Advanced Pterostilbene Manufacturing: High-Purity Synthesis for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for high-value bioactive compounds, and Patent CN1269782C presents a significant breakthrough in the synthesis of Pterostilbene. This potent phytoestrogen, structurally related to resveratrol but with superior bioavailability and antifungal properties, has garnered immense interest for its potential in treating diabetes and various cancers. The patented methodology introduces a streamlined, four-step sequence that bypasses the notorious inefficiencies of traditional Wittig olefinations. By leveraging a base-catalyzed condensation followed by a catalytic reduction and a final diazotization-hydrolysis sequence, this route achieves high conversion rates with minimal environmental impact. For R&D directors and procurement specialists alike, understanding this chemistry is crucial for securing a reliable supply of this critical intermediate.
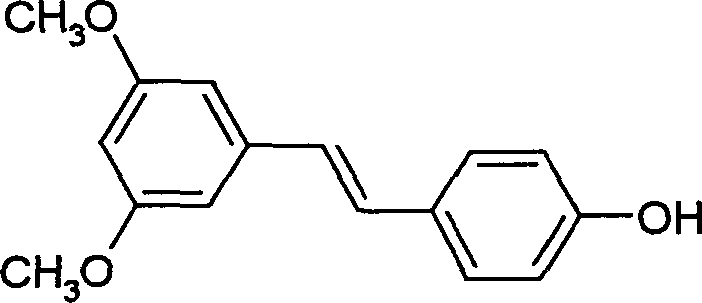
Pterostilbene is widely recognized for its moderate inhibitory activity against COX-1 and strong antibacterial effects, making it a versatile candidate for drug development. However, accessing high-purity material has historically been hindered by complex extraction processes from natural sources or inefficient synthetic routes. The innovation detailed in CN1269782C addresses these bottlenecks by establishing a fully synthetic pathway that is not only chemically elegant but also commercially viable. The process eliminates the need for protecting group strategies that plague other methods, directly installing the phenolic hydroxyl group in the final step through a controlled hydrolysis of a diazonium salt. This strategic design ensures that the final product meets stringent purity specifications required for pharmaceutical applications without the burden of excessive downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of stilbene derivatives like Pterostilbene has relied heavily on the Wittig reaction, a method fraught with significant operational challenges for large-scale manufacturing. The primary drawback of the Wittig approach is the generation of cis-trans isomeric mixtures, which necessitates difficult and costly separation procedures to isolate the biologically active trans-isomer. Furthermore, the Wittig reagents often require the use of benzyl halides, which are inherently unstable and prone to self-polymerization during storage, leading to inconsistent batch quality and supply chain disruptions. Additionally, conventional routes frequently employ bulky protecting groups, such as tert-butyldimethylsiloxy (TBDMS), to mask the phenolic hydroxyl functionality during the olefination step. This adds two extra steps—protection and deprotection—to the synthesis, drastically increasing raw material consumption, waste generation, and overall production time, rendering the process economically unfeasible for cost-sensitive markets.
The Novel Approach
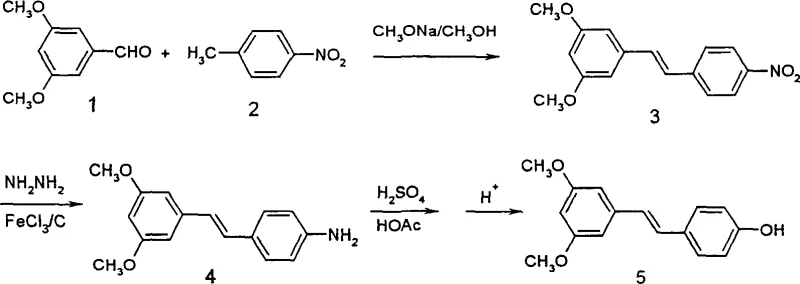
In stark contrast, the methodology disclosed in Patent CN1269782C utilizes a Knoevenagel-type condensation between 3,5-dimethoxybenzaldehyde and p-nitrotoluene to construct the carbon skeleton. This reaction is highly selective for the thermodynamically stable trans-isomer, effectively eliminating the isomer separation nightmare associated with the Wittig reaction. The subsequent transformation involves a catalytic reduction of the nitro group to an amine, followed by a classic diazotization and hydrolysis sequence to reveal the phenol. This linear progression avoids the use of unstable halides and cumbersome protecting groups entirely. The result is a process characterized by short reaction times, high conversion rates, and a simplified post-treatment workflow that relies on straightforward recrystallization rather than complex chromatography. This represents a paradigm shift in how we approach the manufacturing of complex stilbene intermediates.
Mechanistic Insights into FeCl3-Catalyzed Reduction and Diazotization
The core of this synthetic strategy lies in the precise control of reaction conditions to maximize selectivity and yield. The initial condensation step utilizes sodium methoxide in methanol at controlled temperatures between 50°C and 60°C. Under these basic conditions, the acidic methyl protons of p-nitrotoluene are deprotonated to form a carbanion, which attacks the carbonyl carbon of the aldehyde. The electron-withdrawing nature of the nitro group enhances the acidity of the methyl protons, facilitating the formation of the double bond with exclusive trans-selectivity due to steric hindrance in the transition state. This mechanistic feature is critical for R&D teams, as it guarantees a high-purity intermediate without the need for isomeric enrichment, setting a solid foundation for the subsequent steps.
Following the formation of the nitro-stilbene scaffold, the reduction step employs hydrazine hydrate in the presence of activated carbon and a Lewis acid catalyst, specifically ferric chloride hexahydrate (FeCl3·6H2O). This catalytic system is remarkably efficient, operating at reflux temperatures in ethanol to reduce the nitro group to an amine with yields exceeding 97%. The use of FeCl3 likely facilitates electron transfer or activates the hydrazine species, allowing for rapid conversion while minimizing side reactions. The final transformation involves converting the amine into a diazonium salt using sodium nitrite and sulfuric acid under strict ice-salt bath conditions to prevent premature decomposition. Subsequent hydrolysis of this diazonium intermediate in a boiling copper sulfate or dilute sulfuric acid solution cleanly installs the hydroxyl group, completing the synthesis of Pterostilbene with high fidelity and minimal byproduct formation.
How to Synthesize Pterostilbene Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control, particularly during the diazotization phase where thermal instability can compromise safety and yield. The patent outlines a robust protocol where molar ratios are optimized to drive reactions to completion while minimizing excess reagent waste. For instance, the condensation step prefers a slight excess of p-nitrotoluene and base to ensure full consumption of the valuable aldehyde. The reduction step benefits from the heterogeneous catalysis provided by activated carbon, which also aids in the adsorption of colored impurities, resulting in a cleaner crude product that is easily purified by recrystallization from ethanol/water mixtures. These operational details are vital for process chemists aiming to translate this laboratory-scale success into a commercial reality.
- Condense 3,5-dimethoxybenzaldehyde with p-nitrotoluene using sodium methoxide in methanol at 50-60°C to form 3,5-dimethoxy-4'-nitrostilbene.
- Reduce the nitro group to an amine using hydrazine hydrate and activated carbon with a Lewis acid catalyst (FeCl3) in ethanol at 75-82°C.
- Perform diazotization of the amine intermediate using sodium nitrite and sulfuric acid under ice-salt bath conditions, followed by hydrolysis to yield Pterostilbene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from Wittig chemistry to this condensation-reduction-hydrolysis sequence offers profound economic and logistical benefits. The elimination of protecting group manipulations alone translates to a drastic reduction in raw material costs and solvent usage. By removing the need for silyl chlorides and the associated fluoride or acid deprotection reagents, the process becomes significantly leaner. Furthermore, the reliance on stable, commodity-grade starting materials like p-nitrotoluene and 3,5-dimethoxybenzaldehyde mitigates the risk of supply interruptions caused by the degradation of sensitive reagents. This stability ensures consistent lead times and allows for better inventory planning, which is essential for maintaining continuous production schedules in a fast-paced pharmaceutical environment.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the simplified purification workflow. Traditional methods often require column chromatography to separate cis/trans isomers, a technique that is prohibitively expensive and difficult to scale. This new route produces the trans-isomer selectively, allowing for purification via simple recrystallization using low-cost solvents like ethanol and water. Additionally, the high conversion rates observed in the reduction step mean less raw material is wasted, directly lowering the cost of goods sold (COGS) and improving overall margin potential for high-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust intermediates that do not degrade rapidly upon storage. Unlike benzyl halides which polymerize and become unusable over time, the nitro-stilbene and amino-stilbene intermediates generated in this process are chemically stable solids. This stability allows manufacturers to produce and stockpile key intermediates in advance, decoupling the production of the final API intermediate from immediate raw material availability. Such flexibility is invaluable for mitigating risks associated with global logistics delays or sudden spikes in demand for Pterostilbene-based therapeutics.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process is markedly superior. The avoidance of heavy metal phosphine oxides (typical Wittig byproducts) and silicon-containing waste simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. The use of hydrazine hydrate, while requiring careful handling, is a well-understood reagent in industrial settings, and the catalytic nature of the iron salt minimizes heavy metal load in the waste stream. These factors facilitate easier regulatory approval and align with modern green chemistry principles, making the facility more attractive to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is key for stakeholders evaluating its adoption. The following questions address common concerns regarding scalability, purity, and safety, drawing directly from the experimental data and beneficial effects reported in the patent documentation. These insights are intended to provide clarity for technical teams assessing the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this synthesis method improve upon the traditional Wittig reaction?
A: Unlike the Wittig reaction which often produces difficult-to-separate cis/trans mixtures and requires unstable benzyl halides, this method utilizes a selective condensation that favors the trans-isomer exclusively, simplifying purification and improving overall yield stability.
Q: What are the key advantages regarding raw material stability?
A: The process relies on stable starting materials like 3,5-dimethoxybenzaldehyde and p-nitrotoluene, avoiding the self-polymerization issues associated with benzyl halides used in alternative routes, thereby ensuring consistent supply chain reliability.
Q: Is the purification process scalable for industrial production?
A: Yes, the method employs standard recrystallization techniques using environmentally friendly solvents like ethanol/water systems rather than complex chromatographic separations, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pterostilbene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this synthesis are fully realized in practice. We are committed to delivering high-purity Pterostilbene that meets stringent purity specifications, utilizing our rigorous QC labs to monitor every step of the condensation, reduction, and hydrolysis processes. Our infrastructure is designed to handle the specific thermal and safety requirements of diazotization reactions, guaranteeing a safe and consistent supply for your downstream applications.
We invite you to collaborate with us to optimize your supply chain for this high-value intermediate. Whether you are looking to reduce costs in pharmaceutical intermediates manufacturing or secure a long-term source of high-purity Pterostilbene, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis, where we can evaluate your current sourcing strategy against our optimized production capabilities. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis method can add tangible value to your product portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
