Optimizing Sulfachloropyridazine Sodium Production: A Technical Breakdown for Global Supply Chains
The global demand for effective veterinary antimicrobial agents continues to drive innovation in the synthesis of sulfonamide derivatives, particularly Sulfachloropyridazine Sodium. This compound serves as a critical active pharmaceutical ingredient (API) for treating gastrointestinal infections and coccidiosis in livestock, necessitating robust and scalable manufacturing protocols. A pivotal advancement in this domain is detailed in patent CN101914064A, which discloses a refined preparation method that significantly alters the economic and operational landscape of production. By shifting away from traditional, cumbersome isolation techniques toward a telescoped synthetic strategy, this technology offers a compelling value proposition for manufacturers seeking to optimize their supply chains. The core innovation lies in the strategic modification of the chlorination and condensation phases, allowing for direct progression between reaction steps without the need for intermediate purification. This approach not only mitigates the risks associated with handling unstable intermediates but also streamlines the overall workflow, ensuring higher throughput and consistent quality. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for evaluating potential partnerships and securing a reliable source of high-purity veterinary drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
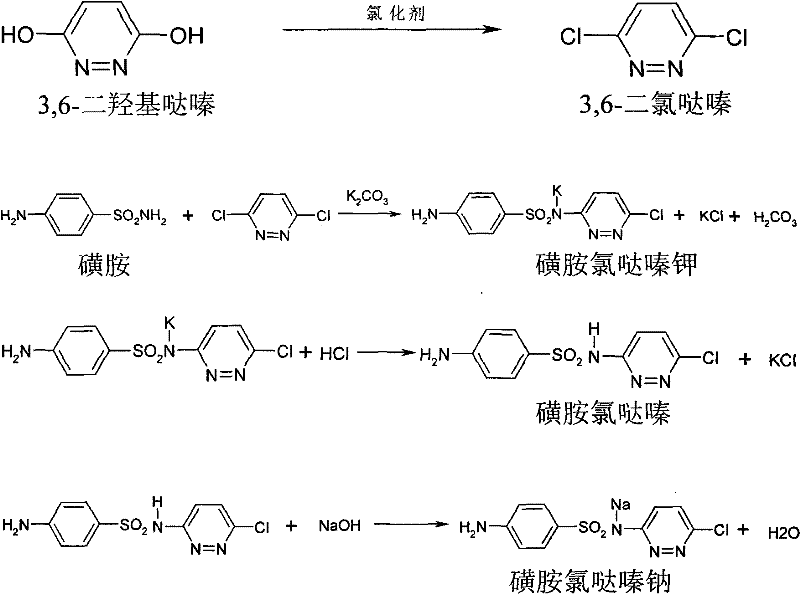
Historically, the industrial synthesis of Sulfachloropyridazine Sodium has relied on a multi-step sequence beginning with the ring expansion of maleic anhydride and hydrazine hydrate to form 3,6-dihydroxypyridazine. The subsequent chlorination step traditionally employs phosphorus oxychloride (POCl3) as the primary chlorinating agent, a reagent known for its high cost and hazardous nature. The post-treatment of this chlorination reaction presents significant engineering challenges, requiring the reaction liquid to flow concurrently with ammonia water under strictly controlled temperature and acidity conditions to precipitate the chloride. Furthermore, the resulting precipitate necessitates a laborious purification process involving melting and water separation to achieve acceptable purity levels. These operational bottlenecks not only inflate the production costs due to energy consumption and reagent expenses but also introduce variability in product quality. The complexity of managing corrosive byproducts and the stringent safety protocols required for handling phosphorus oxychloride further exacerbate the environmental footprint, making the conventional route less attractive for modern, sustainability-focused manufacturing facilities.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a paradigm shift by utilizing more economical chlorinating agents such as thionyl chloride, chlorine gas, phosphorus trichloride, or phosphorus pentachloride. The process operates at a moderate temperature range of 80 to 100°C, facilitating the conversion of 3,6-dihydroxypyridazine to 3,6-dichloropyridazine with high efficiency. Crucially, the invention eliminates the isolation of the 3,6-dichloropyridazine intermediate; instead, the reaction mixture is subjected to solvent extraction using organic solvents like o-dichlorobenzene or chlorobenzene. This extract, containing the crude intermediate, is directly utilized in the subsequent condensation reaction with sulfanilamide and potassium carbonate. By bypassing the isolation, drying, and melting purification steps, the process drastically reduces the number of unit operations and minimizes material loss. The integration of HPLC content measurement allows for precise stoichiometric adjustments before the condensation phase, ensuring that the reaction proceeds with optimal efficiency. This telescoped approach represents a significant leap forward in process intensification, offering a cleaner, faster, and more cost-effective pathway to the final API.
Mechanistic Insights into Chlorination and Condensation Dynamics
The chemical transformation begins with the nucleophilic substitution of the hydroxyl groups on the pyridazine ring, a reaction driven by the electrophilic nature of the chosen chlorinating agents. When agents like phosphorus trichloride are employed, the mechanism involves the activation of the hydroxyl oxygen, rendering it a better leaving group, which is subsequently displaced by a chloride ion. The selection of the solvent system is paramount here; solvents such as o-dichlorobenzene not only facilitate the extraction of the lipophilic 3,6-dichloropyridazine from the polar reaction byproducts but also serve as the reaction medium for the next stage. This dual functionality simplifies the process flow and ensures that the reactive intermediate remains in solution, preventing decomposition that might occur during solid-state handling. The maintenance of the reaction temperature between 80 and 100°C is critical to balance the reaction kinetics against the potential for over-chlorination or ring degradation, ensuring a clean profile of the dichloro-intermediate ready for the next coupling step.
Following the chlorination, the condensation reaction involves the nucleophilic attack of the sulfonamide nitrogen on the electron-deficient carbon of the pyridazine ring, displacing one of the chlorine atoms. This step is promoted by the presence of potassium carbonate, which acts as a base to deprotonate the sulfonamide, thereby increasing its nucleophilicity. The reaction is conducted at elevated temperatures, typically between 140 and 180°C, to overcome the activation energy barrier associated with aromatic nucleophilic substitution. Post-reaction processing includes a decolorization step using activated carbon, which is essential for removing trace colored impurities and residual organic byproducts that could affect the final API's appearance and stability. The subsequent pH adjustment to the range of 4.0 to 5.0 using dilute hydrochloric acid is a precise control point; it ensures the selective precipitation of the sulfachloropyridazine while keeping soluble impurities in the aqueous phase. This careful manipulation of acid-base properties is fundamental to achieving the high purity standards required for veterinary applications, effectively controlling the impurity profile without the need for complex chromatographic separations.
How to Synthesize Sulfachloropyridazine Sodium Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters and sequential processing to maximize yield and purity. The protocol is designed to be robust, allowing for direct transition from the chlorination extract to the condensation reactor, thereby minimizing hold times and exposure of the intermediate to potentially degrading conditions. Operators must ensure rigorous monitoring of the HPLC content of the extracted 3,6-dichloropyridazine solution to accurately calculate the required amounts of sulfanilamide and base for the subsequent step. The following guide outlines the standardized operational framework derived from the patent data, providing a clear roadmap for laboratory and pilot-scale execution.
- Perform chlorination of 3,6-dihydroxypyridazine using cost-effective agents like phosphorus trichloride at 80-100°C, followed by solvent extraction.
- Conduct condensation directly in the extract using sulfanilamide and potassium carbonate at 140-180°C, followed by decolorization and pH adjustment to 4.0-5.0.
- React the resulting sulfachloropyridazine with aqueous sodium hydroxide, cool to crystallize, and filter to obtain the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method translates into tangible strategic benefits that extend beyond simple unit cost calculations. The primary advantage lies in the substantial reduction of raw material costs achieved by replacing expensive phosphorus oxychloride with more abundant and affordable chlorinating agents like phosphorus trichloride or thionyl chloride. Furthermore, the elimination of intermediate isolation and purification steps significantly lowers utility consumption, including energy for heating and cooling, as well as labor costs associated with additional filtration and drying operations. This streamlined process flow enhances the overall equipment effectiveness (OEE) by reducing cycle times and freeing up reactor capacity for additional batches. Consequently, manufacturers can offer more competitive pricing structures while maintaining healthy margins, a critical factor in the price-sensitive veterinary pharmaceutical market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of high-cost reagents and the removal of energy-intensive purification stages. By avoiding the melting and water separation steps required in traditional methods, the facility saves significant amounts of steam and cooling water, directly lowering the variable cost per kilogram of production. Additionally, the ability to recycle solvents like o-dichlorobenzene from the extraction and condensation phases further contributes to cost efficiency, reducing the net consumption of organic solvents. The simplified workflow also reduces the likelihood of batch failures due to handling errors during intermediate transfer, thereby improving the overall yield and reducing waste disposal costs associated with off-spec material.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on readily available commodity chemicals such as sulfanilamide, potassium carbonate, and common chlorinating agents mitigates the risk of raw material shortages. Unlike specialized reagents that may have limited suppliers or long lead times, the inputs for this process are widely sourced from the global chemical market, ensuring continuity of supply even during market fluctuations. The robustness of the telescoped process also means that production schedules are less susceptible to delays caused by equipment bottlenecks in purification units. This reliability allows for more accurate forecasting and inventory management, enabling downstream partners to maintain optimal stock levels of the finished veterinary API without the need for excessive safety buffers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with the solvent extraction and condensation steps being easily adaptable from pilot plants to large-scale commercial reactors. The reduction in waste generation, particularly the avoidance of phosphorus-rich wastewater streams associated with phosphorus oxychloride hydrolysis, simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations. The use of activated carbon for decolorization is a proven, scalable technology that effectively manages organic impurities without generating hazardous sludge. These environmental advantages not only reduce the cost of waste treatment but also enhance the corporate sustainability profile of the manufacturer, aligning with the ESG (Environmental, Social, and Governance) goals of major multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Sulfachloropyridazine Sodium using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality control measures inherent in the process. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this supply source into their existing manufacturing networks.
Q: Why is the traditional phosphorus oxychloride method considered less efficient?
A: Traditional methods utilizing phosphorus oxychloride involve expensive reagents and complex post-treatment controls, such as strict temperature regulation during ammonia water co-current flow, which increases operational complexity and waste generation.
Q: How does the novel telescoped process improve product quality?
A: By eliminating the isolation and melting purification steps of the intermediate 3,6-dichloropyridazine, the new method reduces thermal degradation risks and simplifies impurity removal through direct solvent extraction and activated carbon decolorization.
Q: What are the key cost drivers addressed in this patent technology?
A: The primary cost reductions are achieved by substituting expensive chlorinating agents with cheaper alternatives like phosphorus trichloride and by removing unit operations associated with intermediate purification and drying.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfachloropyridazine Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes in the veterinary pharmaceutical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patented processes like CN101914064A are fully realized in practical manufacturing environments. We are committed to delivering Sulfachloropyridazine Sodium that meets stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify identity and assay. Our infrastructure is designed to handle the specific solvent systems and reaction conditions required for this telescoped synthesis, guaranteeing a consistent supply of high-quality intermediates for your formulation needs.
We invite you to engage with our technical procurement team to discuss how our optimized production capabilities can support your supply chain objectives. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to request specific COA data and route feasibility assessments to validate the compatibility of our material with your downstream processes. Together, we can drive value and efficiency in the global veterinary healthcare market.
