Revolutionizing Vitamin D Precursor Manufacturing via Streamlined Iodination Technology
Revolutionizing Vitamin D Precursor Manufacturing via Streamlined Iodination Technology
The pharmaceutical industry's relentless pursuit of efficient synthetic routes for vital nutrients has found a significant breakthrough in the domain of Vitamin D derivatives. Patent CN111138512A introduces a highly optimized methodology for synthesizing 25-hydroxycholesterol intermediates, which serve as the critical backbone for producing active forms of Vitamin D such as calcitriol and calcifediol. This technology addresses long-standing bottlenecks in the production of these high-value steroidal compounds by replacing cumbersome multi-step sequences with a direct, high-yielding transformation. For R&D directors and process chemists, this represents a pivotal shift towards more atom-economical and operationally simple protocols that can be seamlessly integrated into existing manufacturing lines. The ability to access these key intermediates with greater efficiency not only accelerates drug development timelines but also establishes a more robust foundation for the global supply of essential hormonal therapies.
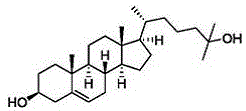
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 25-hydroxycholesterol has been plagued by excessive step counts and harsh reaction conditions that compromise overall yield and operational safety. Traditional pathways often commence with stigmasterol, necessitating a laborious sequence involving sulfonylation, substitution, and notably, ozonolysis to cleave the side chain. As illustrated in the prior art reaction schemes, converting the resulting alcohol into a reactive leaving group typically requires additional activation steps using reagents like WCl before final iodination can occur. This multi-stage approach inherently accumulates material losses at every transfer and purification stage, leading to suboptimal mass balance and increased waste generation. Furthermore, the reliance on ozone generators and cryogenic conditions for ozonolysis introduces significant capital expenditure and safety hazards, making scale-up a complex engineering challenge for procurement and supply chain teams managing large-volume production.
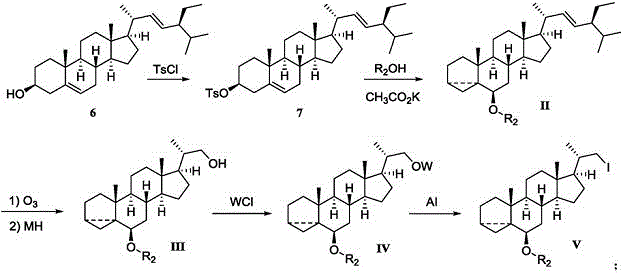
The Novel Approach
In stark contrast to the convoluted legacy processes, the methodology disclosed in CN111138512A offers a radical simplification by enabling the direct conversion of the hydroxyl-bearing intermediate (Compound III) to the corresponding iodide (Compound V) in a single operational step. This innovative route bypasses the need for separate activation and halogen exchange stages, effectively collapsing what was previously a two-step sequence into one streamlined reaction. By utilizing a specific combination of triphenylphosphine, imidazole, and elemental iodine, the process achieves high conversion rates under remarkably mild thermal conditions. This reduction in synthetic complexity translates directly to shorter cycle times and reduced solvent consumption, providing a clear competitive advantage for manufacturers seeking to optimize their cost structures. The elegance of this approach lies in its ability to maintain the stereochemical integrity of the steroid nucleus while efficiently functionalizing the side chain, ensuring that downstream coupling reactions proceed with high fidelity.
Mechanistic Insights into Triphenylphosphine-Mediated Iodination
The core chemical innovation driving this process is the in situ generation of a reactive phosphorus-iodine species that facilitates the nucleophilic substitution of the hydroxyl group. When triphenylphosphine reacts with elemental iodine in the presence of imidazole, it forms a highly electrophilic intermediate capable of activating the alcohol oxygen. This activation converts the poor leaving group (hydroxyl) into a superior leaving group, allowing the iodide anion to displace it via an SN2-type mechanism. The inclusion of imidazole serves a dual purpose: it acts as a base to neutralize the acidic byproducts generated during the reaction and helps to stabilize the reactive intermediates, thereby preventing the degradation of the sensitive steroidal framework. This mechanistic pathway is particularly advantageous for complex molecules like bisnorcholine derivatives, where harsh acidic conditions could lead to unwanted rearrangements or elimination side products.
Impurity control is meticulously managed through precise thermal regulation and reagent dosing strategies. The patent specifies that the reaction is exothermic, and uncontrolled heat release can lead to the formation of unidentified byproducts that complicate purification. To mitigate this, the protocol mandates the batch-wise addition of elemental iodine at temperatures ranging from -20°C to 40°C. This controlled addition profile ensures that the concentration of the reactive iodinating species remains within an optimal window, minimizing side reactions such as over-iodination or oxidation of other sensitive functional groups on the steroid ring. Consequently, the crude reaction mixture exhibits a much cleaner profile, which significantly reduces the burden on downstream purification units and enhances the overall recovery of the target pharmaceutical intermediate.
How to Synthesize 25-Hydroxycholesterol Intermediate Efficiently
Implementing this synthesis requires careful attention to solvent selection and reagent stoichiometry to maximize the yield of the target iodide. The process begins by dissolving the starting alcohol, specifically 6β-methoxy-3β,5α-cyclobisnorcholine-22-ol or its ethoxy analogue, in a non-polar organic solvent such as dichloroethane, toluene, or dichloromethane. Once the substrate is fully solubilized, triphenylphosphine and imidazole are introduced to the reaction vessel. The critical step follows, where elemental iodine is added portion-wise to maintain the reaction temperature within the specified range of -20°C to 40°C. The reaction is typically allowed to proceed for 1 to 3 hours, after which the mixture is worked up to isolate the pure iodide product. Detailed standardized operating procedures for this transformation are provided below to ensure reproducibility and safety.
- Dissolve the starting alcohol (Compound III) in a non-polar organic solvent such as dichloroethane or toluene.
- Add triphenylphosphine and imidazole to the solution, ensuring complete dissolution before proceeding.
- Add elemental iodine in batches at a controlled temperature between -20°C and 40°C, reacting for 1-3 hours to form the target iodide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this streamlined synthetic route offers profound economic and logistical benefits that extend far beyond simple yield improvements. By eliminating multiple unit operations associated with the traditional activation and halogenation steps, manufacturers can drastically reduce the consumption of solvents, reagents, and energy. This reduction in material intensity directly correlates to a lower cost of goods sold (COGS), allowing companies to remain competitive in the pricing of high-value Vitamin D derivatives. Moreover, the simplified workflow reduces the requirement for specialized equipment such as ozone generators, thereby lowering capital investment barriers and maintenance costs for production facilities.
- Cost Reduction in Manufacturing: The consolidation of multiple reaction steps into a single pot operation significantly lowers processing costs by reducing labor hours and utility consumption. Eliminating the need for distinct activation reagents and the associated workup procedures removes entire categories of raw material expenses from the bill of materials. Additionally, the high selectivity of the reaction minimizes the loss of expensive steroidal starting materials, ensuring that a greater proportion of input mass is converted into saleable product. This efficiency gain is compounded by the reduced volume of waste streams requiring treatment, leading to substantial savings in environmental compliance and disposal fees.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the number of potential failure points in the manufacturing process, leading to more consistent batch-to-batch quality and reliable delivery schedules. The use of common, commercially available reagents like triphenylphosphine and iodine mitigates the risk of supply disruptions often associated with specialized or hazardous chemicals. Furthermore, the robustness of the reaction conditions allows for greater flexibility in production scheduling, as the process is less sensitive to minor variations in ambient conditions compared to cryogenic ozonolysis. This stability ensures that manufacturers can meet fluctuating market demands without compromising on product availability or lead times.
- Scalability and Environmental Compliance: The mild thermal profile and absence of hazardous ozone gas make this process exceptionally well-suited for large-scale commercial production. Scaling up exothermic reactions is traditionally challenging, but the batch-wise addition protocol described in the patent provides a safe and controllable method for managing heat release in large reactors. From an environmental perspective, the reduction in solvent usage and the elimination of ozone-depleting or hazardous byproducts align with increasingly stringent global regulations on green chemistry. This alignment not only future-proofs the manufacturing asset against regulatory changes but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this novel iodination technology. These insights are derived directly from the experimental data and process descriptions contained within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is essential for process engineers and quality assurance teams planning to integrate this method into their production portfolios.
Q: What are the critical reaction conditions for the iodination step?
A: The reaction requires strict temperature control between -20°C and 40°C. Elemental iodine must be added in batches to manage the exothermic nature of the reaction and prevent impurity formation.
Q: How does this method improve upon traditional stigmasterol routes?
A: Traditional routes involve multiple steps including ozonolysis and activation. This novel method converts the alcohol intermediate directly to the iodide in a single step, significantly reducing processing time and material loss.
Q: What purification methods are recommended for the final iodide product?
A: The patent recommends removing the solvent via reduced pressure distillation followed by silica gel column chromatography using petroleum ether as the eluent to achieve high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 25-Hydroxycholesterol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in patent literature can be reliably replicated on an industrial scale. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 25-hydroxycholesterol intermediate meets the exacting standards required for pharmaceutical applications. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal controls necessary for this advanced synthesis.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge technology for their Vitamin D supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your next project is built on a foundation of scientific excellence and commercial viability.
