Scalable Catalyst-Free Transamination for High-Purity Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for greener, more cost-effective synthetic routes. A pivotal advancement in this domain is documented in patent CN109293569B, which discloses a novel method for preparing formamide derivatives via a transamination reaction that remarkably proceeds without any catalyst or additional solvent participation. Formamide derivatives are ubiquitous structural motifs found in a vast array of biologically active molecules, ranging from bronchodilators like Formoterol to complex antibiotics and functional polymer materials, as illustrated in the diverse applications shown below. 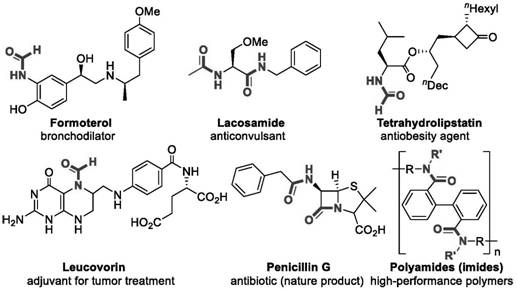 . The traditional reliance on toxic formylating agents and expensive metal catalysts has long been a bottleneck for R&D teams aiming for high purity and supply chain heads seeking cost efficiency. This patent introduces a paradigm shift by utilizing N,N-dimethylformamide (DMF) directly as the formyl source, achieving high yields through a streamlined, one-step thermal process that fundamentally alters the economic and environmental profile of producing these critical fine chemical intermediates.
. The traditional reliance on toxic formylating agents and expensive metal catalysts has long been a bottleneck for R&D teams aiming for high purity and supply chain heads seeking cost efficiency. This patent introduces a paradigm shift by utilizing N,N-dimethylformamide (DMF) directly as the formyl source, achieving high yields through a streamlined, one-step thermal process that fundamentally alters the economic and environmental profile of producing these critical fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of formamide derivatives has been plagued by significant technical and economic hurdles that impact both the purity of the final API intermediate and the overall cost of goods sold. Conventional methodologies frequently necessitate the use of hazardous formylating reagents such as formyl chloride or mixed anhydrides, which pose severe safety risks and require specialized handling infrastructure. Furthermore, many established protocols rely heavily on transition metal catalysts including palladium, ruthenium, gold, or copper complexes to drive the formylation reaction. While effective in a laboratory setting, these metal-catalyzed routes introduce a critical downstream burden: the rigorous removal of trace metal residues to meet stringent pharmaceutical purity specifications. This purification step often involves expensive scavengers or complex chromatographic separations, drastically inflating production costs and extending lead times. Additionally, the use of organic solvents in these traditional methods contributes to a high E-factor, generating substantial chemical waste that complicates environmental compliance and disposal logistics for large-scale manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN109293569B offers a radically simplified pathway that eliminates the need for external catalysts and auxiliary solvents. As depicted in the general reaction scheme below, the process relies on the direct transamination between a low-reactivity tertiary amide (specifically DMF) and a脂肪胺 (aliphatic amine). 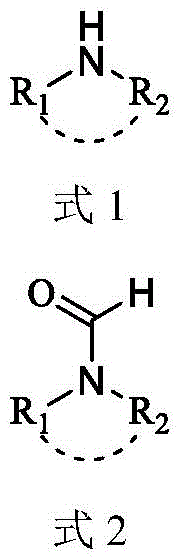 . By heating the reactants to temperatures between 130°C and 150°C under an inert atmosphere, the system achieves sufficient activation energy to drive the equilibrium towards the desired formamide product without metallic assistance. This solvent-free approach not only reduces the volume of reaction mass, thereby increasing reactor throughput, but also simplifies the isolation procedure significantly. The absence of metal catalysts means the resulting crude product is free from heavy metal contamination, a decisive advantage for reliable pharmaceutical intermediate suppliers who must guarantee compliance with ICH Q3D guidelines for elemental impurities. This method transforms a multi-step, waste-intensive process into a concise, atom-economical operation.
. By heating the reactants to temperatures between 130°C and 150°C under an inert atmosphere, the system achieves sufficient activation energy to drive the equilibrium towards the desired formamide product without metallic assistance. This solvent-free approach not only reduces the volume of reaction mass, thereby increasing reactor throughput, but also simplifies the isolation procedure significantly. The absence of metal catalysts means the resulting crude product is free from heavy metal contamination, a decisive advantage for reliable pharmaceutical intermediate suppliers who must guarantee compliance with ICH Q3D guidelines for elemental impurities. This method transforms a multi-step, waste-intensive process into a concise, atom-economical operation.
Mechanistic Insights into Catalyst-Free Transamination
Understanding the mechanistic underpinnings of this catalyst-free transformation is crucial for R&D directors evaluating its robustness and scope. The reaction proceeds through a unique activation mode where the amine substrate itself plays a dual role. Initially, the hydrogen atom on the nitrogen of the aliphatic amine forms a hydrogen bond with the carbonyl oxygen of the DMF molecule. This intermolecular interaction serves to activate the carbonyl carbon, making it more susceptible to nucleophilic attack. Subsequently, the lone pair of electrons on the nitrogen of a second amine molecule (or the same species in a dynamic equilibrium) performs a nucleophilic attack on the activated carbonyl carbon of the DMF. This leads to the formation of a tetrahedral intermediate, which then undergoes fragmentation to release dimethylamine and generate the N-formyl product. The cycle continues as the released dimethylamine can theoretically participate in further equilibria, although the high boiling point and excess of DMF drive the reaction forward. 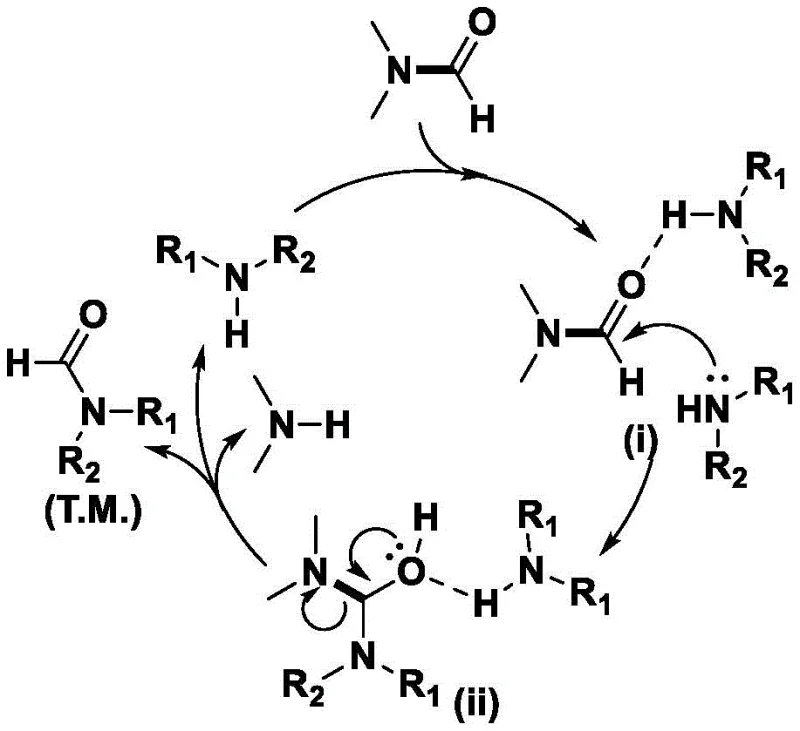 . This mechanism highlights the elegance of the process: it leverages the inherent basicity and nucleophilicity of the amine substrate to catalyze its own transformation, removing the dependency on external Lewis or Brønsted acids.
. This mechanism highlights the elegance of the process: it leverages the inherent basicity and nucleophilicity of the amine substrate to catalyze its own transformation, removing the dependency on external Lewis or Brønsted acids.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Since no transition metals are introduced, there is zero risk of metal-induced side reactions such as oxidative coupling or hydrogenolysis, which often plague sensitive substrates containing halogens or other reducible groups. The primary by-product is dimethylamine, which is volatile and easily removed during the concentration or workup phase, leaving behind a clean product profile. The high selectivity observed across various substrates, including those with electron-withdrawing groups like bromine or trifluoromethyl groups, suggests that the transition state is not significantly destabilized by steric or electronic variations. This broad tolerance ensures that the process can be applied to a wide library of complex amine building blocks without requiring extensive re-optimization of reaction parameters, providing a reliable platform for the synthesis of high-purity pharmaceutical intermediates.
How to Synthesize N-Formyl-1,2,3,4-tetrahydroisoquinoline Efficiently
To implement this technology for the production of key intermediates like N-formyl-1,2,3,4-tetrahydroisoquinoline, operators must adhere to specific thermal and atmospheric conditions to maximize yield and minimize side reactions. The patent data indicates that while the reaction can proceed at temperatures as low as 130°C, optimal conversion is achieved at 150°C over a period of 24 hours. The specific reaction setup for a model substrate is shown below, demonstrating the simplicity of charging the amine and DMF directly into a sealed vessel.  . Maintaining an inert atmosphere, preferably argon, is critical to prevent oxidative degradation of the amine starting material at these elevated temperatures. Following the thermal cycle, the workup involves a straightforward aqueous quench and extraction with ethyl acetate, avoiding the need for complex distillation or crystallization steps often required to remove metal catalysts. The detailed standardized synthesis steps for scaling this process are outlined in the guide below.
. Maintaining an inert atmosphere, preferably argon, is critical to prevent oxidative degradation of the amine starting material at these elevated temperatures. Following the thermal cycle, the workup involves a straightforward aqueous quench and extraction with ethyl acetate, avoiding the need for complex distillation or crystallization steps often required to remove metal catalysts. The detailed standardized synthesis steps for scaling this process are outlined in the guide below.
- Charge the reaction vessel with the target aliphatic amine (e.g., 1,2,3,4-tetrahydroisoquinoline) and a magnetic stir bar under inert atmosphere protection.
- Add N,N-dimethylformamide (DMF) directly to the vessel, serving as both the formyl source and the reaction medium, typically at a molar ratio of 1: 2.5.
- Heat the mixture to 150°C in an oil bath and stir for 24 hours. Upon completion, cool, dilute with water, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free transamination technology translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of expensive noble metal catalysts represents a direct reduction in raw material costs, as there is no longer a need to purchase, recover, or dispose of precious metals like palladium or ruthenium. Furthermore, the solvent-free nature of the reaction significantly reduces the volume of hazardous waste generated per kilogram of product, lowering disposal fees and simplifying environmental reporting. The simplified downstream processing—requiring only basic extraction and drying rather than specialized metal scavenging columns—reduces the consumption of silica gel and eluents, further driving down the cost of goods. These factors combine to create a more resilient supply chain that is less vulnerable to fluctuations in the price of rare earth metals or specialty reagents.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of catalyst costs and the associated purification expenses. In traditional routes, the cost of the catalyst and the materials needed to reduce metal residues to ppm levels can constitute a significant portion of the total manufacturing cost. By utilizing DMF as both reagent and solvent, the process achieves substantial cost savings through material consolidation. Additionally, the high atom economy of the transamination reaction ensures that the majority of the input mass is converted into valuable product or easily removable volatile by-products, minimizing waste treatment costs and maximizing the efficiency of raw material utilization in commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like DMF and simple aliphatic amines drastically improves supply security compared to routes dependent on specialized, single-source catalysts. DMF is a globally available bulk chemical with a stable supply chain, reducing the risk of production stoppages due to reagent shortages. The robustness of the reaction conditions (130-150°C) also means that the process is less sensitive to minor variations in reagent quality, allowing for greater flexibility in sourcing raw materials. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturing schedules are met without delay caused by complex purification bottlenecks or catalyst procurement issues.
- Scalability and Environmental Compliance: The absence of solvent and catalyst simplifies the engineering requirements for scale-up. Reactors do not need to be lined for corrosion resistance against strong acids or equipped for high-pressure hydrogenation, making the technology adaptable to existing multipurpose facilities. The reduced waste stream aligns perfectly with modern green chemistry initiatives and increasingly strict environmental regulations. By minimizing the use of volatile organic compounds (VOCs) and eliminating heavy metal waste, manufacturers can achieve better environmental compliance scores and lower their carbon footprint. This sustainability advantage is becoming a key differentiator in vendor selection processes for major multinational pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst-free transamination technology. These answers are derived directly from the experimental data and scope defined in patent CN109293569B, providing clarity on substrate compatibility, reaction conditions, and purification strategies. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this method require expensive transition metal catalysts?
A: No, the patented process (CN109293569B) operates entirely without catalysts. It utilizes the intrinsic reactivity of DMF at elevated temperatures (130-150°C), eliminating the need for costly palladium, ruthenium, or copper catalysts and the subsequent removal steps.
Q: What is the typical yield for N-formyl-1,2,3,4-tetrahydroisoquinoline?
A: Under optimized conditions (150°C, 24 hours, argon atmosphere), the process achieves yields up to 81% for 1,2,3,4-tetrahydroisoquinoline, with specific substrates like 3-phenyl-1-propylamine reaching yields as high as 99%.
Q: Can this method be scaled for industrial production?
A: Yes, the solvent-free nature and lack of sensitive catalysts make this highly scalable. The simple workup procedure involving aqueous dilution and extraction is compatible with standard large-scale manufacturing equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Formyl-1,2,3,4-tetrahydroisoquinoline Supplier
The technological potential of catalyst-free transamination represents a significant leap forward for the efficient production of formamide derivatives, and NINGBO INNO PHARMCHEM is uniquely positioned to leverage this innovation for your projects. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, particularly for elemental impurities, guaranteeing that every batch of N-Formyl-1,2,3,4-tetrahydroisoquinoline meets the highest global standards. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our commitment to process excellence ensures reliable delivery of critical intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our formamide derivative portfolio and to discuss route feasibility assessments for your custom synthesis needs. Together, we can engineer a more efficient and sustainable future for your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
