Scalable Metal-Free Formylation Technology for High-Purity Pharmaceutical Intermediates Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, sustainable, and cost-effective methodologies for constructing essential molecular scaffolds, particularly formamide derivatives which serve as critical building blocks in bioactive molecules and functional materials. Patent CN107827816B introduces a groundbreaking approach to synthesizing these valuable compounds through a graphene oxide catalyzed formylation reaction that fundamentally shifts the paradigm away from traditional metal-dependent catalysis. This innovation leverages the unique surface chemistry of oxidized graphene to activate amine substrates and formyl sources simultaneously, enabling a one-pot transformation that operates under relatively mild thermal conditions while delivering exceptional selectivity. The technology addresses long-standing challenges in organic synthesis by providing a metal-free alternative that circumvents the regulatory and environmental burdens associated with heavy metal residues in active pharmaceutical ingredients. By utilizing inexpensive and industrially available reagents such as DMF as the carbonyl source, this method offers a streamlined pathway that enhances both the economic viability and the environmental profile of manufacturing processes for reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of formamide derivatives has relied heavily on a diverse array of carbonyl sources and catalytic systems, each carrying significant drawbacks that hinder efficient large-scale production. Traditional methods utilizing formic acid or its salts often suffer from high reagent costs, strong reducibility issues, and stringent storage requirements due to toxicity concerns, making them less ideal for sensitive manufacturing environments. Alternative approaches employing esters as carbonyl sources necessitate the use of nickel catalysis alongside expensive additives like imidazolium salts and aluminum trialkoxides, which drastically inflate the overall production cost and complicate downstream purification processes. Furthermore, methods based on methanol require precious ruthenium catalysts and strong bases such as potassium tert-butoxide, introducing safety hazards and increasing the financial barrier for entry. Even when using DMF, a common industrial solvent, prior art methods reported by researchers like Jagtap and Sheppard have relied on toxic boron derivatives or expensive boronic acid catalysts that are difficult to separate from the final product, leading to potential contamination and increased waste treatment expenses. These conventional pathways often exhibit limited substrate scope, particularly struggling with secondary amines, and frequently result in lower yields accompanied by complex byproduct profiles that demand resource-intensive purification steps.
The Novel Approach
In stark contrast to these established limitations, the novel methodology disclosed in the patent utilizes graphene oxide as a heterogeneous, metal-free catalyst that effectively activates both the amine substrate and the DMF acylating agent through hydrogen bonding interactions. This innovative catalytic system operates efficiently across a broad spectrum of substrates, including both primary and secondary amines, achieving high conversion rates and isolated yields that frequently exceed 90% for optimized conditions. The use of graphene oxide eliminates the need for toxic heavy metals or expensive organoboron reagents, thereby simplifying the regulatory compliance landscape for pharmaceutical manufacturers aiming for high-purity outputs. The reaction proceeds in a simple one-pot fashion where the catalyst can be easily recovered via filtration after the reaction is complete, allowing for direct reuse in subsequent batches without significant loss of catalytic activity. This approach not only reduces the consumption of raw materials but also minimizes the generation of hazardous waste, aligning perfectly with modern green chemistry principles and the demands of a cost reduction in API manufacturing strategy. The versatility of this system is further evidenced by its compatibility with various formyl sources, although DMF remains the most economically advantageous choice due to its widespread availability and low cost in the global chemical market.

Mechanistic Insights into Graphene Oxide Catalyzed Formylation
The catalytic mechanism underpinning this transformative synthesis relies on the multifunctional oxygen-containing groups present on the graphene oxide surface, which act as dual activators for the reactants involved in the formylation process. Initially, the graphene oxide catalyst engages with both the substituted amine and the DMF molecule through a network of hydrogen bonds, forming a highly organized active species that lowers the activation energy required for the nucleophilic attack. In this activated complex, the amine nitrogen acts as a potent nucleophile that attacks the carbonyl carbon of the DMF, facilitated by the proton-shuttling capability of the catalyst's hydroxyl and carboxyl groups. This interaction leads to the formation of a key tetrahedral intermediate, which subsequently undergoes a proton transfer sequence that regenerates the graphene oxide catalyst while releasing dimethylamine as a volatile byproduct. The precise arrangement of functional groups on the graphene lattice ensures that the reaction proceeds with high regioselectivity and minimal side reactions, effectively suppressing the formation of unwanted impurities that often plague metal-catalyzed processes. This mechanistic pathway highlights the unique ability of carbon-based nanomaterials to mimic enzymatic active sites, providing a biomimetic environment that enhances reaction efficiency without the introduction of foreign metallic elements into the reaction matrix.
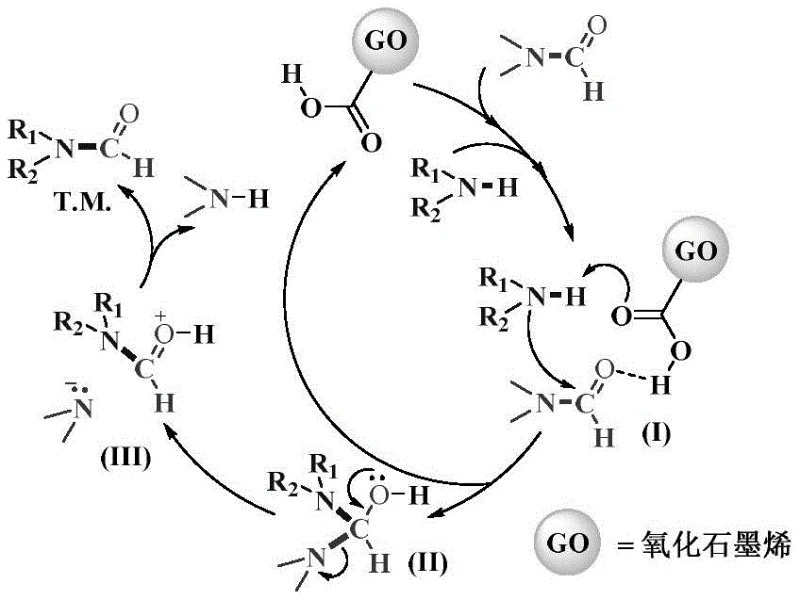
From an impurity control perspective, the heterogeneous nature of the graphene oxide catalyst plays a pivotal role in ensuring the purity of the final formamide derivatives. Unlike homogeneous catalysts that dissolve completely in the reaction medium and require complex extraction or chromatography to remove trace metal residues, the solid graphene oxide particles remain suspended or settled, allowing for physical separation via simple filtration techniques. This physical separation capability ensures that the final product is free from catalyst-derived contaminants, which is a critical quality attribute for pharmaceutical intermediates intended for human consumption. Furthermore, the mild reaction conditions employed, typically ranging between 130°C and 160°C, prevent the thermal degradation of sensitive functional groups on the amine substrates, thereby preserving the structural integrity of complex molecules. The absence of strong acids or bases in the catalytic cycle further reduces the risk of hydrolysis or racemization, which are common pitfalls in traditional formylation protocols. By maintaining a clean reaction profile with minimal byproduct formation, this technology significantly reduces the burden on downstream purification units, leading to higher overall process yields and reduced solvent consumption during the workup phase.
How to Synthesize Formamide Derivatives Efficiently
The practical implementation of this graphene oxide catalyzed formylation involves a straightforward protocol that begins with the precise weighing of the amine substrate and the graphene oxide catalyst, typically used at a loading of 15 to 40 weight percent relative to the amine. These components are dissolved or suspended in DMF, which serves as both the solvent and the formyl source, creating a homogeneous or semi-homogeneous reaction mixture that is then subjected to heating under an inert atmosphere to prevent oxidation side reactions. The reaction is maintained at an elevated temperature, optimally around 150°C, for a duration of 6 to 24 hours depending on the specific steric and electronic properties of the amine substrate being processed. Upon completion, the mixture is cooled to room temperature, and the solid catalyst is removed by filtration, leaving a filtrate that contains the crude formamide product along with excess DMF and dimethylamine byproducts. Detailed standardized synthesis steps see the guide below.
- Mix the amine compound and graphene oxide catalyst (15-40 wt% relative to amine) in DMF solvent under inert gas protection.
- Heat the reaction mixture to 130-160°C (optimally 150°C) and stir for 6-24 hours to complete the formylation.
- Cool the reaction, filter to recover the graphene oxide catalyst, and purify the filtrate via extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this graphene oxide catalyzed technology presents a compelling value proposition driven by substantial reductions in raw material costs and operational complexity. The elimination of expensive noble metal catalysts such as ruthenium or nickel, which are subject to volatile market pricing and supply constraints, translates directly into a more stable and predictable cost structure for long-term production contracts. Additionally, the replacement of toxic boron-based reagents with benign carbon materials removes the need for specialized hazardous waste disposal services and reduces the regulatory compliance overhead associated with handling dangerous chemicals. The ability to recover and recycle the graphene oxide catalyst for multiple cycles further amplifies these cost savings by extending the useful life of the catalytic material and minimizing the frequency of catalyst replenishment orders. This operational efficiency is complemented by the use of DMF, a commodity chemical with a robust global supply chain, ensuring that raw material availability remains consistent even during periods of market fluctuation.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost dynamics of formamide synthesis by removing the most expensive line items from the bill of materials. By avoiding the procurement of high-purity noble metals and specialized ligands, manufacturers can achieve significant cost optimization without compromising on reaction performance or product quality. The simplified workup procedure, which relies on filtration rather than complex extraction or scavenging resins to remove metal traces, further reduces labor costs and solvent usage, contributing to a leaner and more profitable manufacturing operation. These cumulative savings allow companies to offer more competitive pricing to their clients while maintaining healthy profit margins in a highly competitive marketplace.
- Enhanced Supply Chain Reliability: Dependence on scarce or geographically concentrated resources poses a significant risk to supply chain continuity, a vulnerability that this new technology effectively mitigates by utilizing abundant carbon-based catalysts. Graphene oxide can be produced from graphite, a widely available natural resource, ensuring that the catalyst supply is not bottlenecked by the same geopolitical or mining constraints that affect rare earth metals. Furthermore, the robustness of the catalyst allows for extended storage and transport without degradation, simplifying inventory management and reducing the risk of production delays due to expired or compromised reagents. This reliability is crucial for maintaining just-in-time delivery schedules and meeting the rigorous demand forecasts of large-scale pharmaceutical partners.
- Scalability and Environmental Compliance: The scalability of this process is demonstrated by successful gram-scale experiments that maintain high yields, indicating a clear path towards ton-scale commercial production without the need for extensive process re-engineering. The heterogeneous nature of the catalyst facilitates easy integration into existing reactor infrastructure, whether batch or continuous flow, minimizing capital expenditure requirements for technology adoption. From an environmental standpoint, the reduction in heavy metal waste and toxic reagents aligns with increasingly stringent global environmental regulations, reducing the risk of fines and enhancing the corporate sustainability profile. This alignment with green chemistry principles not only satisfies regulatory bodies but also appeals to environmentally conscious stakeholders and customers who prioritize sustainable sourcing in their supply chains.
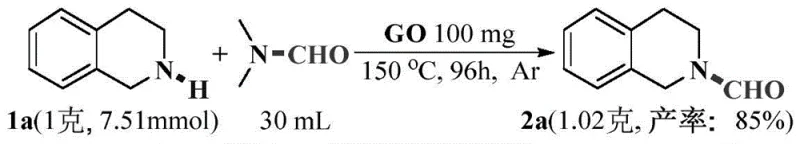
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this graphene oxide catalyzed formylation technology, providing clarity on its operational parameters and strategic benefits. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this synthesis route. Understanding these details is essential for assessing the feasibility of integrating this method into existing manufacturing workflows and for projecting the long-term value it can deliver to the organization.
Q: What are the advantages of using Graphene Oxide over traditional catalysts for formylation?
A: Graphene Oxide eliminates the need for expensive noble metals like Ruthenium or Nickel and avoids toxic boron reagents. It is cheap, non-toxic, easily separable by filtration, and can be recycled for multiple cycles without significant loss of activity.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent demonstrates successful scale-up reactions (e.g., 1g scale yielding 85% product). The simple one-pot procedure, use of industrial solvent DMF, and easy catalyst recovery make it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: Can the graphene oxide catalyst be reused?
A: Experimental data confirms that the graphene oxide catalyst maintains high catalytic efficiency for at least 5 consecutive recycling cycles, significantly reducing waste and operational costs compared to single-use homogeneous catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formamide Derivatives Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM stands ready to leverage this advanced graphene oxide catalyzed technology to deliver high-quality formamide derivatives that meet the exacting standards of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of material we supply is free from the metal contaminants that often plague traditionally synthesized intermediates. Our investment in state-of-the-art processing equipment allows us to fully capitalize on the scalability and efficiency of this metal-free method, providing our partners with a secure and sustainable source of critical building blocks.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs, offering a pathway to Customized Cost-Saving Analysis that aligns with your budgetary goals. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this greener, more economical manufacturing process. Let us help you optimize your supply chain and accelerate your development timelines with our superior technical capabilities and unwavering commitment to quality excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
