Optimizing Sacubitril Intermediate Production: A Technical Analysis of Novel Coupling Strategies
Optimizing Sacubitril Intermediate Production: A Technical Analysis of Novel Coupling Strategies
The global pharmaceutical landscape is increasingly focused on cardiovascular therapeutics, with heart failure representing a critical area of unmet medical need. The dual-action inhibitor Sacubitril/Valsartan has revolutionized treatment protocols, driving immense demand for high-quality active pharmaceutical ingredients and their precursors. Central to this supply chain is the efficient production of the Sacubitril intermediate, specifically N-tert-butyloxycarbonyl-amino-4, 4'-biphenyl-R-alanine methyl ester. Recent intellectual property, specifically patent CN112661671A, discloses a transformative synthetic methodology that addresses longstanding bottlenecks in manufacturing this key building block. By shifting away from toxic and costly activation reagents toward a more economical halogenation and coupling strategy, this technology offers a compelling value proposition for generic drug manufacturers and CDMOs alike. The structural complexity of the target molecule, featuring a biphenyl moiety attached to an amino acid backbone, necessitates precise stereochemical control and robust process chemistry, as illustrated in the molecular architecture below.
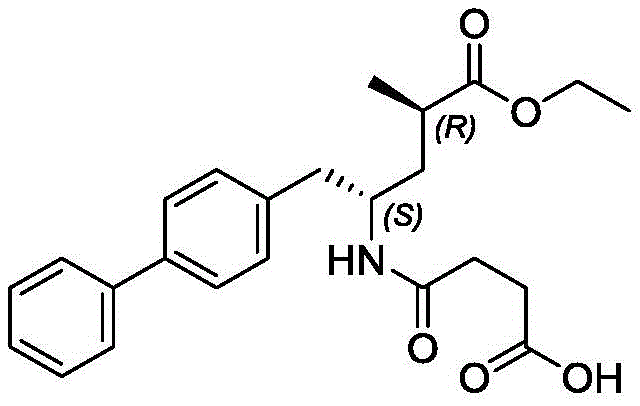
This technical insight report dissects the proprietary synthesis route detailed in the patent, evaluating its mechanistic underpinnings and commercial viability. For R&D directors, the focus lies on the improved impurity profile and the elimination of hazardous reagents like triflic anhydride. For procurement and supply chain leaders, the substitution of expensive palladium catalysts and boronic acids with commodity chemicals represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing. As we delve into the specifics of this novel pathway, it becomes evident that this method not only enhances operational safety but also streamlines the purification workflow, ensuring a reliable supply of high-purity materials essential for regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the biphenyl alanine scaffold relied heavily on classical Suzuki-Miyaura coupling conditions that, while effective on a small scale, present severe drawbacks for industrial application. The traditional route typically begins with BOC-D-tyrosine methyl ester, which must be activated to participate in cross-coupling reactions. This activation step conventionally employs trifluoromethanesulfonic anhydride to generate a triflate leaving group. Triflic anhydride is not only prohibitively expensive but also poses significant handling hazards due to its corrosive nature and moisture sensitivity, complicating reactor maintenance and operator safety protocols. Furthermore, the subsequent coupling step requires phenylboronic acid and substantial quantities of tetrakis(triphenylphosphine)palladium(0).
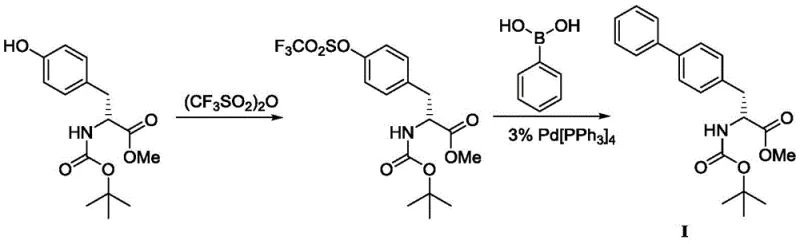
The reliance on Pd(PPh3)4 is particularly problematic from a cost and sustainability perspective. This catalyst is air-sensitive, requiring inert atmosphere handling which increases operational complexity and capital expenditure for specialized equipment. Moreover, the stoichiometry often demands large equivalents of the catalyst to drive the reaction to completion, leading to high residual palladium levels in the crude product. Removing these heavy metal impurities to meet stringent pharmaceutical specifications (<10 ppm) necessitates additional purification steps, such as scavenging or recrystallization, which erode overall yield and extend cycle times. Consequently, the conventional method suffers from poor atom economy, high raw material costs, and a cumbersome waste stream profile that is increasingly untenable in modern green chemistry frameworks.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112661671A introduces a paradigm shift by utilizing a halogenation-coupling sequence that bypasses the need for triflate activation entirely. The new route initiates with the conversion of the hydroxyl group on the tyrosine derivative directly into a halide using phosphorus trihalides (PX3), such as PBr3 or PCl3. These reagents are commodity chemicals, widely available at a fraction of the cost of triflic anhydride, and they react efficiently under mild conditions to generate the reactive aryl halide intermediate (Compound II). This strategic modification simplifies the synthetic tree and drastically reduces the hazard profile of the initial activation step, facilitating safer scale-up operations.
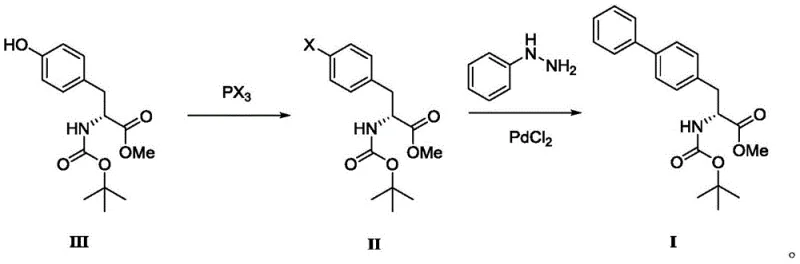
Following halogenation, the process employs a palladium-catalyzed coupling with phenylhydrazine instead of phenylboronic acid. Phenylhydrazine is a significantly more economical feedstock compared to boronic acids, offering immediate raw material cost savings. Crucially, the catalytic system utilizes PdCl2, a stable and inexpensive palladium(II) salt, rather than the sensitive Pd(0) complex. The addition of simple promoters like CuI, KI, or NaI facilitates the catalytic cycle, allowing for low catalyst loadings while maintaining high conversion rates. This approach not only lowers the direct cost of goods sold (COGS) but also minimizes the burden on downstream purification, as the removal of inorganic salts and simple palladium species is generally more straightforward than removing bulky phosphine ligands. The result is a streamlined, cost-effective process ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into PdCl2-Catalyzed Hydrazine Coupling
The core innovation of this synthetic route lies in the efficient construction of the carbon-carbon bond between the tyrosine-derived aryl ring and the phenyl ring using phenylhydrazine as the coupling partner. Mechanistically, this transformation likely proceeds through a palladium-catalyzed cycle where the aryl halide (Compound II) undergoes oxidative addition to the palladium center. While traditional Suzuki coupling involves transmetallation with a boron species, the use of phenylhydrazine suggests a variation where the hydrazine serves as a source of the phenyl nucleophile, potentially via an in situ generated diazonium species or direct transmetallation facilitated by the iodide additives. The presence of additives like KI or CuI is critical; iodide ions can enhance the rate of oxidative addition by converting aryl chlorides or bromides into more reactive aryl iodides in situ, or they may stabilize the active palladium species.
From an impurity control perspective, this mechanism offers distinct advantages over the boronic acid route. One of the primary challenges in biphenyl synthesis is the formation of homocoupling byproducts (e.g., biphenyl from two phenyl rings or biaryl from two tyrosine units). The specific catalytic environment created by PdCl2 and the chosen additives appears to suppress these side reactions effectively, as evidenced by the high purity reports in the patent examples. Furthermore, the absence of boronic acid eliminates the formation of difficult-to-remove boron-containing impurities and phenol byproducts that often plague Suzuki reactions. The reaction conditions, typically ranging from 60°C to 150°C in polar aprotic solvents like DMF or NMP, provide sufficient energy to overcome activation barriers without compromising the stereochemical integrity of the chiral alpha-carbon, ensuring the retention of the (R)-configuration essential for biological activity.
How to Synthesize Sacubitril Intermediate Efficiently
Implementing this novel synthesis requires careful attention to reaction parameters to maximize yield and purity. The process is divided into two distinct stages: the halogenation of the protected tyrosine derivative and the subsequent cross-coupling reaction. The first stage must be conducted at controlled low temperatures to prevent racemization and manage the exotherm associated with phosphorus trihalide addition. The second stage involves heating the reaction mixture to facilitate the coupling, followed by a rigorous workup procedure to remove metal residues. The following guide outlines the generalized operational framework derived from the patent embodiments, serving as a foundational reference for process development teams aiming to adopt this technology for reliable Sacubitril intermediate supplier capabilities.
- React the starting material (Compound III) with phosphorus trihalide (PX3) in a solvent like dichloromethane at low temperature (-10°C to 0°C) to form the halogenated intermediate (Compound II).
- Purify Compound II through aqueous workup, washing with sodium bicarbonate and water, followed by drying and concentration.
- Couple Compound II with phenylhydrazine using a PdCl2 catalyst and additive (CuI/KI/NaI) in a polar solvent at elevated temperatures (60-150°C) to yield the final biphenyl intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond simple unit price reductions. The shift from specialized, high-cost reagents to commodity chemicals fundamentally alters the risk profile of the supply chain. By eliminating the dependency on trifluoromethanesulfonic anhydride and large quantities of specialized palladium catalysts, manufacturers can mitigate the risks associated with supplier shortages and price volatility of niche fine chemicals. This resilience is crucial for maintaining continuous production schedules in the face of global supply chain disruptions, ensuring that downstream API manufacturing remains uninterrupted.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive reagents with cost-effective alternatives. Replacing triflic anhydride with phosphorus trihalides and phenylboronic acid with phenylhydrazine results in a drastic reduction in raw material expenditures. Furthermore, the use of PdCl2 instead of Pd(PPh3)4 significantly lowers catalyst costs, as the former is a bulk chemical while the latter is a specialized ligand-complexed catalyst. These changes collectively contribute to substantial cost savings in pharmaceutical intermediates manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: The raw materials required for this novel route, such as PBr3, PCl3, and phenylhydrazine, are produced on a massive industrial scale for various chemical sectors. This widespread availability ensures a stable and redundant supply base, reducing lead time for high-purity pharmaceutical intermediates. Unlike specialized boronic acids which may have limited suppliers and long lead times, these commodity chemicals can be sourced from multiple vendors globally, providing procurement teams with greater negotiating power and supply security.
- Scalability and Environmental Compliance: From an operational standpoint, the simplified workup procedures and the use of robust catalysts enhance the scalability of the process. The elimination of air-sensitive reagents reduces the need for complex inert atmosphere setups, allowing for the use of standard glass-lined reactors. Additionally, the reduced generation of hazardous waste and the avoidance of toxic triflates align with increasingly stringent environmental regulations, facilitating easier permitting and waste disposal management for large-scale production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the practical implications for industrial adoption. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing lines.
Q: Why is phenylhydrazine preferred over phenylboronic acid in this synthesis?
A: Phenylhydrazine is significantly more cost-effective than phenylboronic acid. Additionally, the new route avoids the use of expensive trifluoromethanesulfonic anhydride required for activating the tyrosine precursor in traditional boronic acid coupling methods.
Q: What are the purity levels achievable with this new method?
A: The patent data demonstrates that this method consistently achieves high purity levels, with reported values exceeding 99% (e.g., 99.4% to 99.6%) across multiple examples, which is critical for pharmaceutical grade intermediates.
Q: Is the catalyst system scalable for industrial production?
A: Yes, the process utilizes PdCl2, a robust and inexpensive palladium source, often with simple additives like KI or CuI. This contrasts with air-sensitive and expensive Pd(0) complexes used in prior art, making the process highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sacubitril Intermediate Supplier
The technological advancements detailed in patent CN112661671A represent a significant leap forward in the manufacturing efficiency of cardiovascular drug intermediates. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory processes into robust, commercial-scale operations. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required, while our rigorous QC labs enforce stringent purity specifications to guarantee product quality that meets global regulatory standards.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this cost-effective synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and reduce the overall cost of goods for your Sacubitril-based formulations.
