Advanced One-Step Synthesis of Bisphosphine Oxides for Commercial Ligand Production
Advanced One-Step Synthesis of Bisphosphine Oxides for Commercial Ligand Production
The landscape of asymmetric catalysis relies heavily on the availability of high-purity chiral ligands, yet their synthesis often presents significant bottlenecks in terms of cost and scalability. Patent CN1224019A introduces a transformative single-step process for the manufacture of bisphosphine oxide compounds and bisphosphonates, which serve as critical intermediates for producing valuable bisphosphine ligands. This methodology replaces the cumbersome traditional multi-step routes with a streamlined oxidative coupling reaction performed at mild temperatures ranging from -70°C to 20°C. By utilizing lithium or magnesium amide compounds followed by the addition of oxidatively-acting metal salts, this innovation offers a robust pathway for generating complex biaryl structures essential for transition metal complexes used in asymmetric hydrogenation. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more efficient and environmentally compliant manufacturing protocols for these high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of titanium dioxide phosphine compounds and bisphosphonates has relied on a laborious two-step approach that poses substantial challenges for industrial scale-up. The conventional route typically begins with the halogenation of a precursor compound at cryogenic temperatures around -70°C using bromine or iodine in the presence of lithium dialkyl amides, a step that often yields aryl halides with only approximately 70% efficiency. The subsequent step involves an Ullmann linking reaction, which necessitates harsh conditions including temperatures between 110°C and 200°C and the use of stoichiometric amounts of copper(0). This high-temperature coupling not only consumes excessive energy but also generates intractable by-product precipitates and significant heavy metal waste, creating severe environmental and disposal issues that complicate regulatory compliance and increase overall production costs for pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent data achieves the formation of the biaryl backbone in a single stage, effectively bypassing the isolation of unstable aryl halide intermediates. This streamlined process involves reacting a phosphine oxide compound with 0.5 to 3 equivalents of a lithium or magnesium amide compound in an organic solvent such as tetrahydrofuran (THF) at controlled temperatures between -70°C and 20°C. Following this lithiation or magnesiation step, an oxidatively-acting metal salt or metal salt complex, such as iron(III) chloride or copper(II) chloride, is added directly to the suspension to induce oxidative dimerization. This method eliminates the need for stoichiometric copper and extreme heat, thereby drastically simplifying the workup procedure and enhancing the safety profile of the manufacturing process while maintaining high purity standards required for downstream catalytic applications.
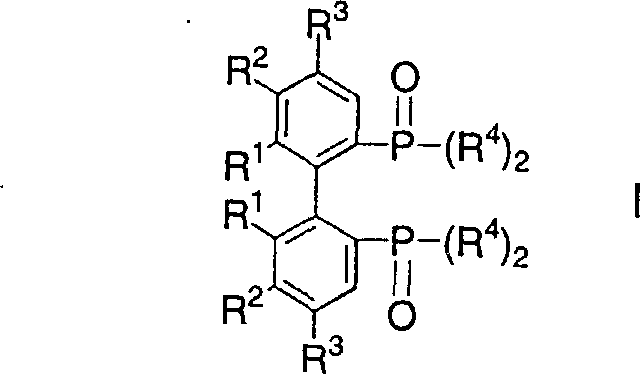
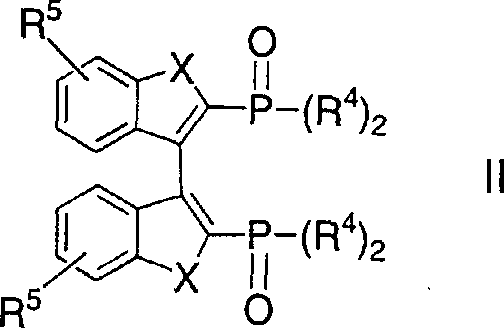
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The core of this technological breakthrough lies in the precise control of the oxidative coupling mechanism mediated by transition metal salts. The reaction initiates with the deprotonation of the phosphine oxide precursor using strong bases like lithium diisopropylamide (LDA) or tetraalkyl piperidyl lithium, generating a reactive nucleophilic species at the ortho-position relative to the phosphorus group. Upon the introduction of the oxidant, typically an Fe(III) salt like FeCl3·xTHF or a Cu(II) salt, a single-electron transfer process facilitates the formation of a radical intermediate which rapidly dimerizes to form the carbon-carbon bond linking the two aromatic systems. This mechanism is highly sensitive to temperature, with the patent specifying a preferred range of -30°C to 0°C during the addition of the metal salt to prevent over-oxidation or side reactions, ensuring that the resulting racemate of the bisphosphine oxide compound is obtained with minimal impurity profiles that could otherwise poison downstream catalytic cycles.
Furthermore, the process allows for exceptional control over the stereochemical outcome through subsequent resolution steps, which is critical for producing enantiomerically pure ligands. The racemic mixture obtained from the oxidative coupling can be effectively separated using chiral resolving agents such as (-)- or (+)-O,O'-dibenzoyl tartaric acid (DBT) or di-p-tolyl tartrate (DTT) in solvents like ethyl acetate or alcohols. This resolution step forms diastereomeric salts that can be isolated via crystallization, followed by liberation of the free optically active phosphine oxide using mineral alkali. The ability to integrate this resolution seamlessly after the coupling step ensures that the final product meets the stringent enantiomeric excess (e.e.) specifications demanded by the pharmaceutical industry for asymmetric hydrogenation catalysts, thereby validating the process as a reliable source for high-purity pharmaceutical intermediates.
How to Synthesize Bisphosphine Oxide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory discovery to pilot plant production, emphasizing the importance of reagent quality and thermal management. The procedure begins with the preparation of the amide base, such as reacting diisopropylamine with butyllithium in THF under an inert argon atmosphere at temperatures below 0°C to ensure complete formation of the lithium amide species. Once the base is ready, the phosphine oxide substrate is added slowly to maintain the reaction temperature below -15°C, preventing thermal runaway during the exothermic deprotonation phase. After the lithiation is complete, the pre-cooled solution of the oxidizing metal salt is introduced, and the mixture is allowed to warm to room temperature over several hours to drive the coupling to completion before quenching with ammonium hydroxide to remove metal residues.
- React a phosphine oxide compound with 0.5-3 equivalents of a lithium or magnesium amide compound in an organic solvent at temperatures between -70°C and 20°C.
- Add 0.5-3 equivalents of an oxidatively-acting metal salt or metal salt complex to the suspension within the same temperature range to obtain a racemate.
- Optionally perform racemate cleavage using resolving agents like dibenzoyl tartaric acid, followed by conversion to bisphosphine oxides if starting from bisphosphonates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this single-step oxidative coupling technology offers profound strategic advantages regarding cost structure and supply continuity. By eliminating the high-temperature Ullmann coupling step and the associated need for stoichiometric copper, the process significantly reduces raw material costs and energy consumption, leading to substantial cost savings in fine chemical intermediates manufacturing. The removal of harsh reaction conditions also translates to reduced wear and tear on reactor equipment and lower maintenance downtime, further enhancing the economic viability of large-scale production. Additionally, the simplified workup procedure, which avoids the filtration of intractable copper sludge, accelerates batch turnover times and improves overall plant throughput, allowing suppliers to respond more agilely to market demand fluctuations without compromising on delivery schedules.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric copper reagents and the reduction of energy-intensive high-temperature steps directly lower the variable cost per kilogram of the final product. Furthermore, the improved yield profile and reduced formation of intractable by-products minimize waste disposal costs and maximize the utilization of starting materials, creating a leaner and more cost-effective production model that enhances competitiveness in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: The use of readily available reagents such as iron chloride and common organic solvents like THF reduces dependency on scarce or volatile raw materials, thereby stabilizing the supply chain against external shocks. The milder reaction conditions also improve operational safety, reducing the risk of unplanned shutdowns due to thermal incidents, which ensures a consistent and reliable flow of high-purity pharmaceutical intermediates to downstream customers who depend on uninterrupted supply for their own drug manufacturing processes.
- Scalability and Environmental Compliance: The process is inherently scalable due to its manageable exotherm and lack of hazardous high-pressure or high-temperature requirements, facilitating the commercial scale-up of complex polymer additives and ligand precursors from kilogram to multi-ton scales. Moreover, the significant reduction in heavy metal waste aligns with increasingly stringent environmental regulations, simplifying the permitting process and reducing the environmental footprint of the manufacturing facility, which is a key consideration for sustainable sourcing strategies adopted by major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is essential for process chemists aiming to adapt this technology for specific substrate classes or to troubleshoot potential variations in reaction performance. The answers provided reflect the specific conditions and parameters validated in the patent examples, ensuring that the guidance is grounded in proven experimental results rather than theoretical speculation.
Q: What are the critical temperature controls for the oxidative coupling step?
A: The process requires strict temperature maintenance between -70°C and 20°C, with a preferred range of -30°C to 0°C during the addition of oxidizing metal salts to ensure optimal yield and minimize by-product formation.
Q: Which resolving agents are suitable for separating the racemic bisphosphine oxides?
A: Effective resolution can be achieved using (-)- or (+)-O,O'-dibenzoyl tartaric acid (DBT) or (-)- or (+)-O,O'-di-p-tolyl tartrate (DTT) in inert organic solvents such as chloroform, ethyl acetate, or alcohols at temperatures between 0°C and 60°C.
Q: How does this method compare to traditional Ullmann coupling for these intermediates?
A: Unlike the traditional two-step Ullmann reaction which requires stoichiometric copper and high temperatures up to 200°C, this novel method utilizes a single-step oxidative coupling at mild temperatures, significantly reducing energy consumption and heavy metal waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthesis technologies play in securing the supply chain for next-generation catalytic systems. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications and rigorous QC labs testing every batch to guarantee performance in your asymmetric hydrogenation applications. Our infrastructure is designed to handle the specific thermal and safety requirements of oxidative coupling reactions, providing a secure and compliant manufacturing environment for your most sensitive projects.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this single-step methodology for your specific application needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your R&D timelines and your long-term procurement strategy for high-value chemical intermediates.
