Advanced Manufacturing of Cefpiramide Sodium: A Direct Salting-Out Strategy for High Purity
Advanced Manufacturing of Cefpiramide Sodium: A Direct Salting-Out Strategy for High Purity
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational efficiency, particularly for complex beta-lactam antibiotics. Patent CN112442048B introduces a significant technological advancement in the preparation of cefpiramide sodium, a third-generation cephalosporin with broad-spectrum antibacterial activity. Unlike conventional methods that rely on cumbersome amine salt intermediates and multi-solvent systems, this novel approach utilizes a direct salting-out strategy employing sodium isooctanoate. This shift represents a critical optimization in the commercial scale-up of complex pharmaceutical intermediates, offering a streamlined route that enhances product stability while drastically simplifying the downstream processing requirements. By focusing on a single-solvent crystallization system based on acetone, the process addresses long-standing challenges related to solvent residue and crystal morphology, positioning it as a superior choice for reliable API intermediate supplier networks aiming for green chemistry compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for cefpiramide sodium, such as those described in prior art documents like CN201310225796.6 and 201811294558.X, are fraught with inefficiencies that impact both cost and quality. These legacy processes typically involve a multi-step salt exchange mechanism where cefpiramide acid is first converted into an amine salt using organic bases like triethylamine or N,N-diisopropylethylamine. This intermediate amine salt must then be reacted with a sodium source to generate the final sodium salt, a redundant step that introduces additional unit operations and potential points of failure. Furthermore, these conventional crystallization protocols often necessitate the use of binary or ternary solvent systems, frequently incorporating hazardous or difficult-to-remove solvents such as acetonitrile and ethyl acetate alongside methanol. The reliance on these complex solvent mixtures complicates the drying process, often leading to products with high residual solvent levels that struggle to meet stringent pharmacopoeial standards without extensive and energy-intensive vacuum drying cycles. Additionally, the crystal habit obtained through these older methods can be poor, resulting in filter cakes that are difficult to wash, thereby trapping mother liquor impurities and extending the overall production cycle time significantly.
The Novel Approach
The innovative methodology disclosed in the patent data fundamentally reengineers the salification and crystallization sequence to overcome these historical bottlenecks. By employing sodium isooctanoate as the specific salt-forming agent, the process achieves a direct conversion of cefpiramide acid to cefpiramide sodium, effectively eliminating the need for the intermediate amine salt formation entirely. This direct route not only reduces the number of reaction steps but also minimizes the introduction of extraneous organic bases that can act as impurities. A cornerstone of this novel approach is the adoption of acetone as a single primary solvent for the crystallization phase. The process utilizes a sophisticated four-stage addition protocol for acetone, acting as an antisolvent to precisely modulate supersaturation. This controlled addition, combined with specific stirring speeds and temperature gradients ranging from 15°C to 35°C, ensures the formation of well-defined crystals that are inherently easier to filter and dry. The result is a manufacturing process that is not only greener due to the ease of acetone recovery but also yields a product with superior physical properties, including improved flowability and significantly reduced moisture content, facilitating cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Direct Salting-Out and Controlled Crystallization
The chemical elegance of this synthesis lies in the interaction between the carboxylic acid moiety of the cefpiramide nucleus and the sodium isooctanoate. In the initial pulping stage, cefpiramide acid is suspended in a mixed solution of acetone and water, creating a heterogeneous system that is ready for transformation. Upon the addition of the sodium isooctanoate solution, an acid-base reaction occurs where the sodium ion displaces the proton on the carboxylic acid, forming the soluble sodium salt in situ. The choice of sodium isooctanoate is critical; its lipophilic octanoate counter-ion helps maintain solubility in the organic-rich phase initially, preventing premature precipitation before the system is ready for controlled crystallization. Following decolorization with activated carbon to remove trace colored impurities and potential degradation products, the solution is clarified, setting the stage for the critical crystallization event. The absence of amine salts means there is no risk of amine-related degradation pathways or the formation of difficult-to-remove amine-acid complexes, which contributes directly to the observed improvement in the high-purity API profile.
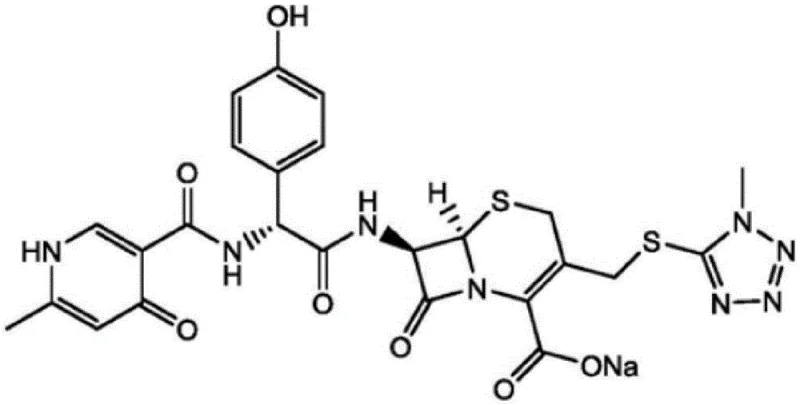
The crystallization mechanism is driven by the precise manipulation of solubility through the four-stage addition of acetone. In the first stage, a small volume of acetone is introduced to initiate nucleation, often aided by the addition of seed crystals to ensure polymorphic consistency. As the process moves to the second and third stages, increased stirring speeds (up to 350 rpm) and controlled temperature drops (from 34°C down to 25°C) manage the growth rate of the crystals. This gradual increase in antisolvent concentration prevents the "oiling out" phenomenon, where the product separates as a liquid phase rather than a solid, which is a common issue in cephalosporin processing. The final stage ensures complete precipitation while maintaining a slurry that is easy to handle. Post-filtration, the washing step employs a specific ternary mixture of methanol, water, and acetone. This tailored wash solvent is designed to dissolve surface-adhered impurities, such as residual sodium isooctanoate or trace starting materials, without redissolving the cefpiramide sodium crystal lattice itself. This selective washing capability is key to achieving the low impurity profiles demonstrated in the experimental data, specifically regarding the reduction of 1-methyl-5-mercaptotetrazole and other related substances.
How to Synthesize Cefpiramide Sodium Efficiently
Implementing this synthesis route requires strict adherence to the specified stoichiometric ratios and thermal profiles to ensure reproducibility at scale. The process begins with the preparation of the salifying agent, followed by the dissolution of the acid substrate, and culminates in the staged antisolvent crystallization. Operators must carefully monitor the stirring speeds and addition times during the four acetone dosing phases, as these parameters dictate the final crystal size distribution and filtration characteristics. While the general workflow is simplified compared to legacy methods, the precision required in the crystallization control loop is paramount for achieving the reported quality benchmarks. For a detailed breakdown of the specific operational parameters, including exact flow rates and temperature setpoints for each stage, please refer to the standardized synthesis guide below.
- Prepare sodium isooctanoate solution in acetone and dissolve cefpiramide acid in an acetone-water mixture.
- Mix the solutions, decolorize with activated carbon, and filter to obtain a clear cefpiramide sodium solution.
- Add acetone in four controlled stages to induce crystallization, followed by filtration and dual-solvent washing.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this direct salting-out technology offers compelling advantages that extend beyond mere technical specifications. The elimination of the amine salt intermediate step translates directly into a reduction in raw material consumption and a decrease in the overall processing time per batch. By removing the need for expensive organic amines and the subsequent salt exchange reagents, the bill of materials is simplified, leading to substantial cost savings in the procurement of starting materials. Furthermore, the transition to a single-solvent system based on acetone significantly streamlines the solvent recovery infrastructure. Acetone is a widely available, low-cost commodity chemical with well-established recycling protocols, unlike the more specialized and regulated solvents like acetonitrile used in older processes. This shift reduces the environmental burden associated with solvent waste disposal and lowers the energy costs associated with distilling complex azeotropic mixtures, thereby enhancing the overall economic viability of the production line.
- Cost Reduction in Manufacturing: The streamlined process architecture eliminates entire unit operations, specifically the formation and isolation of the amine salt intermediate. This reduction in processing steps decreases labor hours, equipment occupancy time, and utility consumption. Additionally, the high efficiency of the acetone-based crystallization allows for greater recovery and reuse of the mother liquor, minimizing raw material loss. The avoidance of complex solvent mixtures also reduces the capital expenditure required for specialized solvent recovery columns, allowing for a more lean and cost-effective manufacturing footprint that drives down the cost of goods sold (COGS) without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of this crystallization method improves the predictability of production schedules. Traditional methods often suffer from variable filtration times due to poor crystal morphology, which can create bottlenecks in the drying department. The superior crystal habit achieved through the four-stage acetone addition ensures rapid filtration and consistent drying times, enabling tighter turnaround times between batches. This reliability is crucial for maintaining continuous supply chains for essential antibiotics, reducing the risk of stockouts and allowing for more accurate demand forecasting. The use of common, non-restricted solvents like acetone also mitigates supply chain risks associated with the procurement of regulated or scarce chemical reagents.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, with parameters that translate linearly from laboratory to pilot and commercial scales. The green chemistry attributes of the method, particularly the low residual solvent profile and the use of a single recyclable organic solvent, align perfectly with modern environmental, social, and governance (ESG) goals. Reduced solvent waste generation lowers the burden on wastewater treatment facilities and minimizes the facility's carbon footprint. This environmental compliance not only future-proofs the manufacturing site against tightening regulations but also enhances the brand value of the final pharmaceutical product by associating it with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cefpiramide sodium preparation method. These insights are derived directly from the comparative data and process descriptions found in the patent literature, highlighting the specific improvements over prior art technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term value proposition of this manufacturing route.
Q: How does the new method improve solvent residue profiles compared to traditional amine salt routes?
A: By utilizing a single organic solvent (acetone) system for crystallization instead of complex mixtures involving methanol and acetonitrile, the new method significantly lowers residual solvent levels, particularly eliminating detectable ethyl acetate and reducing acetone residues well below pharmacopoeia limits.
Q: What is the advantage of using sodium isooctanoate over traditional transaminants?
A: Sodium isooctanoate allows for the direct conversion of cefpiramide acid to its sodium salt, bypassing the intermediate amine salt formation step. This simplification reduces process time, eliminates the need for subsequent salt exchange reactions, and improves overall product stability and color grade.
Q: Why is the four-stage addition of acetone critical in this crystallization process?
A: The staged addition of acetone precisely controls the supersaturation level of the solution. This prevents rapid, uncontrolled precipitation which can trap impurities or form oils, ensuring the growth of uniform, easily filterable crystals with high purity and consistent particle size distribution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpiramide Sodium Supplier
The technical advancements detailed in this report underscore the potential for significant process intensification in cephalosporin manufacturing. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative patent technologies into robust, commercial-scale realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of direct salting-out and controlled crystallization are fully realized in large-volume manufacturing. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the highest international standards for residual solvents and related substances, providing our partners with unwavering confidence in product quality and consistency.
We invite global pharmaceutical partners to engage with us to explore how this optimized synthesis route can enhance your supply chain resilience. By leveraging our technical expertise, you can achieve meaningful efficiencies and secure a stable source of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your next project milestone.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
