Advanced Telescoped Synthesis of Sulfachloropyridazine Sodium for Scalable Veterinary Applications
Introduction to Next-Generation Sulfachloropyridazine Sodium Manufacturing
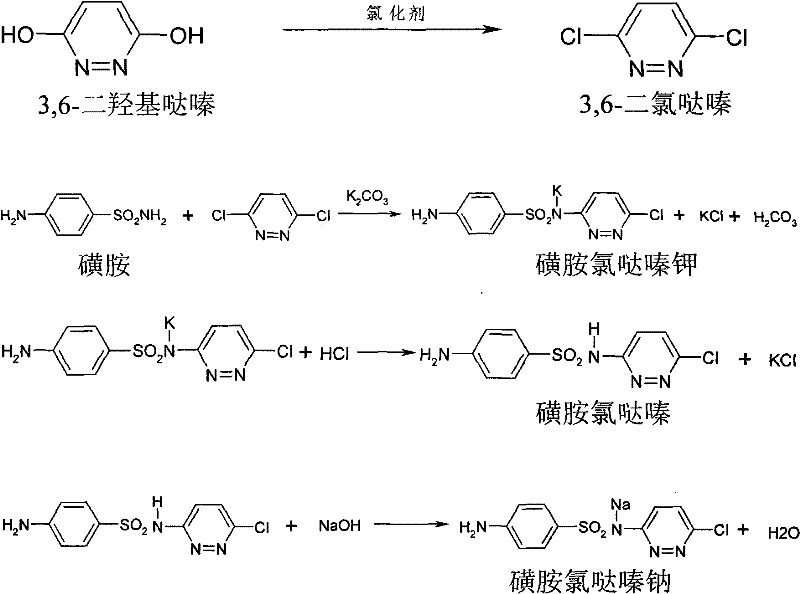
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways for producing essential veterinary intermediates, and the methodology detailed in patent CN101914064B represents a significant leap forward in this domain. This specific intellectual property outlines a robust, cost-effective preparation method for Sulfachloropyridazine Sodium, a critical sulfonamide antiseptic widely utilized for treating digestive tract infections and coccidiosis in livestock. Unlike traditional synthetic routes that rely on expensive reagents and laborious isolation steps, this innovation introduces a telescoped process that streamlines the conversion of 3,6-dihydroxypyridazine directly into the final active pharmaceutical ingredient precursor. By integrating chlorination and condensation steps without the need for intermediate purification, the technology addresses key pain points regarding operational efficiency and environmental impact. For R&D directors and procurement specialists alike, understanding this shift from batch-heavy processing to continuous-flow-friendly chemistry is essential for maintaining competitiveness in the global supply chain for veterinary drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sulfachloropyridazine Sodium has been plagued by high operational costs and complex downstream processing requirements. Traditional protocols typically employ phosphorus oxychloride (POCl3) as the primary chlorinating agent, which is not only prohibitively expensive but also generates significant corrosive waste streams that complicate environmental compliance. Furthermore, the conventional workflow necessitates the complete isolation and purification of the intermediate, 3,6-dichloropyridazine, often involving tedious fusion and water separation techniques that are difficult to control on a large scale. These multi-step isolation procedures introduce multiple points of potential yield loss and increase the overall cycle time, making the supply chain vulnerable to delays. The rigorous temperature and acidity controls required during the separation of muriate salts further burden the production team, leading to higher energy consumption and increased safety risks associated with handling hazardous intermediates in solid form.
The Novel Approach
In stark contrast, the patented methodology revolutionizes this landscape by adopting a telescoped strategy that bypasses the isolation of 3,6-dichloropyridazine entirely. Instead of relying on costly phosphorus oxychloride, the process utilizes more economical chlorinating agents such as phosphorus trichloride, thionyl chloride, or even chlorine gas, reacting them with 3,6-dihydroxypyridazine at moderate temperatures between 80°C and 100°C. Following the chlorination, the reaction mixture undergoes solvent extraction using organic solvents like o-dichlorobenzene, allowing the intermediate to proceed directly into the condensation phase with sulfanilamide. This seamless transition eliminates the need for refining and purification of the chlorinated intermediate, as the content is simply verified via HPLC before moving to the next stage. The result is a drastically simplified operation that reduces solvent usage, minimizes waste generation, and accelerates the overall production timeline.
Mechanistic Insights into Telescoped Chlorination and Condensation
The core chemical transformation in this process involves a nucleophilic substitution where the hydroxyl groups of the pyridazine ring are replaced by chlorine atoms, facilitated by the chosen chlorinating agent. In the initial stage, the reaction between 3,6-dihydroxypyridazine and agents like phosphorus trichloride proceeds through the formation of reactive chlorophosphite intermediates, which effectively convert the dihydroxy structure into 3,6-dichloropyridazine. The patent specifies a mass ratio of chlorinating agent to substrate ranging from 1:1 to 5:1, ensuring complete conversion while allowing for the recovery of excess reagent under reduced pressure. This step is critical because incomplete chlorination would lead to impurities that could interfere with the subsequent coupling reaction, yet the process is robust enough to tolerate minor variations due to the subsequent purification steps. The use of solvents such as o-dichlorobenzene or chlorobenzene not only facilitates the extraction of the product but also serves as the reaction medium for the next phase, creating a homogeneous environment for the condensation.
Following the chlorination, the mechanism shifts to a nucleophilic aromatic substitution where sulfanilamide attacks the electron-deficient chloropyridazine ring in the presence of potassium carbonate as a base. This condensation occurs at elevated temperatures between 140°C and 180°C, promoting the displacement of one chlorine atom by the sulfonamide nitrogen. A crucial aspect of quality control in this mechanism is the post-reaction workup, where the mixture is treated with hot water and activated carbon to remove colored impurities and byproducts. The pH of the resulting solution is then carefully adjusted to between 4.0 and 5.0 using dilute hydrochloric acid, which induces the precipitation of the free acid form of sulfachloropyridazine. This precise pH control exploits the isoelectric properties of the molecule to maximize recovery while leaving soluble impurities in the aqueous phase, ensuring that the final conversion to the sodium salt yields a product with high purity specifications suitable for veterinary use.
How to Synthesize Sulfachloropyridazine Sodium Efficiently
Implementing this synthesis route requires careful attention to thermal management and stoichiometry to ensure optimal yields and safety. The process begins with the charging of 3,6-dihydroxypyridazine and the chlorinating agent into a reactor, where the exothermic nature of the reaction must be managed by controlling the heating rate to maintain the temperature within the 80°C to 100°C window. Once the chlorination is complete and excess reagent is recovered, the crude solution is extracted and transferred for condensation with sulfanilamide and potassium carbonate. The detailed standardized operating procedures for temperature ramping, solvent recovery, and crystallization parameters are critical for reproducibility and are outlined in the technical guide below.
- Perform chlorination of 3,6-dihydroxypyridazine using phosphorus trichloride or thionyl chloride at 80-100°C, followed by solvent extraction without isolating the intermediate.
- Conduct condensation reaction by adding sulfanilamide and potassium carbonate to the extracted solution at 140-180°C, followed by decolorization and pH adjustment to 4.0-5.0.
- React the isolated sulfachloropyridazine with 30% sodium hydroxide solution and crystallize at temperatures below 5°C to obtain the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits that extend beyond simple chemical efficiency. By eliminating the isolation and purification of the intermediate 3,6-dichloropyridazine, the process removes an entire unit operation from the manufacturing flow, which directly translates to reduced labor costs and lower capital expenditure on equipment. The ability to use cheaper chlorinating agents like phosphorus trichloride instead of phosphorus oxychloride significantly lowers the raw material cost base, providing a competitive edge in pricing for the final veterinary active ingredient. Furthermore, the simplified workflow reduces the overall cycle time, allowing manufacturers to respond more rapidly to market demand fluctuations and shorten lead times for high-purity veterinary intermediates.
- Cost Reduction in Manufacturing: The elimination of the intermediate isolation step removes the need for energy-intensive drying and purification processes, leading to significant savings in utility consumption and operational overhead. Additionally, the substitution of expensive reagents with more affordable alternatives like phosphorus trichloride reduces the direct material cost per kilogram of product. The telescoped nature of the reaction also minimizes solvent losses typically associated with multiple filtration and washing steps, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: Simplifying the synthesis pathway reduces the number of potential failure points in the manufacturing process, thereby increasing the reliability of supply for downstream pharmaceutical formulators. The robustness of the reaction conditions, which tolerate a range of chlorinating agents and solvents, ensures that production can continue even if specific raw materials face temporary shortages. This flexibility allows supply chain managers to diversify their vendor base for inputs without compromising the quality or consistency of the final Sulfachloropyridazine Sodium output.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the removal of solid-state intermediate handling reduces the risk of blockages and equipment fouling in large reactors. From an environmental perspective, the reduced generation of solid waste and the ability to recover and recycle solvents like o-dichlorobenzene align with increasingly stringent global regulations on chemical manufacturing emissions. This eco-friendly profile not only mitigates regulatory risk but also enhances the brand value of the final product in markets that prioritize sustainable sourcing of animal health ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable foundation for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the advantages of the telescoped process over conventional methods?
A: The novel method eliminates the need to isolate and purify the intermediate 3,6-dichloropyridazine, significantly reducing operational complexity and solvent consumption compared to traditional fusion and water separation techniques.
Q: Which chlorinating agents are suitable for this synthesis?
A: The process allows for the use of cost-effective chlorinating agents such as phosphorus trichloride, thionyl chloride, or chlorine gas, offering flexibility over the traditionally expensive phosphorus oxychloride.
Q: How is product purity controlled during the condensation step?
A: Purity is managed through activated carbon decolorization and precise pH control between 4.0 and 5.0 during precipitation, ensuring high-quality crude material before final salt formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfachloropyridazine Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern veterinary pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101914064B are fully realized in practical manufacturing environments. We are committed to delivering high-purity Sulfachloropyridazine Sodium that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific thermal and solvent requirements of this telescoped process, guaranteeing a consistent supply of this vital intermediate for your animal health formulations.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized production methods can enhance your operational efficiency and reduce total landed costs.
