Revolutionizing Brivaracetam Production: A High-Purity Chiral Pool Synthesis Strategy
Revolutionizing Brivaracetam Production: A High-Purity Chiral Pool Synthesis Strategy
The pharmaceutical landscape for antiepileptic drugs continues to evolve, driven by the demand for higher purity intermediates and more sustainable manufacturing processes. Patent CN112110843A introduces a groundbreaking preparation method for Brivaracetam, a third-generation antiepileptic agent developed by UCB. This novel approach leverages a chiral pool strategy, utilizing commercially available (R)-4-propyl-dihydro-2(3H)-furanone as the foundational building block. By bypassing the traditional reliance on racemic mixtures and subsequent resolution, this technology offers a direct pathway to high-optical-purity Brivaracetam. The significance of this development lies in its ability to streamline the supply chain for reliable pharmaceutical intermediate suppliers, ensuring consistent quality without the bottleneck of chiral chromatography. As we delve into the technical specifics, it becomes evident that this four-step synthesis represents a paradigm shift in cost reduction in API manufacturing, addressing both economic and regulatory pressures facing modern generic drug producers.
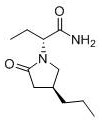
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Brivaracetam has been plagued by inefficiencies inherent in racemic synthesis strategies. Traditional routes often commence with racemic 4-propyl-dihydrofuran-2-one, necessitating a complex downstream separation process to isolate the desired enantiomer. As illustrated in prior art methodologies, these pathways typically involve ring-opening and acylation followed by a closure reaction that yields a mixture of the target molecule and its diastereomers. The critical flaw in this approach is the absolute requirement for silica gel column chromatography and chiral preparative HPLC to achieve pharmaceutical-grade purity. These purification steps are not only capital-intensive but also significantly limit throughput, creating a bottleneck for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the use of racemic starting materials inherently caps the theoretical yield at 50%, generating substantial chemical waste and increasing the environmental footprint of the production process, which is increasingly scrutinized by global regulatory bodies.
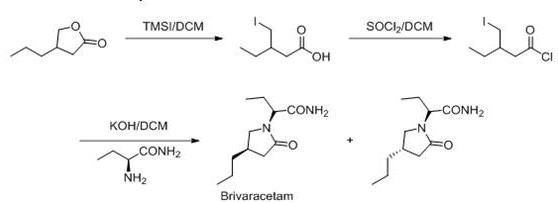
The Novel Approach
In stark contrast, the methodology disclosed in CN112110843A circumvents these historical hurdles by employing a chirally pure starting material from the outset. This strategic choice fundamentally alters the process economics and technical feasibility. The new route proceeds through a logical sequence of ring-opening, halogenation, amidation, and finally, a highly stereoselective cyclization. By locking the stereochemistry early in the synthesis via the (R)-configured lactone, the process eliminates the formation of unwanted enantiomeric impurities that plague racemic routes. This results in a streamlined workflow where the final product can be obtained with exceptional purity simply through recrystallization, completely obviating the need for chiral column separation. For procurement teams, this translates to a drastic simplification of the Bill of Materials (BOM) and a significant reduction in processing time, directly enhancing the reliability of the Brivaracetam supplier network by removing fragile purification dependencies.
Mechanistic Insights into Base-Mediated Intramolecular Cyclization
The cornerstone of this synthesis is the final cyclization step, which transforms the linear diamide precursor into the bicyclic Brivaracetam structure. This transformation is mediated by strong, non-nucleophilic bases such as Lithium Diisopropylamide (LDA) or Potassium Bis(trimethylsilyl)amide (KHMDS). The reaction mechanism involves the deprotonation of the alpha-carbon adjacent to the amide carbonyl, generating a reactive enolate species. This enolate then undergoes an intramolecular nucleophilic attack on the adjacent carbon bearing the halogen leaving group (iodine or bromine), displacing the halide and closing the five-membered pyrrolidinone ring. The success of this step relies heavily on precise temperature control, typically maintained between -80°C and -70°C. These cryogenic conditions are essential to suppress competing side reactions, such as elimination or non-selective deprotonation, which could lead to impurity formation. The stereochemical outcome is dictated by the existing chiral center derived from the starting lactone, ensuring that the newly formed stereocenter aligns perfectly to produce the (2S, 4R) configuration required for biological activity.
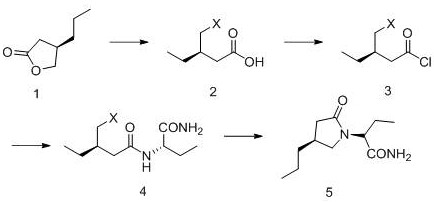
Impurity control is rigorously managed throughout this mechanistic pathway, particularly during the workup of the cyclization reaction. The patent specifies quenching the reaction with saturated ammonium chloride solution, followed by extraction and drying. A critical aspect of the purification strategy is the recrystallization process using aprotic solvents like ethyl acetate and methyl tert-butyl ether. This step is highly effective at rejecting trace organic impurities and residual salts, yielding a final product with an HPLC purity exceeding 99.70%. The ability to achieve such high purity standards without chromatographic intervention is a testament to the robustness of the reaction design. For R&D directors, this implies a stable and reproducible process where critical quality attributes (CQAs) are built into the chemistry rather than relying solely on downstream purification, thereby reducing the risk of batch failure and ensuring consistent supply continuity.
How to Synthesize Brivaracetam Efficiently
The execution of this synthesis requires careful attention to reagent stoichiometry and thermal management, particularly during the exothermic acylation and the cryogenic cyclization steps. The process begins with the ring opening of the chiral lactone using trimethyliodosilane (TMSI), followed by conversion to the acid chloride using thionyl chloride. The subsequent coupling with S-2-aminobutanamide forms the key linear precursor. While the general chemistry is straightforward, the specific operational parameters—such as the dropwise addition of reagents and the maintenance of inert atmospheres—are critical for maximizing yield and safety. The detailed standardized operating procedures, including exact molar ratios and specific workup protocols necessary for GMP compliance, are outlined in the technical guide below.
- Ring Opening: React (R)-4-propyl-dihydro-2(3H)-furanone with trimethyliodosilane (TMSI) in dichloromethane to generate the open-chain iodo-acid intermediate.
- Activation and Amidation: Convert the acid to an acid chloride using thionyl chloride, then couple with S-2-aminobutanamide to form the diamide precursor.
- Cyclization: Treat the diamide precursor with a strong base like LDA at -70°C to induce intramolecular cyclization, forming the pyrrolidinone ring of Brivaracetam.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains. By shifting from a resolution-based process to a chiral pool synthesis, manufacturers can achieve substantial cost savings and operational efficiencies. The elimination of chiral chromatography not only reduces the consumption of expensive stationary phases and solvents but also drastically shortens the cycle time per batch. This efficiency gain allows for higher throughput in existing facilities, effectively increasing capacity without the need for significant capital expenditure on new equipment. Furthermore, the use of commodity chemicals like thionyl chloride and standard strong bases ensures that the raw material supply chain remains robust and less susceptible to geopolitical disruptions compared to specialized chiral catalysts.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the removal of the chiral separation step. In traditional processes, chiral preparative HPLC is a major cost center, consuming vast amounts of solvent and requiring specialized, high-maintenance equipment. By designing a route that inherently produces the correct stereoisomer, this method eliminates those variable costs entirely. Additionally, the overall yield improvement—avoiding the theoretical 50% loss of racemic resolution—means that less starting material is required to produce the same amount of API. This fundamental increase in atom economy translates directly to a lower cost of goods sold (COGS), providing a competitive edge in the generic marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, commercially sourced starting materials. The primary chiral building block, (R)-4-propyl-dihydro-2(3H)-furanone, is a stable and accessible chemical, reducing the risk of supply shortages associated with custom-synthesized chiral intermediates. Moreover, the simplified process flow, consisting of only four distinct chemical transformations, reduces the number of potential failure points in the manufacturing line. Fewer unit operations mean less handling, lower risk of cross-contamination, and faster turnaround times, ensuring that high-purity Brivaracetam can be delivered to customers with greater consistency and shorter lead times.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its reliance on standard unit operations such as liquid-liquid extraction and crystallization, which are easily transferred from pilot plant to multi-ton production scales. The avoidance of heavy metal catalysts or exotic reagents simplifies waste stream management and reduces the burden on wastewater treatment facilities. The process generates fewer hazardous byproducts compared to resolution methods, aligning with green chemistry principles. This environmental compatibility facilitates easier regulatory approval and reduces the long-term liability associated with hazardous waste disposal, making it an attractive option for manufacturers aiming to meet stringent sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and performance of the method. Understanding these nuances is crucial for technical teams evaluating the transfer of this technology to their own production sites.
Q: Does this synthesis route require chiral chromatography?
A: No, the patented method utilizes a chiral starting material ((R)-4-propyl-dihydro-2(3H)-furanone) and stereoselective cyclization, eliminating the need for expensive chiral preparative HPLC separation.
Q: What is the achieved HPLC purity of the final Brivaracetam product?
A: The process yields Brivaracetam with an HPLC purity greater than 99.70%, with individual impurities controlled below 0.1%, meeting strict API specifications.
Q: What are the critical reaction conditions for the final cyclization step?
A: The final ring-closing step requires cryogenic conditions, specifically between -80°C and -70°C, using strong bases such as LDA or KHMDS to ensure high diastereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Supplier
The technical sophistication of this four-step chiral synthesis underscores the complexity involved in producing high-quality antiepileptic intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex stereoselective reactions like the LDA-mediated cyclization are executed with precision. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace diastereomers down to 0.1% levels. We understand that the transition from lab-scale patent examples to industrial reality requires deep process engineering expertise, which is exactly what our CDMO team brings to every partnership.
We invite you to explore how this optimized route can transform your supply chain economics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term API sourcing strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
