Revolutionizing Tulathromycin Production: A Safer, Three-Step Metal-Coordinated Synthesis Route
The pharmaceutical landscape for veterinary antibiotics is constantly evolving, driven by the need for safer, more efficient, and cost-effective manufacturing processes. Patent CN103641869A introduces a groundbreaking synthetic method for Tulathromycin (CAS 217500-96-4), a third-generation macrolide antibiotic widely used for treating respiratory diseases in cattle and swine. This innovation addresses critical bottlenecks in the conventional production of this high-value active pharmaceutical ingredient (API). Unlike traditional routes that rely on hazardous hydrogenation and expensive protecting groups, this novel approach utilizes a sophisticated metal-coordination strategy to achieve high selectivity. By leveraging the specific interaction between metal salts and hydroxyl groups on the Azithromycin A macrocycle, the process enables direct oxidation without the need for cumbersome protection-deprotection sequences. This technical leap not only enhances operational safety by removing high-pressure hydrogen requirements but also streamlines the supply chain through a simplified three-step workflow. For global procurement teams and R&D directors, this patent represents a pivotal shift towards greener, more economical antibiotic manufacturing.
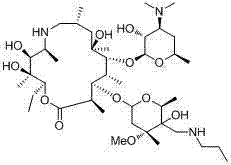
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tulathromycin has been plagued by significant safety and economic inefficiencies inherent to the standard protection-deprotection strategies. Conventional methodologies typically necessitate the use of benzyloxy carbonyl chloride (Cbz-Cl) to protect the 4-hydroxyl group before oxidizing the 5-hydroxyl position. This adds unnecessary synthetic steps, increasing the overall cycle time and reducing the final yield due to cumulative losses at each stage. Furthermore, the removal of the Cbz protecting group traditionally requires a palladium on carbon (Pd/C) catalytic system under a high-pressure hydrogen atmosphere. This reliance on noble metal catalysts introduces substantial raw material costs and creates a vulnerability in the supply chain due to the fluctuating price of palladium. More critically, the use of high-pressure hydrogen gas poses severe safety risks in a manufacturing environment, requiring specialized explosion-proof infrastructure and rigorous safety protocols that drive up capital expenditure. Additionally, palladium catalysts are prone to poisoning by sulfur-containing impurities often present in macrolide intermediates, leading to inconsistent reaction performance and potential batch failures.
The Novel Approach
The synthetic route disclosed in patent CN103641869A fundamentally reengineers this process by eliminating the need for Cbz-Cl protection and Pd/C hydrogenation entirely. Instead, it employs a clever in situ metal-coordination mechanism where a metal salt selectively complexes with the 4-hydroxyl group and its adjacent alpha-carbon. This temporary coordination effectively masks the 4-position, allowing for the selective oxidation of the 5-hydroxyl group to a ketone using an N-chlorosuccinimide and dimethyl sulfide system. Following oxidation, the ketone is converted into an epoxide via a sulfur ylide reaction, which is subsequently opened by tri-n-propylamine to yield the final Tulathromycin structure. This streamlined three-step sequence operates under much milder conditions, avoiding the ultra-low temperatures often required in prior art. The result is a robust, scalable process that significantly reduces energy consumption and eliminates the hazards associated with high-pressure hydrogenation, making it an ideal candidate for reliable veterinary drug supplier operations seeking to optimize their manufacturing footprint.
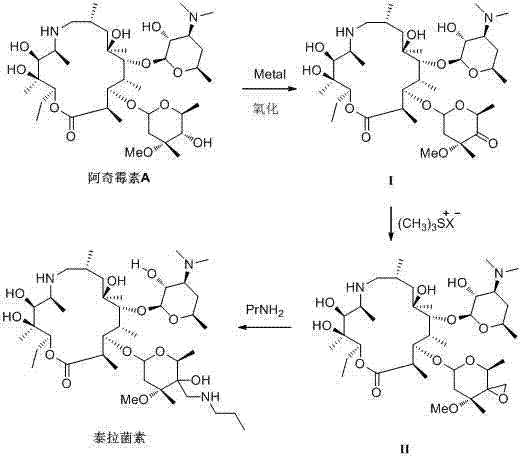
Mechanistic Insights into Metal-Coordinated Selective Oxidation
The core scientific breakthrough of this methodology lies in the precise control of regioselectivity during the oxidation phase, achieved through the formation of a transient metal complex. As illustrated in the mechanistic diagrams, the addition of specific metal salts—such as lithium, cobalt, nickel, or zinc salts—to the reaction mixture containing Azithromycin A facilitates the formation of a stable coordination complex. In this complex, the metal ion simultaneously coordinates with the oxygen atom of the 4-hydroxyl group and the nitrogen atom of the desosamine sugar or adjacent carbon centers. This steric and electronic shielding effectively deactivates the 4-hydroxyl group towards oxidation, despite its inherent reactivity. Consequently, when the oxidizing agent, generated from N-chlorosuccinimide and dimethyl sulfide, is introduced, it selectively targets the exposed 5-hydroxyl group. This level of chemoselectivity is crucial for maintaining the integrity of the macrolide ring and preventing the formation of difficult-to-remove regioisomeric impurities. The ability to achieve this differentiation without covalent protecting groups represents a significant advancement in fine chemical intermediates synthesis, offering a cleaner reaction profile and simplifying downstream purification.
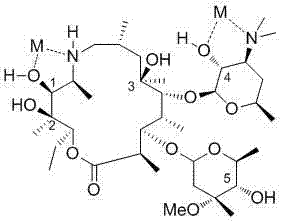
Furthermore, the subsequent transformation of the ketone intermediate into the final product demonstrates excellent control over stereochemistry and impurity profiles. The conversion of the ketone to an epoxide using a sulfur ylide, generated from trimethylammonium halogenation sulfonium and a strong base, proceeds with high efficiency. The choice of base and solvent plays a pivotal role here; the patent details the use of various bases like potassium tert-butoxide or sodium hydride in solvents such as dichloromethane or tetrahydrofuran to optimize the yield of the epoxy intermediate. The final ring-opening step with tri-n-propylamine is equally critical, as it installs the characteristic amino-propyl side chain of Tulathromycin. The reaction conditions are tuned to ensure complete conversion while minimizing side reactions that could lead to polymeric byproducts. This meticulous control over each mechanistic step ensures that the final API meets stringent purity specifications required for veterinary applications, thereby reducing the burden on quality control laboratories and ensuring consistent batch-to-batch quality.
How to Synthesize Tulathromycin Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to maximize yield and safety. The process begins with the preparation of the oxidizing species by reacting N-chlorosuccinimide with dimethyl sulfide at low temperatures, followed by the addition of the Azithromycin A-metal salt complex. The subsequent epoxidation and ring-opening steps utilize common organic solvents and reagents that are readily available on the global market, facilitating easy technology transfer. The detailed procedural nuances, including specific molar ratios and workup procedures, are critical for achieving the reported high yields. For process chemists looking to adopt this methodology, understanding the interplay between the metal salt choice and the oxidation efficiency is paramount. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Selective Oxidation: React Azithromycin A with N-chlorosuccinimide and dimethyl sulfide in the presence of a metal salt (e.g., lithium, zinc, or cobalt salts) to form a coordination complex that protects the 4-hydroxyl group while oxidizing the 5-hydroxyl to a ketone.
- Epoxidation: Treat the resulting ketone intermediate with a trimethylammonium halogenation sulfonium salt and a strong base to generate a sulfur ylide, which converts the ketone into an epoxy compound.
- Ring Opening and Crystallization: React the epoxy compound with tri-n-propylamine in an alcoholic solvent to open the epoxide ring, followed by solvent recovery, concentration, and recrystallization to obtain pure Tulathromycin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-coordinated synthesis route offers profound advantages for procurement managers and supply chain directors focused on cost reduction in antibiotic manufacturing. The most immediate impact is the drastic simplification of the supply chain raw material list. By removing the requirement for benzyloxy carbonyl chloride and palladium catalysts, manufacturers can bypass the volatility associated with noble metal markets and the regulatory complexities of handling controlled precursors. This simplification translates directly into a more predictable cost structure and reduced exposure to supply disruptions. Furthermore, the ability to recover and reuse dimethyl sulfide, a key reagent in the oxidation step, contributes to substantial cost savings and aligns with modern sustainability goals. The elimination of the hydrogenation step also removes the need for specialized high-pressure reactors, allowing production to be scaled up in standard glass-lined or stainless steel vessels, thereby lowering capital investment barriers for contract development and manufacturing organizations (CDMOs).
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in unit operations and the elimination of expensive catalysts. Traditional routes involving palladium-catalyzed hydrogenation incur high costs not only for the metal itself but also for the filtration and recovery systems required to meet residual metal limits in the final API. By circumventing this step entirely, the new method removes a significant cost center. Additionally, the shorter three-step sequence reduces solvent consumption, labor hours, and utility usage per kilogram of product. The qualitative improvement in process efficiency means that resources are utilized more effectively, leading to a lower cost of goods sold (COGS) without compromising on the quality of the high-purity veterinary drug produced.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals rather than specialized reagents. The metal salts used for coordination, such as zinc chloride or lithium hydroxide, are abundant and sourced from stable supply chains, unlike palladium which is subject to geopolitical constraints. Moreover, the operational safety improvements—specifically the avoidance of high-pressure hydrogen—reduce the risk of unplanned plant shutdowns due to safety incidents or regulatory inspections. This reliability ensures a consistent flow of commercial scale-up of complex macrolides to the market, meeting the demands of the global animal health industry without the delays often associated with hazardous chemical processing. The robustness of the reaction conditions also allows for greater flexibility in manufacturing scheduling.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the mild reaction conditions and the absence of extreme parameters. The oxidation and epoxidation steps can be performed at temperatures ranging from ambient to moderately elevated levels, which are easily manageable in large-scale reactors. From an environmental standpoint, the process generates less hazardous waste compared to traditional methods. The recovery of dimethyl sulfide minimizes volatile organic compound (VOC) emissions, and the lack of heavy metal waste streams simplifies wastewater treatment protocols. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturer, making it an attractive partner for environmentally conscious pharmaceutical companies seeking reducing lead time for high-purity veterinary drugs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Tulathromycin synthesis method. These insights are derived directly from the comparative data and experimental results presented in patent CN103641869A, providing clarity on why this route is gaining traction in the industry. Understanding these specifics is essential for stakeholders evaluating the feasibility of switching from legacy processes to this advanced metal-coordination technology. The answers highlight the balance between chemical innovation and practical manufacturing realities.
Q: Why is the metal-coordination method superior to traditional Cbz-Cl protection?
A: Traditional methods require benzyloxy carbonyl chloride (Cbz-Cl) protection and subsequent removal using a Pd/C-H2 system, which involves expensive palladium catalysts and dangerous high-pressure hydrogen. The metal-coordination method achieves selectivity in situ without these hazardous and costly steps.
Q: How does this process improve safety in large-scale manufacturing?
A: By eliminating the need for ultra-low temperatures and high-pressure hydrogenation equipment, the process significantly reduces operational hazards. Additionally, the reagents used, such as dimethyl sulfide, can be recovered and reused, minimizing waste and exposure risks.
Q: What represents the primary cost-saving driver in this synthetic route?
A: The primary cost drivers are the elimination of noble metal catalysts (Palladium) and the reduction of synthetic steps from a multi-step protection/deprotection sequence to a direct three-step transformation, which lowers both material consumption and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tulathromycin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in patent CN103641869A for the production of high-value veterinary antibiotics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our state-of-the-art facilities are equipped to handle the specific requirements of macrolide synthesis, including the precise temperature control and metal handling capabilities necessary for this coordination chemistry. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Tulathromycin meets the highest international standards for animal health applications. Our commitment to technical excellence ensures that the theoretical benefits of this patent—safety, efficiency, and cost-effectiveness—are fully realized in the final commercial product.
We invite global pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical team is ready to provide specific COA data and route feasibility assessments to demonstrate how this metal-coordinated synthesis can optimize your procurement strategy. Contact our technical procurement team today to discuss how we can support your long-term goals for reliable, high-quality Tulathromycin supply.
