additives to achieve moderate yields, which drastically increases energy consumption and complicates waste stream management in large-scale operations. The inability to broadly accommodate diverse substrate scopes, particularly with simple acrylates and aromatic aldehydes, has further limited the utility of these conventional methods in the rapid synthesis of versatile pharmaceutical scaffolds.
The Novel Approach
The innovative strategy presented in the patent data overcomes these deficiencies by engineering a bifunctional catalyst architecture that synergistically combines Lewis basicity with strong hydrogen-bonding activation. By tethering a chiral tertiary phosphine moiety to a thiourea group via a rigid dehydroabietylamine linker, the new catalyst creates a well-defined chiral pocket capable of simultaneous activation of both the nucleophilic acrylate and the electrophilic aldehyde. This dual-activation mechanism not only accelerates the reaction rate, allowing it to proceed efficiently at ambient temperatures, but also enforces a strict geometric orientation on the transition state, resulting in dramatically improved enantioselectivity. The versatility of this approach is evidenced by its ability to tolerate a wide range of substituents on the aromatic aldehyde, including electron-withdrawing groups like nitro and cyro functionalities, delivering yields up to 94% and ee values up to 89% without the need for specialized equipment or extreme conditions.

Mechanistic Insights into Bifunctional Organocatalysis
The exceptional performance of these dehydroabietylamine-derived catalysts stems from their sophisticated molecular design, which integrates multiple non-covalent interactions to govern reactivity and selectivity. The tertiary phosphine center acts as a soft nucleophile, attacking the beta-position of the acrylate to generate a zwitterionic enolate intermediate, a critical step in the MBH manifold. Concurrently, the thiourea moiety functions as a potent hydrogen-bond donor, coordinating with the carbonyl oxygen of the aromatic aldehyde to lower its LUMO energy and enhance its electrophilicity. This cooperative activation brings the reacting partners into close proximity within a chiral environment defined by the bulky, rigid tricyclic diterpene skeleton of the dehydroabietylamine unit. The steric bulk of the isopropyl and methyl groups on the phenanthrene ring system effectively shields one face of the approaching electrophile, thereby directing the formation of the new carbon-carbon bond with high facial selectivity.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions such as polymerization of the acrylate or self-condensation of the aldehyde, which are common pitfalls in uncatalyzed or poorly catalyzed MBH reactions. The precise spatial arrangement enforced by the catalyst ensures that the desired cross-coupling pathway is kinetically favored over competing processes, leading to cleaner reaction profiles and simplified downstream purification. For quality assurance teams, this translates to a reduced burden on analytical resources, as the crude product streams contain fewer structurally related impurities that are difficult to separate. The stability of the catalyst under ambient conditions further contributes to process robustness, ensuring consistent batch-to-batch reproducibility which is a critical parameter for regulatory compliance in the manufacture of active pharmaceutical ingredients (APIs) and their precursors.
How to Synthesize Chiral Tertiary Phosphine Thiourea Efficiently
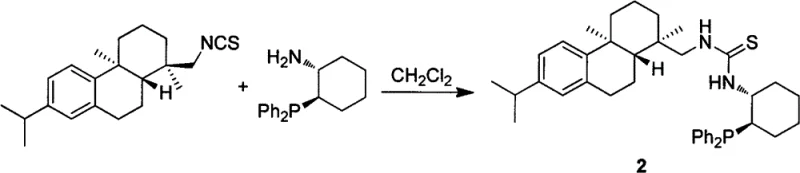
The preparation of these high-performance catalysts is remarkably straightforward, relying on a direct condensation reaction between readily accessible chiral building blocks. The synthetic route utilizes dehydroabietylamine-derived isothiocyanate, which serves as the electrophilic partner, and reacts it with enantiomerically pure 2-(dihydrocarbyl)phosphino-1-primary amines. This atom-economical transformation proceeds smoothly in common organic solvents such as dichloromethane or chloroform, requiring no exotic reagents or complex protection-deprotection sequences. The simplicity of the synthesis facilitates rapid access to a library of catalyst variants by simply varying the phosphine substituent, allowing process developers to fine-tune steric and electronic properties for specific substrate challenges. Detailed standardized synthesis steps for the most effective variant, Catalyst 2, are outlined in the guide below to assist technical teams in replicating these results.
- Dissolve dehydroabietylamine-derived isothiocyanate and chiral 2-(dihydrocarbyl)phosphino-1-primary amine in an organic solvent such as dichloromethane.
- Stir the reaction mixture under nitrogen protection at room temperature to reflux temperature for 1 to 24 hours to ensure complete condensation.
- Purify the resulting crude product using silica gel column chromatography with a gradient elution of petroleum ether and ethyl acetate to obtain the pure white solid catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dehydroabietylamine-based catalytic technology presents a compelling value proposition centered on cost efficiency, supply security, and environmental sustainability. The primary driver for cost reduction lies in the origin of the chiral scaffold; dehydroabietylamine is derived from rosin, a renewable and abundant byproduct of the paper pulping industry, making it significantly cheaper than many synthetic chiral ligands that require multi-step total synthesis. This inherent cost advantage at the raw material level cascades through the entire production value chain, enabling substantial cost savings in fine chemical manufacturing without compromising on the quality or performance of the final catalyst. Furthermore, the elimination of transition metals from the catalytic cycle removes the need for expensive metal scavenging resins and rigorous heavy metal testing, streamlining the purification process and reducing the overall operational expenditure associated with quality control and waste disposal.
- Cost Reduction in Manufacturing: The economic benefits of this technology are multifaceted, extending beyond just the price of the starting materials to include significant operational efficiencies. By operating effectively at room temperature, the process eliminates the energy costs associated with cryogenic cooling or prolonged heating, which are often required for less active catalysts. Additionally, the high turnover and selectivity of the catalyst mean that lower catalyst loadings can be utilized to achieve the same throughput, further driving down the cost per kilogram of the produced intermediate. The simplified workup procedure, facilitated by the clean reaction profile, reduces solvent consumption and labor hours, contributing to a leaner and more profitable manufacturing operation that maximizes resource utilization.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically enhanced by the use of bio-based feedstocks that are less susceptible to the geopolitical and petrochemical market fluctuations that often plague synthetic organic chemistry. The robust nature of the catalyst synthesis, which tolerates standard industrial solvents and ambient conditions, ensures that production can be maintained consistently even in the face of minor variations in utility availability. This reliability is paramount for maintaining continuous flow in the production of high-purity pharmaceutical intermediates, where interruptions can lead to significant delays in drug development timelines. The ability to source the key chiral precursor from a stable, renewable supply chain mitigates the risk of raw material shortages and provides a strategic buffer against market volatility.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this organocatalytic approach aligns perfectly with modern green chemistry principles and increasingly stringent regulatory requirements. The absence of toxic heavy metals simplifies the environmental impact assessment and reduces the regulatory burden associated with metal residue limits in final drug products. The process generates minimal hazardous waste, and the use of benign solvents allows for easier recycling and recovery, supporting corporate sustainability goals. Scalability is inherently supported by the simple reaction setup, which does not require specialized high-pressure or low-temperature reactors, allowing for seamless translation from gram-scale laboratory optimization to multi-ton commercial production facilities with minimal engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, aiming to clarify the practical aspects of adopting this novel synthetic methodology. Understanding these details is crucial for technical decision-makers evaluating the feasibility of integrating this catalyst into their existing process workflows for the production of complex chiral molecules.
Q: What are the primary advantages of using dehydroabietylamine-based catalysts over traditional phosphine catalysts?
A: Traditional simple phosphine catalysts often suffer from limited enantioselectivity and narrow substrate scope in Morita-Baylis-Hillman reactions. The novel dehydroabietylamine-based bifunctional catalysts introduce a rigid chiral backbone and a thiourea hydrogen-bonding donor, which simultaneously activates both nucleophiles and electrophiles, significantly enhancing stereocontrol and yielding up to 89% ee.
Q: Can these catalysts be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the synthesis involves readily available starting materials derived from natural rosin and standard condensation reactions under mild conditions. The absence of expensive transition metals and the use of common organic solvents make the process highly scalable and suitable for commercial manufacturing of complex chiral intermediates.
Q: What represents the optimal reaction conditions for the Morita-Baylis-Hillman reaction using these catalysts?
A: The patent data indicates that the reaction proceeds efficiently at room temperature (25°C) in tetrahydrofuran (THF) with a catalyst loading of 10 mol%. Under these mild conditions, the system achieves high conversion rates with yields reaching up to 94% for various aromatic aldehydes and acrylates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Thiourea Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be reliably translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral catalyst or intermediate we deliver meets the highest international standards. We are committed to supporting our clients in navigating the complexities of asymmetric synthesis, providing the technical expertise and manufacturing capacity needed to bring life-saving therapies to market faster and more efficiently.
We invite you to engage with our technical procurement team to discuss how this dehydroabietylamine-based catalytic technology can be tailored to your specific synthesis challenges. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your target molecule. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to serve as your trusted partner in the commercial scale-up of complex organic intermediates. Let us collaborate to optimize your supply chain and drive innovation in your pharmaceutical manufacturing processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
