Scalable Industrial Synthesis of Ropinirole Intermediates via Novel Nitration-Cyclization Route
Scalable Industrial Synthesis of Ropinirole Intermediates via Novel Nitration-Cyclization Route
The pharmaceutical landscape for Parkinson's disease treatment relies heavily on the consistent supply of high-quality dopamine agonists, with Ropinirole standing as a cornerstone therapeutic agent. A pivotal advancement in the manufacturing of this critical active pharmaceutical ingredient (API) is detailed in patent CN100457732C, which outlines a robust, eight-step synthetic pathway designed to overcome the limitations of prior art. This innovative methodology utilizes commercially available phenylacetic acid or its 4-substituted derivatives as the foundational starting material, initiating a sequence that includes nitration, amidation, selective reductions, and a sophisticated cyclization protocol. For global procurement teams and R&D directors, this patent represents a significant leap forward in process chemistry, offering a route that is not only chemically elegant but also inherently safer and more economically viable than historical alternatives. By shifting away from hazardous reagents and complex multi-step sequences, this technology establishes a new benchmark for the reliable production of Ropinirole intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ropinirole has been plagued by significant operational hazards and inefficiencies that complicate supply chain stability. Early methodologies, such as those disclosed in US4452808, relied heavily on the use of sodium cyanide, a highly toxic reagent that necessitates rigorous safety protocols, specialized waste treatment infrastructure, and increases the overall environmental footprint of the manufacturing process. Furthermore, these legacy routes often involved upwards of ten discrete reaction steps, each introducing potential yield losses and impurity profiles that are difficult to control at scale. Alternative approaches reported in EP300614 attempted to streamline the process but introduced new bottlenecks, specifically requiring photochemical bromination steps that demand specialized irradiation equipment and result in low yields due to competing elimination by-products. These technical barriers create substantial friction for a reliable agrochemical intermediate supplier or pharma partner, as they translate directly into higher production costs, longer lead times, and increased regulatory scrutiny regarding residual toxicants.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach described in the patent data leverages a direct nitration strategy on readily accessible phenylacetic acid scaffolds to construct the core architecture of the molecule. This route elegantly bypasses the need for toxic cyanide sources and eliminates the dependency on light-sensitive bromination reactions, thereby simplifying the engineering requirements for commercial scale-up. The process flows logically from nitration to amidation, followed by a strategic reduction sequence that installs the diisopropylaminoethyl side chain early in the synthesis, ensuring that the bulky amine functionality is present during the critical ring-closing steps. This structural foresight minimizes steric hindrance issues during the final cyclization and reduction phases. As illustrated in the comprehensive reaction scheme below, the pathway is linear and convergent, utilizing standard unit operations that are easily replicated in multipurpose chemical plants.
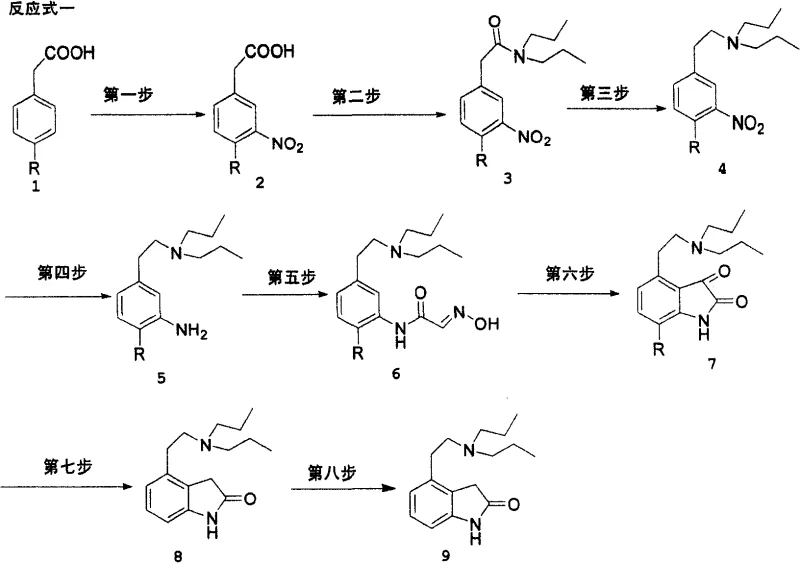
The visual representation of the full synthetic pathway highlights the seamless transition from simple aromatic acids to the complex indolinone core, demonstrating how each functional group transformation is optimized for maximum atom economy and minimal waste generation. This streamlined design is crucial for achieving cost reduction in API intermediate manufacturing, as it reduces the total number of isolation and purification steps required before reaching the final drug substance.
Mechanistic Insights into Isatin Cyclization and Carbonyl Reduction
A critical juncture in this synthetic route is the formation of the isatin intermediate, which serves as the precursor to the final indolinone structure of Ropinirole. This transformation involves the condensation of the aniline derivative with chloral hydrate and hydroxylamine, followed by an acid-mediated cyclization. The mechanistic precision required here is paramount; the reaction conditions must be carefully controlled to ensure regioselective ring closure without degrading the sensitive amine side chain. The patent specifies the use of polyphosphoric acid or strong mineral acids at temperatures between 50°C and 140°C to drive this cyclization. This acidic environment facilitates the dehydration and intramolecular attack necessary to form the five-membered lactam ring, a structural motif essential for the biological activity of the final dopamine agonist. The ability to perform this cyclization under relatively mild thermal conditions compared to traditional fusion methods represents a significant process advantage, preserving the integrity of the molecule while driving the reaction to completion.
Following the successful construction of the isatin core, the final reduction of the carbonyl group to the methylene functionality completes the synthesis of the Ropinirole backbone. This step often employs catalytic hydrogenation using Raney Nickel or similar catalysts, which simultaneously effects the reduction of the ketone and, if a halogen substituent was used in the starting material, the dehalogenation of the aromatic ring. This dual-functionality of the reduction step is a masterstroke of process design, as it accomplishes two distinct chemical transformations in a single operation, thereby reducing solvent usage, processing time, and overall manufacturing costs. The mechanistic understanding of this hydrogenolysis allows for fine-tuning of pressure and temperature parameters to maximize yield while minimizing the formation of over-reduced by-products, ensuring a high-purity API intermediate suitable for subsequent salt formation.
How to Synthesize Ropinirole Efficiently
The execution of this synthesis requires strict adherence to the optimized reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the controlled nitration of the phenylacetic acid derivative, where temperature management is critical to prevent polynitration or oxidative degradation of the substrate. Subsequent steps involve the activation of the carboxylic acid, typically via thionyl chloride or carbodiimide coupling agents, followed by amidation with dipropylamine. The reduction of the resulting amide to the amine is achieved using powerful hydride donors like lithium aluminum hydride or borane complexes, necessitating anhydrous conditions and careful quenching protocols. For a detailed breakdown of the specific reagents, stoichiometry, and workup procedures for each stage, please refer to the standardized guide below.
- Nitration of 4-substituted phenylacetic acid using nitric acid or nitronium salts in acidic media to form 3-nitro derivatives.
- Conversion of the carboxylic acid to an amide via acyl chloride or coupling agents, followed by reduction to the corresponding amine.
- Reduction of the nitro group to an aniline, followed by condensation with chloral hydrate and hydroxylamine to form the isatin core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend far beyond simple chemical yield. The primary advantage lies in the radical simplification of the raw material portfolio; by utilizing commodity chemicals like phenylacetic acid and dipropylamine, manufacturers can decouple their supply chains from volatile markets for exotic or highly regulated precursors. This shift significantly enhances supply chain reliability, as the sourcing of starting materials becomes less susceptible to geopolitical disruptions or single-supplier bottlenecks. Furthermore, the elimination of sodium cyanide and photochemical steps removes entire categories of regulatory compliance costs and hazardous waste disposal fees, contributing to substantial cost savings in the overall production budget. The robustness of the reaction conditions, which operate within standard temperature and pressure ranges for fine chemical processing, ensures that the technology can be transferred to existing manufacturing facilities without the need for capital-intensive retrofitting or specialized equipment installation.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom efficiency and the avoidance of expensive, toxic reagents. By replacing cyanide-based chemistry with safe nitration and reduction sequences, the process eliminates the need for costly heavy metal scavengers and complex effluent treatment systems associated with cyanide detoxification. Additionally, the convergence of the dehalogenation and carbonyl reduction into a single catalytic step reduces the consumption of hydrogen and catalyst, lowering the variable cost per kilogram of the final product. These efficiencies compound over large production volumes, resulting in a markedly lower cost of goods sold (COGS) compared to legacy manufacturing routes.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures a stable and continuous supply of inputs, mitigating the risk of production stoppages due to raw material shortages. The synthetic route is designed with flexibility in mind, allowing for the use of various halogenated or non-halogenated starting materials depending on market availability, which provides procurement teams with multiple sourcing options. This adaptability is crucial for maintaining business continuity in the face of global supply chain fluctuations, ensuring that the delivery of high-purity pharmaceutical intermediates remains uninterrupted regardless of external market pressures.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior due to its benign reagent profile and mild operating conditions. The absence of genotoxic alkylating agents and highly toxic cyanides simplifies the environmental permitting process and reduces the long-term liability associated with hazardous waste storage. The process is inherently scalable, as demonstrated by the patent examples which show consistent yields and purity profiles that are reproducible from gram to kilogram scales. This scalability ensures that the technology can meet the growing global demand for Ropinirole without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is essential for making informed decisions about technology transfer and process optimization.
Q: What are the advantages of this Ropinirole synthesis route over traditional methods?
A: This route avoids highly toxic reagents like sodium cyanide used in older methods and eliminates the need for specialized photochemical bromination equipment, significantly improving safety and scalability.
Q: What is the purity profile of the intermediates generated in this process?
A: The patent data indicates that key intermediates, such as the final Ropinirole base, can achieve purity levels exceeding 99% after standard recrystallization, meeting stringent pharmaceutical specifications.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions utilize mild temperatures ranging from 0°C to 140°C and employ commercially available solvents and reagents, making it highly adaptable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ropinirole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of this nitration-cyclization route are managed with precision. We are committed to delivering Ropinirole intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical instrumentation to verify identity and assay. Our facility is equipped to handle the specific safety requirements of nitration and hydrogenation reactions, providing a secure and compliant environment for the manufacture of this critical Parkinson's disease medication.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the quality and consistency of our output against your internal standards before committing to a long-term supply agreement.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
