Optimizing Cefpiramide Sodium Production: Advanced Catalytic Strategies for Commercial Scale
The pharmaceutical landscape for third-generation cephalosporins continues to evolve, driven by the relentless demand for higher purity antibiotics with robust stability profiles. A pivotal advancement in this domain is documented in patent CN101671350B, which details a novel preparation method for the Cefpiramide sodium compound, also known industrially as Wy-44635. This specific chemical entity represents a critical milestone in antimicrobial therapy, offering a broad spectrum of activity against gram-positive and gram-negative organisms, including anaerobes, while maintaining significant stability against beta-lactamases. For R&D Directors and Procurement Managers seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this synthesis is paramount. The patent outlines a sophisticated coupling strategy that overcomes the historical limitations of crystalline inhomogeneity and low purity associated with earlier production methods. By leveraging a Triphosgene and Triphenylphosphine oxide catalytic system, the process achieves exceptional yield and purity, directly addressing the stringent quality requirements of modern regulatory bodies. This report delves deep into the technical mechanics and commercial implications of this route, providing a comprehensive analysis for stakeholders focused on cost reduction in pharmaceutical manufacturing and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
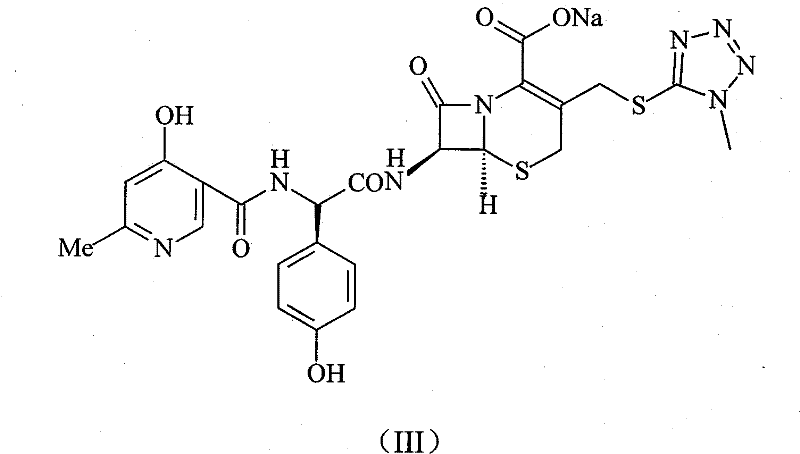
Prior to the innovations described in CN101671350B, the industry relied heavily on methods disclosed in patents such as CN1594324A and CN101037444A. These conventional approaches typically involved the direct reaction of Cefpiramide Acid with sodium transforming agents to generate the sodium salt. While conceptually straightforward, these legacy methods suffered from significant technical deficiencies that impacted the final product's commercial viability. The primary drawback was the formation of inhomogeneous crystalline forms, which led to inconsistent dissolution rates and bioavailability issues in downstream formulation. Furthermore, the purity levels achieved through these older salt formation techniques were often suboptimal, necessitating extensive and costly recrystallization steps that eroded profit margins. From a supply chain perspective, the poor stability of the resulting crystals posed risks during storage and transportation, potentially leading to product degradation before it even reached the formulation stage. For a procurement manager, these factors translate into higher rejection rates, increased quality control costs, and potential supply disruptions. The inability to consistently produce high-purity [Pharmaceutical Intermediates] using these traditional routes created a bottleneck for manufacturers aiming to scale production efficiently without compromising on the stringent quality standards required for injectable antibiotics.
The Novel Approach
The methodology introduced in the focal patent represents a paradigm shift by focusing on the optimization of the coupling reaction itself rather than just the final salt formation. This novel approach utilizes a Triphosgene and Triphenylphosphine oxide system to activate the carboxylic acid moiety of the side chain intermediate, D-alpha-(6-methyl-4-hydroxy niacinamide)-p-hydroxyphenyl acetic acid. This activation strategy is particularly effective given the large molecular structure and significant steric hindrance of the intermediate, which traditionally resulted in low yields. By employing this specific catalytic combination, the reaction proceeds with much higher efficiency, leading to a dramatic improvement in both compound purity and overall yield. The process is designed to be operationally simple, utilizing common organic solvents and straightforward equipment, which facilitates easier purification and isolation of the final product. For stakeholders evaluating cost reduction in electronic chemical manufacturing or similar high-value sectors, the principle of simplifying complex coupling reactions to enhance yield is universally applicable. This method not only solves the stability issues inherent in prior art but also ensures a more robust and reproducible manufacturing process. The result is a high-purity Cefpiramide Sodium compound that meets the rigorous demands of clinical applications, offering a distinct competitive advantage in the global market for specialty chemical suppliers.
Mechanistic Insights into Triphosgene-Mediated Amide Coupling
The core of this technological breakthrough lies in the precise mechanistic interaction between the Triphosgene activator and the sterically hindered acid intermediate. In traditional peptide or amide couplings, reagents like DCC or EDC are often used, but they can introduce difficult-to-remove urea byproducts. The Triphosgene system, however, generates the reactive acid chloride or mixed anhydride in situ under mild conditions, which then reacts rapidly with the amine component of the 7-TMCA intermediate. The addition of Triphenylphosphine oxide plays a crucial role in stabilizing the transition state and facilitating the nucleophilic attack, effectively overcoming the steric bulk of the 6-methyl-4-hydroxy niacinamide group. This catalytic cycle ensures that the reaction proceeds to completion with minimal formation of racemized byproducts, which is critical for maintaining the biological activity of the beta-lactam antibiotic. The control of reaction parameters, specifically the temperature range of -5°C to 5°C, is vital to prevent the hydrolysis of the activated intermediate and to preserve the integrity of the sensitive beta-lactam ring. For R&D teams, understanding this mechanism provides a blueprint for optimizing similar coupling reactions in other complex molecule syntheses. The ability to manage steric effects through specific catalytic additives is a key competency in advanced organic synthesis, allowing for the production of high-purity OLED material or complex polymer additives where structural precision is non-negotiable.
Impurity control is another critical aspect where this mechanism excels, directly impacting the commercial viability of the process. The high selectivity of the Triphosgene/Triphenylphosphine oxide system minimizes the formation of side products that typically arise from over-activation or degradation of the starting materials. In the context of antibiotic production, impurities can lead to severe immunogenic reactions or reduced efficacy, making their control a top priority for regulatory compliance. The process described allows for the direct precipitation of the product by adding acetone to the reaction mixture after pH adjustment, which effectively washes away soluble impurities and residual reagents. This simplifies the downstream processing significantly, eliminating the need for complex chromatographic separations that are often cost-prohibitive at scale. The resulting solid exhibits superior stability and homogeneity, addressing the key failures of previous methods. For a supply chain head, this translates to a more predictable manufacturing timeline and reduced waste generation. The robustness of this impurity control mechanism ensures that every batch meets the stringent purity specifications required for clinical use, thereby reducing the risk of batch failures and ensuring a continuous supply of critical medical ingredients.
How to Synthesize Cefpiramide Sodium Efficiently
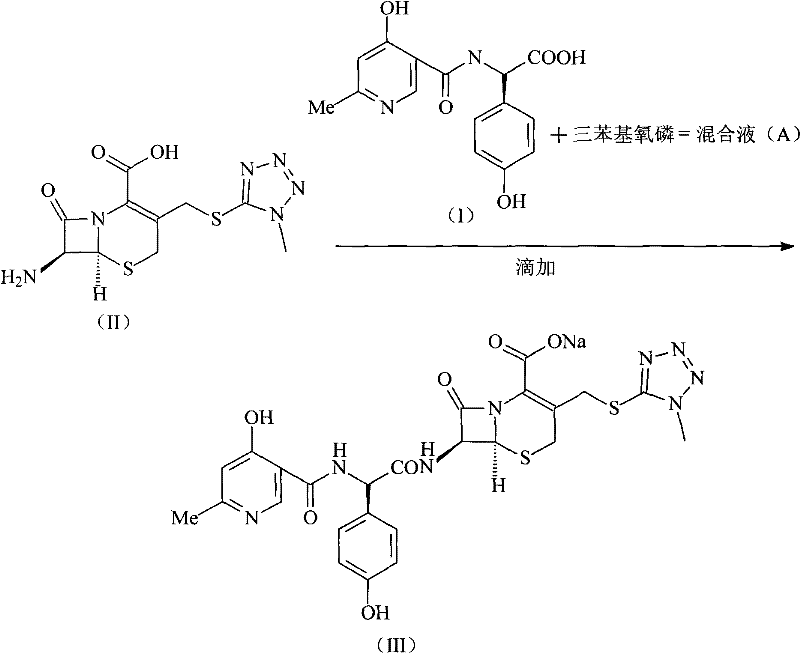
The practical implementation of this synthesis route requires careful attention to solvent selection and temperature control to maximize the benefits of the novel catalytic system. The process begins with the dissolution of Triphosgene in a suitable organic solvent, such as methylene dichloride or toluene, followed by the preparation of a separate solution containing the acid intermediate and Triphenylphosphine oxide. These two streams are then combined to form Mixed Solution A, where the activation of the acid occurs under strictly controlled low-temperature conditions. This mixture is subsequently added dropwise into an aqueous solution of the 7-TMCA intermediate, with the pH carefully maintained between 7 and 8 using an alkaline solution like sodium hydroxide. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations essential for scaling this reaction.
- Dissolve Triphosgene in an organic solvent such as methylene dichloride and prepare a separate solution of D-alpha-(6-methyl-4-hydroxy niacinamide)-p-hydroxy phenylacetic acid with triphenylphosphine oxide.
- Mix the two solutions to form Mixed Solution A while strictly controlling the reaction temperature between -5°C and 5°C to prevent side reactions.
- Add Mixed Solution A dropwise into an aqueous solution of 7-TMCA, regulate pH to 7-8 using alkaline solution, and isolate the solid product via acetone precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this advanced synthesis route offers substantial benefits that extend far beyond simple chemical yield improvements. For procurement managers and supply chain leaders, the primary value proposition lies in the significant optimization of manufacturing costs and the enhancement of supply reliability. The elimination of complex purification steps and the use of readily available, cost-effective reagents like Triphosgene and common organic solvents drastically reduce the raw material expenditure per kilogram of finished product. Furthermore, the high yield and purity achieved reduce the volume of waste generated, leading to lower environmental compliance costs and a smaller carbon footprint. This aligns perfectly with the growing global emphasis on sustainable manufacturing practices. The robustness of the process also means that production schedules are more predictable, reducing the risk of delays that can disrupt the entire pharmaceutical supply chain. For companies seeking a reliable agrochemical intermediate supplier or pharmaceutical partner, these operational efficiencies are key indicators of a mature and dependable manufacturing capability.
- Cost Reduction in Manufacturing: The implementation of the Triphosgene-mediated coupling strategy leads to significant cost savings by streamlining the production workflow and minimizing material loss. By achieving higher conversion rates and reducing the need for extensive recrystallization, the overall cost of goods sold is substantially lowered. The use of inexpensive and widely available catalysts further contributes to this economic advantage, making the process highly competitive in the global market. Additionally, the simplified isolation procedure reduces energy consumption associated with solvent recovery and drying, adding another layer of cost efficiency. These cumulative savings allow for more aggressive pricing strategies or higher margins, providing a strong financial incentive for adopting this technology in large-scale commercial operations.
- Enhanced Supply Chain Reliability: The stability and reproducibility of this synthesis method directly translate to a more reliable supply chain for critical antibiotic ingredients. The ability to consistently produce high-quality Cefpiramide Sodium reduces the incidence of batch rejections and the need for safety stock buffers. This reliability is crucial for maintaining continuous production lines in downstream formulation facilities, preventing costly shutdowns due to material shortages. The use of stable intermediates and robust reaction conditions ensures that the manufacturing process is less susceptible to variations in raw material quality or environmental factors. For supply chain heads, this means reduced lead time for high-purity pharmaceutical intermediates and a more resilient procurement strategy that can withstand market fluctuations and demand spikes.
- Scalability and Environmental Compliance: This process is inherently designed for scalability, utilizing standard chemical engineering unit operations that can be easily transferred from pilot plant to full commercial scale. The straightforward precipitation and filtration steps are well-suited for large-scale reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates without significant engineering hurdles. Moreover, the reduced generation of hazardous waste and the use of recoverable solvents align with strict environmental regulations, minimizing the risk of compliance issues. The ability to scale efficiently while maintaining environmental standards is a critical competitive advantage in the modern chemical industry. This ensures long-term operational sustainability and protects the company from potential regulatory fines or production halts, securing the supply of essential medicines for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Cefpiramide Sodium. These insights are derived directly from the patent data and industry best practices, providing clarity for decision-makers evaluating this technology. Understanding these details is essential for assessing the feasibility of integrating this synthesis route into existing manufacturing portfolios. The answers highlight the specific advantages of the Triphosgene method and its alignment with current industry standards for quality and efficiency.
Q: What are the primary advantages of the Triphosgene method over conventional salt formation?
A: The Triphosgene-mediated approach significantly improves product purity and stability compared to prior art methods which often resulted in inhomogeneous crystalline forms and lower stability profiles.
Q: How does the low-temperature control impact the impurity profile?
A: Maintaining the reaction temperature between -5°C and 5°C is critical for minimizing degradation of the beta-lactam ring and ensuring high stereochemical integrity of the final Cefpiramide Sodium compound.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the process utilizes common organic solvents like methylene dichloride and toluene and involves straightforward precipitation steps, making it highly suitable for commercial scale-up from 100 kgs to 100 MT.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpiramide Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality antibiotics in the global healthcare ecosystem. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative synthesis routes like the one described in CN101671350B can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex beta-lactam chemistry positions us as a strategic partner for pharmaceutical companies seeking to optimize their supply chains. By leveraging our technical expertise and manufacturing capacity, we help clients navigate the challenges of commercial scale-up, ensuring a steady supply of high-purity Cefpiramide Sodium that meets the highest international standards.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your specific production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the tangible value of our manufacturing capabilities. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving efficiency and quality in the production of essential pharmaceutical ingredients. Let us help you secure your supply of critical antibiotics with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
