Industrial Scale-Up of Memantine Hydrochloride via Optimized Ritter Reaction and Green Solvent Systems
Introduction to Advanced Memantine Hydrochloride Manufacturing
The global demand for effective treatments for Alzheimer's disease continues to drive the need for reliable, high-quality active pharmaceutical ingredients (APIs) and their precursors. Among these, Memantine Hydrochloride stands out as a critical NMDA receptor antagonist, necessitating manufacturing processes that balance efficiency with stringent safety standards. The technical methodology detailed in patent CN103553932A represents a significant evolution in the synthetic landscape of this compound, addressing historical bottlenecks related to thermal runaway and toxic solvent usage. By refining the classic Ritter reaction pathway, this approach offers a viable solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring that production facilities can meet rigorous regulatory requirements while maintaining operational safety. This report analyzes the technical merits of this optimized route, providing strategic insights for R&D directors and supply chain leaders seeking to enhance their manufacturing capabilities for neurotherapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Memantine Hydrochloride has been plagued by methodologies that pose severe risks during industrial amplification. Traditional approaches, such as the urea method, require excessively high reaction temperatures and harsh conditions that degrade equipment and compromise product purity. Alternative routes involving Grignard reagents demand strictly anhydrous and oxygen-free environments, often necessitating expensive ultrasonic equipment and specialized operational expertise that increases overhead costs. Furthermore, earlier iterations of the acetonitrile-based Ritter reaction, while chemically sound, suffered from uncontrollable exothermic profiles; experimental data indicates that without precise intervention, reaction temperatures can skyrocket to 160°C, leading to dangerous material ejection. Additionally, legacy processes heavily relied on carcinogenic solvents like benzene and chloroform for extraction and recrystallization, creating substantial liabilities regarding worker safety and environmental compliance that modern facilities can no longer tolerate.
The Novel Approach
The optimized protocol introduced in the referenced patent fundamentally restructures the synthesis to prioritize thermal management and green chemistry principles. By implementing a strict temperature control regime during the initial acetylamidation—specifically maintaining the internal temperature below 20°C during acid addition and monitoring the system's color transition—the process effectively mitigates the risk of violent exothermic events. This methodological shift transforms a hazardous laboratory reaction into a robust industrial operation. Moreover, the substitution of toxic extraction and recrystallization solvents with safer alternatives like ethyl acetate, dichloromethane, ethanol, and isopropanol drastically simplifies waste treatment protocols. This holistic improvement not only enhances the safety profile but also streamlines the downstream processing, making it an ideal candidate for cost reduction in API manufacturing where solvent recovery and disposal constitute a major portion of operational expenditure.
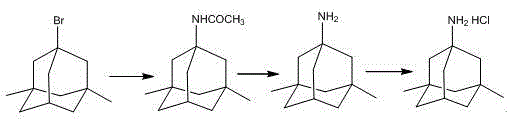
Mechanistic Insights into Controlled Ritter Reaction and Hydrolysis
The core of this synthetic strategy relies on a meticulously managed Ritter reaction, where 1-bromo-3,5-dimethyladamantane reacts with acetonitrile in the presence of concentrated sulfuric acid. Mechanistically, the strong acid facilitates the ionization of the carbon-bromine bond to generate a stable tertiary carbocation, which is subsequently trapped by the nitrile nitrogen of acetonitrile. The critical innovation lies in the kinetic control of this step; the observed color change from colorless to light yellow and finally to deep red serves as a visual indicator of the reaction's progression and thermal status. By coupling this visual cue with active cooling (ice bath) when the temperature rises between 35°C and 50°C, the process prevents the accumulation of thermal energy that typically leads to side reactions and decomposition. This level of control ensures that the intermediate, 1-acetylamino-3,5-dimethyladamantane, is formed with high fidelity, minimizing the formation of polymeric byproducts that often complicate purification in less controlled environments.
Following the amidation, the subsequent hydrolysis step utilizes a high-boiling solvent system comprising ethylene glycol and water with sodium hydroxide to cleave the acetamide group. The use of ethylene glycol is particularly advantageous as it allows the reaction mixture to reach the necessary 150-160°C range without requiring high-pressure vessels, thereby facilitating the complete conversion to 1-amino-3,5-dimethyladamantane. The final salt formation with hydrochloric acid is straightforward, but the true mechanistic elegance is found in the recrystallization phase. By employing a mixed solvent system of polar alcohols and esters, the process leverages differential solubility to exclude impurities effectively. This careful manipulation of solubility parameters ensures that the final Memantine Hydrochloride meets stringent purity specifications, a critical factor for any reliable pharmaceutical intermediates supplier aiming to serve regulated markets.
How to Synthesize Memantine Hydrochloride Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the amide intermediate under strictly cooled conditions, followed by a high-temperature hydrolysis in a glycol medium, and concludes with acidification and recrystallization. The detailed standardized synthetic steps, including precise molar ratios and workup procedures, are outlined below to assist technical teams in replicating this efficient pathway.
- Perform Ritter reaction on 1-bromo-3,5-dimethyladamantane with acetonitrile and sulfuric acid, strictly controlling temperature below 20°C initially and monitoring color changes to prevent exothermic runaway.
- Hydrolyze the resulting 1-acetylamino-3,5-dimethyladamantane using sodium hydroxide in an ethylene glycol and water mixture at 150-160°C to yield the free amine.
- Convert the amine to memantine hydrochloride using concentrated hydrochloric acid, followed by recrystallization using a non-toxic mixture of ethanol or isopropanol with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthesis route offers tangible strategic benefits beyond mere chemical efficiency. The elimination of hazardous reagents and the stabilization of reaction conditions directly translate to reduced operational risks and lower insurance premiums for manufacturing sites. By removing the dependency on specialized equipment like ultrasonic reactors or high-pressure hydrogenation units, capital expenditure is significantly minimized, allowing for faster deployment of production lines. Furthermore, the use of commodity chemicals such as acetic acid, ethylene glycol, and common esters ensures a stable supply chain that is less susceptible to the volatility often seen with niche reagents. This robustness is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive consistent supplies without interruption.
- Cost Reduction in Manufacturing: The replacement of toxic solvents like chloroform and benzene with ethyl acetate and alcohols results in substantial cost savings related to waste disposal and environmental compliance. Since these greener solvents are cheaper to procure and easier to recycle, the overall cost of goods sold (COGS) is positively impacted without compromising quality. Additionally, the avoidance of expensive catalysts such as Palladium on Carbon (used in nitration reduction methods) further drives down raw material costs, making the process economically superior to alternative synthetic routes.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production is not bottlenecked by the scarcity of specialized reagents. Unlike methods requiring strictly anhydrous conditions or ultra-pure gases, this protocol tolerates standard industrial-grade materials, simplifying logistics and inventory management. This flexibility allows manufacturers to source materials from multiple vendors, thereby mitigating the risk of supply disruptions and ensuring continuous operation even during market fluctuations.
- Scalability and Environmental Compliance: The inherent safety features of the temperature-controlled Ritter reaction make this process highly scalable from pilot plant to full commercial production. The ability to manage exotherms safely means that reactor sizes can be increased without proportional increases in risk, facilitating the commercial scale-up of complex adamantane derivatives. Moreover, the reduced toxicity of the solvent system aligns with increasingly strict global environmental regulations, future-proofing the manufacturing asset against potential regulatory crackdowns on hazardous chemical usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on critical process parameters and safety considerations for engineering teams evaluating this technology.
Q: Why is strict temperature control critical during the initial acetylamidation step?
A: The Ritter reaction between 1-bromo-3,5-dimethyladamantane and acetonitrile is highly exothermic. Without controlling the internal temperature below 20°C during acid addition and monitoring the subsequent color change (which indicates reaction progress between 35°C and 50°C), the reaction can violently spike to 160°C, causing material ejection and significant safety hazards during scale-up.
Q: How does this method improve environmental safety compared to conventional processes?
A: This protocol eliminates the use of highly toxic solvents such as benzene and chloroform, which were traditionally used for extraction and recrystallization. By substituting these with ethyl acetate, dichloromethane, ethanol, or isopropanol, the process significantly reduces operator health risks and lowers the environmental burden associated with hazardous waste disposal.
Q: What is the expected overall yield for this three-step synthesis?
A: According to the experimental data provided in the patent examples, the optimized process achieves a total yield ranging from 56% to 64% over the three steps. This yield is considered robust for industrial production, especially given the enhanced safety profile and the use of cheaper, greener solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Memantine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Memantine Hydrochloride or related intermediate meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the neurotherapeutic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
