Advanced Manufacturing of Memantine Hydrochloride: A Technical Breakthrough for Global Supply Chains
The global demand for effective treatments against neurodegenerative disorders, particularly Alzheimer's disease, has placed immense pressure on the pharmaceutical supply chain to deliver high-quality active pharmaceutical ingredients (APIs) and their precursors. Among these, Memantine Hydrochloride stands out as a critical NMDA receptor antagonist, widely recognized for its efficacy in treating moderate to severe dementia. However, traditional manufacturing routes have often been plagued by safety hazards, low yields, and complex purification steps that hinder cost-effective mass production. The technical landscape shifted significantly with the disclosure of patent CN103288650A, which introduces a streamlined, two-step synthetic methodology. This innovation utilizes 1-bromo-3,5-dimethyladamantane as a robust starting material, leveraging phosphoric acid catalysis to drive a highly efficient Ritter reaction. For R&D directors and procurement strategists alike, this patent represents a pivotal opportunity to optimize the commercial scale-up of complex pharmaceutical intermediates, ensuring a more reliable and economically viable supply of this essential neurological medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Memantine has been fraught with significant operational challenges that impact both safety and profitability. Early methodologies, such as those disclosed in US3391142, relied heavily on the use of diethyl ether during the recrystallization phase. While chemically effective, diethyl ether possesses an extremely low boiling point and high flammability, creating substantial explosion risks in large-scale reactor environments. Furthermore, alternative routes described in patents like CN1400205 utilized urea-based reactions requiring prolonged heating at temperatures exceeding 200°C. These harsh thermal conditions not only result in excessive energy consumption but also promote the formation of difficult-to-remove byproducts, necessitating tedious purification protocols. Another approach, detailed in CN1556094, attempted a 'one-pot' synthesis but suffered from poor raw material utilization and an overall yield of merely 50%, rendering it commercially unattractive for high-volume manufacturing where margin preservation is critical.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN103288650A offers a refined pathway that prioritizes safety, efficiency, and purity. By selecting 1-bromo-3,5-dimethyladamantane as the initial substrate and reacting it with nitriles under the catalytic influence of phosphoric acid, the invention achieves a remarkably clean conversion to the acetamido intermediate. This reaction proceeds under moderate thermal conditions, typically between 50°C and 120°C, effectively mitigating the thermal hazards associated with previous high-temperature urea routes. The subsequent hydrolysis step utilizes common mineral alkalis in alcohol solvents, avoiding the need for hazardous ether extractions. This strategic shift not only simplifies the operational workflow but also enhances the cost reduction in pharmaceutical intermediates manufacturing by minimizing waste generation and eliminating expensive purification columns. The visual representation of this streamlined transformation highlights the elegance of the new synthetic design.
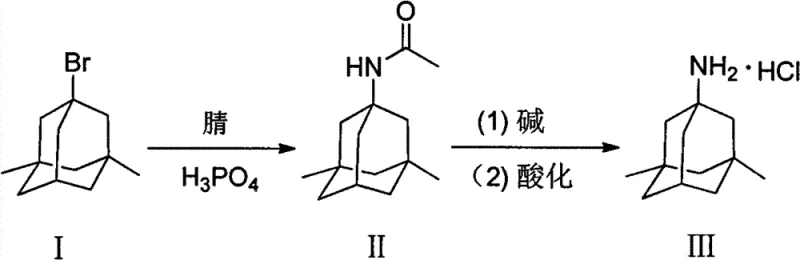
Mechanistic Insights into Phosphoric Acid-Catalyzed Ritter Reaction
The core of this technological advancement lies in the precise manipulation of the Ritter reaction mechanism using phosphoric acid rather than the traditionally employed sulfuric acid. In this catalytic cycle, the phosphoric acid acts as a proton donor to facilitate the ionization of the carbon-bromine bond in the adamantane derivative, generating a stable tertiary carbocation intermediate. This carbocation is then rapidly intercepted by the nitrogen atom of the nitrile solvent, typically acetonitrile, forming a nitrilium ion. The presence of phosphoric acid ensures a controlled acidic environment that promotes this nucleophilic attack while suppressing competing elimination reactions that could lead to olefinic impurities. Subsequent hydration of the nitrilium ion yields the desired acetamido intermediate with exceptional fidelity. This mechanistic control is crucial for maintaining high purity profiles, as it prevents the formation of polymeric tars often seen in harsher acidic conditions, thereby reducing the burden on downstream purification units.
Furthermore, the purification strategy embedded within this process is designed to maximize yield without compromising quality. Unlike methods requiring silica gel chromatography—which are impractical for ton-scale production due to solvent volume and waste issues—this patent leverages differential solubility through crystallization. The intermediate 1-acetamido-3,5-dimethyladamantane can be isolated with high purity simply by adjusting solvent ratios, often using acetone and water mixtures. This physical separation method is inherently more scalable and environmentally benign. The final hydrolysis step, conducted in propanol or similar alcohols with strong bases like KOH or NaOH, cleaves the amide bond efficiently. The resulting free amine is immediately captured as the hydrochloride salt, precipitating out of the solution. This 'react-and-crash' approach ensures that the final high-purity pharmaceutical intermediates are obtained with minimal exposure to atmospheric contaminants, safeguarding the integrity of the supply chain.
How to Synthesize Memantine Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to replicate the high yields reported in the patent literature. The process is divided into two distinct stages: the formation of the acetamido precursor and its subsequent hydrolysis to the final amine salt. Operators must maintain strict temperature controls during the initial Ritter reaction to prevent over-reaction or degradation of the sensitive adamantane cage structure. Following the isolation of the intermediate, the hydrolysis step demands robust agitation and precise pH management to ensure complete conversion before salification. The following guide outlines the standardized operational procedure derived from the patent's preferred embodiments, serving as a foundational protocol for process engineers aiming to establish this production line.
- React 1-bromo-3,5-dimethyladamantane with acetonitrile in the presence of phosphoric acid at 50-120°C to form 1-acetamido-3,5-dimethyladamantane.
- Hydrolyze the intermediate amide using potassium or sodium hydroxide in a C3-C5 alcohol solvent at 80-150°C to release the free amine.
- Salify the resulting amine with hydrochloric acid and purify the final Memantine Hydrochloride through crystallization using ethanol and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates directly into tangible operational improvements and risk mitigation. The shift away from hazardous solvents like ether and the elimination of high-energy consumption steps fundamentally alters the cost structure of Memantine production. By utilizing readily available and inexpensive reagents such as acetonitrile and phosphoric acid, manufacturers can significantly lower their raw material expenditure. Moreover, the simplification of the purification process removes the need for costly chromatographic media and the associated solvent disposal costs, leading to substantial savings in waste management. These factors combined create a more resilient supply chain capable of withstanding market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy of the Ritter reaction and the elimination of expensive purification technologies. By achieving yields approaching quantitative levels in the first step, the process minimizes the loss of valuable starting materials, which are often the most significant cost driver in fine chemical synthesis. Additionally, the avoidance of silica gel chromatography in favor of crystallization drastically reduces solvent consumption and waste treatment costs. This lean manufacturing approach allows for a more competitive pricing structure without sacrificing product quality, making it an attractive option for generic drug manufacturers seeking margin optimization.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized or hazardous reagents that face regulatory shipping restrictions. This synthesis route utilizes commodity chemicals like phosphoric acid, sodium hydroxide, and propanol, which are globally sourced and less susceptible to supply disruptions. The moderate reaction conditions also reduce the wear and tear on reactor equipment, decreasing maintenance downtime and extending the lifecycle of production assets. Consequently, manufacturers can offer more reliable lead times to their clients, ensuring a steady flow of high-purity pharmaceutical intermediates to meet global demand.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental emissions intensify, the ability to produce chemicals with a lower environmental footprint becomes a key competitive advantage. This process generates significantly less three-waste (wastewater, waste gas, and solid waste) compared to traditional methods, thanks to the high selectivity of the reaction and the recyclability of the alcohol solvents. The absence of volatile ether solvents further enhances workplace safety and reduces the risk of environmental contamination. These attributes facilitate easier regulatory approval for new production facilities and support the long-term sustainability goals of modern pharmaceutical enterprises.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its potential integration into their existing manufacturing portfolios. The following questions address common inquiries regarding the practical implementation, yield expectations, and purity standards associated with this patented technology. These insights are derived directly from the experimental data and technical specifications provided in the source documentation, offering a transparent view of the process capabilities.
Q: What are the primary safety advantages of this Memantine synthesis route compared to prior art?
A: Unlike previous methods that utilized diethyl ether for recrystallization—a solvent with a low boiling point and high explosion risk—this patented process employs safer solvents like propanol and ethyl acetate. Furthermore, it avoids the extreme temperatures (200°C) required in urea-based routes, significantly reducing energy consumption and thermal hazard risks.
Q: How does the phosphoric acid catalyst improve the overall yield?
A: The use of phosphoric acid facilitates a highly efficient Ritter reaction with acetonitrile, minimizing side reactions common in sulfuric acid-catalyzed processes. Experimental data indicates yields for the intermediate step can reach up to 99%, drastically reducing raw material waste and improving the economic viability of the production line.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrial scalability. It eliminates the need for complex silica gel chromatography purification, relying instead on robust crystallization techniques. The use of inexpensive, readily available reagents and moderate reaction conditions makes it ideal for multi-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Memantine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN103288650A are fully realized in practice. We operate state-of-the-art facilities equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Memantine Hydrochloride meets the exacting standards required by global regulatory bodies. Our commitment to technical precision ensures that the impurity profiles remain well within acceptable limits, safeguarding the efficacy and safety of the final drug product.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data. Let us help you secure a stable, cost-effective, and high-quality supply of this critical neurological intermediate, driving value for your organization and patients alike.
