Scalable Copper-Catalyzed Decarboxylation for High-Purity E-Diarylethene Intermediates
The pharmaceutical and nutraceutical industries have long recognized the potent biological activities of E-diarylethene derivatives, particularly compounds like Pterostilbene and Trans-Resveratrol, which exhibit significant antioxidant, anti-inflammatory, and metabolic regulatory properties. However, the commercial realization of these high-value active pharmaceutical ingredients (APIs) has historically been bottlenecked by inefficient synthetic routes that rely on complex protection strategies and difficult purification processes. Patent CN101343214B introduces a transformative preparation method that addresses these critical manufacturing challenges by utilizing a direct decarboxylation and isomerization strategy. This innovative approach leverages inexpensive copper powder as a catalyst within a nitrogenous basic solvent system, enabling the direct conversion of E-2,3-diaryl acrylic acid derivatives into the target E-diarylethenes. By operating at elevated temperatures between 190°C and 230°C, this process achieves high yields ranging from 41% to 85% while fundamentally simplifying the operational workflow. For R&D directors and process chemists, this patent represents a pivotal shift away from multi-step sequences toward a streamlined, atom-economical pathway that enhances both purity profiles and production scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the stilbene backbone, such as the Wittig reaction, have plagued process development teams with inherent inefficiencies and scalability hurdles. As illustrated in the reaction schemes below, classical methods often necessitate the use of sensitive reagents and extensive protecting group manipulations that drastically increase the step count and overall cost of goods sold (COGS). For instance, the Wittig reaction typically requires the formation of phosphonium salts and the use of strong bases, often generating stoichiometric amounts of triphenylphosphine oxide waste that is difficult to remove. Furthermore, these reactions frequently produce mixtures of Z and E isomers, necessitating energy-intensive chromatographic separations or recrystallizations to isolate the biologically active E-isomer. The requirement to protect phenolic hydroxyl groups with bulky silyl ethers like TBDMS adds further complexity, introducing additional reagents, reaction steps, and potential impurities that must be rigorously controlled to meet regulatory standards for pharmaceutical intermediates.
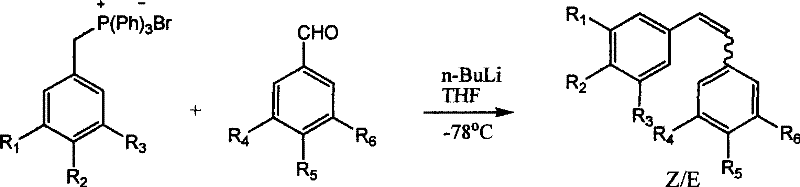
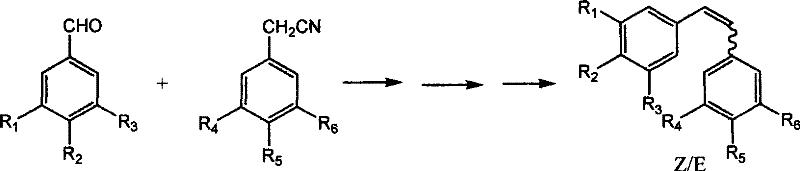
Similarly, alternative approaches like aldol condensation or Heck coupling present their own distinct sets of operational drawbacks that hinder large-scale adoption. Aldol condensation routes, while conceptually straightforward, often suffer from poor stereocontrol, yielding substantial quantities of the undesired Z-isomer alongside the target product, which complicates downstream purification. The Heck reaction, although powerful, relies on expensive palladium catalysts and aryl halides, which are not only cost-prohibitive for bulk manufacturing but also introduce risks of heavy metal contamination that require stringent clearance protocols. The cumulative effect of these limitations is a manufacturing process that is fragile, expensive, and environmentally burdensome, creating significant supply chain vulnerabilities for companies relying on these critical bioactive intermediates.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN101343214B offers a robust and elegant solution by exploiting the thermal decarboxylation of cinnamic acid derivatives. This novel approach bypasses the need for external coupling reagents or precious metal catalysts, utilizing instead a heterogeneous copper powder system that is both inexpensive and easily recoverable. The reaction mechanism inherently favors the formation of the thermodynamically stable E-isomer during the decarboxylation event, effectively solving the stereochemical issues that plague Wittig and aldol routes. By conducting the reaction in high-boiling nitrogenous solvents such as quinoline or imidazole, the process maintains the necessary thermal energy to drive the reaction to completion while simultaneously acting as a base to facilitate the elimination of carbon dioxide. This integration of reaction and isomerization into a single operational unit significantly reduces the physical footprint of the manufacturing process and minimizes the generation of hazardous waste streams.
Mechanistic Insights into Copper-Catalyzed Decarboxylation and Isomerization
The core of this technological advancement lies in the synergistic interaction between the copper catalyst surface and the nitrogenous solvent medium under high-temperature conditions. The mechanism likely initiates with the coordination of the carboxylic acid substrate to the copper surface, facilitating the electron transfer required for the decarboxylation step. As the carbon dioxide molecule is expelled, a transient vinyl-copper intermediate or a radical species is formed, which is highly susceptible to isomerization. The high thermal energy provided by heating the reaction mixture to 190°C–230°C ensures that the system has sufficient activation energy to overcome the rotational barrier of the double bond, allowing it to relax into the lower-energy E-configuration. The nitrogenous base, whether it be quinoline, imidazole, or methylimidazole, plays a dual role: it acts as a solvent to dissolve the polar intermediates and as a proton shuttle to assist in the final protonation step that yields the neutral E-diarylethene product. This mechanistic pathway is remarkably clean, as the only byproduct is gaseous CO2, which simply vents from the reactor, leaving behind a reaction mixture that is predominantly the desired product and the recoverable catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over competing technologies. Because the reaction does not involve the formation of phosphine oxides or palladium complexes, the impurity profile is significantly simplified, consisting mainly of unreacted starting material or minor thermal degradation products that are easily removed. The absence of protecting groups means there are no residual silyl ethers or benzyl fragments to track and eliminate, which streamlines the analytical validation process. Furthermore, the heterogeneous nature of the copper catalyst allows for its physical removal via simple filtration prior to any aqueous workup, preventing metal leaching into the product stream. This inherent purity advantage reduces the burden on downstream purification units, such as crystallization or distillation columns, and ensures that the final API intermediate meets the stringent quality specifications required by global regulatory bodies without the need for extensive chromatographic polishing.
How to Synthesize E-Diarylethene Derivatives Efficiently
The implementation of this synthesis protocol is designed for operational simplicity, making it highly accessible for pilot plant and commercial scale-up operations. The process begins by charging a reactor with the specific E-2,3-diaryl acrylic acid precursor, copper powder, and the chosen nitrogenous solvent in precise mass ratios optimized for maximum conversion. The detailed standardized synthesis steps, including specific stirring rates, heating ramps, and quenching procedures, are outlined in the guide below to ensure reproducibility and safety during execution.
- Combine E-2,3-diaryl acrylic acid derivative, copper powder catalyst, and a nitrogenous basic solvent such as quinoline or imidazole in a reaction vessel.
- Heat the mixture to 190-230°C under a nitrogen atmosphere for 2 to 12 hours to facilitate simultaneous decarboxylation and isomerization.
- Filter off the copper catalyst, wash the filtrate with acid to remove the solvent, and purify the crude product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed decarboxylation route translates directly into enhanced supply security and substantial cost optimization. By eliminating the reliance on volatile and expensive reagents such as palladium catalysts or specialized phosphonium salts, the raw material cost structure is significantly flattened. The ability to recycle the copper catalyst and the nitrogenous solvent further amplifies these savings, creating a circular process flow that minimizes waste disposal costs and reduces the overall consumption of chemicals per kilogram of product. This efficiency gain is critical in a market where margin compression is a constant pressure, allowing manufacturers to maintain competitive pricing while adhering to strict quality standards. Moreover, the simplification of the workflow reduces the dependency on highly skilled labor for complex multi-step operations, thereby lowering operational expenditures associated with training and error mitigation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in step count and the elimination of costly protecting group chemistry. Traditional routes often require three or more discrete steps involving protection, coupling, and deprotection, each incurring yield losses and material costs; in contrast, this one-pot decarboxylation consolidates these transformations into a single unit operation. The removal of expensive transition metals like palladium and the substitution with commodity-grade copper powder results in a direct decrease in catalyst expenditure. Additionally, the recovery and reuse of the high-boiling nitrogenous solvents mean that solvent purchase volumes are drastically reduced over the lifecycle of the campaign, contributing to a leaner and more cost-effective manufacturing model that maximizes return on investment.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials, specifically substituted cinnamic acid derivatives, which are commercially accessible from multiple global vendors. Unlike specialized reagents that may have long lead times or single-source dependencies, the inputs for this process are commodity chemicals with robust supply networks. The robustness of the reaction conditions, which tolerate a range of temperatures and times without catastrophic failure, further ensures consistent batch-to-batch output, reducing the risk of production delays due to process upsets. This reliability allows supply chain planners to forecast inventory levels with greater confidence and respond more agilely to fluctuations in market demand for high-value nutraceutical and pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this technology aligns perfectly with modern green chemistry principles and regulatory expectations. The primary byproduct of the reaction is carbon dioxide, a gas that does not require complex aqueous treatment or hazardous waste disposal protocols, thereby simplifying environmental compliance reporting. The heterogeneous catalyst system facilitates easy separation without the need for extensive extraction or filtration aids, reducing the volume of wastewater generated. As the process scales from kilograms to metric tons, the heat transfer and mixing requirements remain manageable due to the homogeneous liquid phase of the reactants, ensuring that the high yields observed in the laboratory can be faithfully reproduced in large-scale industrial reactors without compromising safety or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: Does this method require hydroxyl protection groups?
A: No, a key advantage of this copper-catalyzed decarboxylation is that it proceeds directly on substrates containing free phenolic hydroxyl or acetoxy groups, eliminating the need for tedious protection and deprotection steps common in Wittig or Heck reactions.
Q: How is the E/Z isomer ratio controlled?
A: The reaction conditions promote thermodynamic control, driving the isomerization towards the more stable E-isomer during the decarboxylation process, thereby avoiding the difficult separation of Z/E mixtures often encountered in aldol condensations.
Q: Can the copper catalyst be recycled?
A: Yes, the heterogeneous copper powder catalyst can be easily recovered by simple filtration after the reaction, washed with organic solvents like ethyl acetate, and reused, contributing to significant cost reduction and waste minimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable E-Diarylethene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN101343214B can be realized in your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch, guaranteeing that our E-diarylethene intermediates meet the exacting standards required for downstream API synthesis. Our commitment to quality assurance extends beyond mere compliance; we actively collaborate with clients to optimize process parameters for their specific facility constraints, ensuring a seamless technology transfer.
We invite you to engage with our technical procurement team to discuss how this advanced decarboxylation technology can be integrated into your existing manufacturing portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this more efficient route. We encourage you to contact us today to obtain specific COA data for our current inventory and to receive comprehensive route feasibility assessments tailored to your project timelines. Let us help you secure a sustainable and cost-effective supply of these critical high-value intermediates.
