Advanced Manufacturing of RET Kinase Inhibitor Intermediates: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of RET Kinase Inhibitor Intermediates: A Technical Breakthrough for Global Supply Chains
The pharmaceutical landscape for oncology treatments is rapidly evolving, with RET kinase inhibitors like Selpercatinib (LOXO-292) representing a critical frontier in targeted therapy for thyroid and lung cancers. As demand for these life-saving medications surges, the efficiency and reliability of their supply chains become paramount. Patent CN111548349A introduces a transformative synthetic methodology for producing key intermediates, specifically compounds 5a and 5b, which serve as the foundational scaffolds for this class of inhibitors. This technical insight report analyzes the profound implications of this novel route, contrasting it against legacy methods to highlight its potential for revolutionizing the manufacturing economics of high-purity pharmaceutical intermediates. By leveraging hydroxylamine-O-sulfonic acid as a versatile nitrogen source, this innovation addresses long-standing bottlenecks related to reagent stability, cost, and environmental impact.
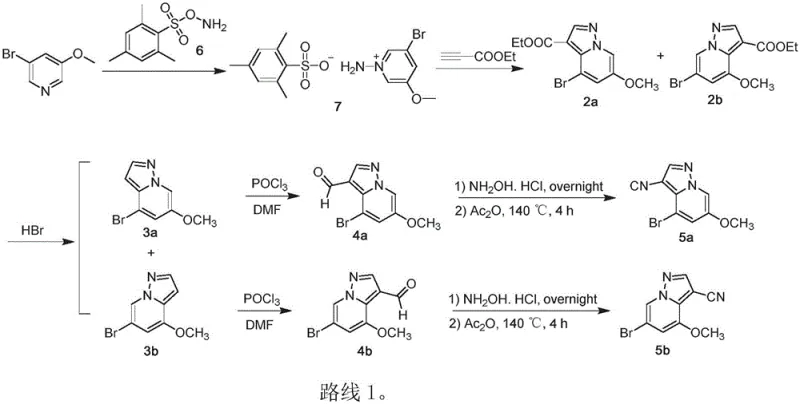
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these critical pyrazolo[1,5-a]pyridine scaffolds has been plagued by significant economic and operational inefficiencies, as illustrated in the prior art Route 1. The conventional pathway relies heavily on the preparation of compound 7, which necessitates the use of compound 6. Compound 6 is derived from the reaction of 2,4,6-trimethylbenzenesulfonyl chloride and BocNHOH, both of which are prohibitively expensive reagents that severely inflate the cost of goods sold (COGS). Furthermore, compound 6 exhibits poor chemical stability, introducing substantial risk into the supply chain regarding shelf-life and batch consistency. From a process chemistry perspective, the atom economy of this traditional route is suboptimal, generating excessive waste that complicates downstream purification. Additionally, the conversion of aldehydes 4a/4b to the final nitriles 5a/5b traditionally requires a cumbersome two-step sequence involving oxime formation followed by dehydration with acetic anhydride. This dehydration step demands strictly anhydrous conditions and elevated temperatures, often resulting in deeply colored products that require extensive purification and generating difficult-to-treat acidic waste streams.
The Novel Approach
In stark contrast, the methodology disclosed in CN111548349A offers a streamlined, cost-effective alternative that fundamentally reimagines the construction of the pyrazole ring and the installation of the nitrile group. As depicted in the comprehensive reaction scheme below, the new route bypasses the unstable sulfonamide intermediates entirely. Instead, it employs a direct cycloaddition strategy using hydroxylamine-O-sulfonic acid and alkyl propiolates on the pyridine substrate. This shift not only drastically reduces raw material costs but also enhances the robustness of the process by utilizing stable, commodity-grade chemicals. The most striking improvement is observed in the final transformation, where the aldehyde-to-nitrile conversion is achieved in a single pot. This eliminates the need for acetic anhydride, allowing the reaction to proceed under mild, aqueous-compatible conditions. The result is a whiter, higher-purity solid product with simplified waste management, representing a paradigm shift towards greener and more sustainable pharmaceutical manufacturing.
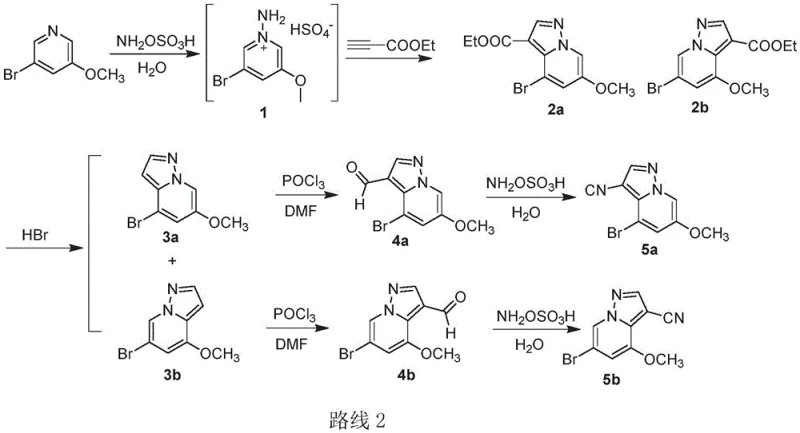
Mechanistic Insights into Hydroxylamine-O-Sulfonic Acid Mediated Cyclization
The core innovation of this synthetic strategy lies in the dual utility of hydroxylamine-O-sulfonic acid as both a nitrogen donor for ring closure and a dehydrating agent for nitrile formation. In the initial cyclization step, the reaction likely proceeds via the in situ generation of an N-aminopyridinium species, which subsequently undergoes a [3+2] cycloaddition with the electron-deficient alkyne (ethyl propiolate). This mechanism is highly efficient, yielding the ester intermediates 2a and 2b in excellent yields (over 80%) without the need for protecting groups. The subsequent hydrolysis and decarboxylation using hydrobromic acid cleanly afford the pyrazolo[1,5-a]pyridine cores 3a and 3b. The formylation step utilizes the classic Vilsmeier-Haack mechanism, where phosphorus oxychloride activates DMF to generate an electrophilic iminium species that attacks the electron-rich positions of the heterocycle. This step is remarkably high-yielding, often exceeding 95%, demonstrating the high reactivity of the substrate.
Perhaps the most mechanistically intriguing aspect is the direct conversion of aldehydes 4a/4b to nitriles 5a/5b. Traditionally, this transformation involves isolating an oxime intermediate and subjecting it to harsh dehydrating conditions. However, in this novel protocol, hydroxylamine-O-sulfonic acid facilitates the formation of the oxime and its subsequent dehydration in a continuous sequence within an aqueous amide solvent system. The presence of water, typically detrimental to nitrile synthesis, is tolerated and even beneficial here, likely assisting in the hydrolysis of the sulfonate leaving group. This tolerance for moisture eliminates the need for rigorous drying of solvents and reagents, significantly lowering the operational complexity. The reaction proceeds at ambient temperatures (15-35°C), preserving the integrity of the sensitive heterocyclic scaffold and preventing the formation of polymeric impurities often associated with thermal stress.
How to Synthesize RET Kinase Inhibitor Intermediates Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the yield and purity of the final intermediates 5a and 5b. The process begins with the careful addition of hydroxylamine-O-sulfonic acid to an aqueous suspension of 3-bromo-5-methoxypyridine, followed by heating to initiate the N-amination. Subsequent addition of the base and alkyne must be controlled to manage the exotherm of the cycloaddition. The downstream processing involves standard acid-base workups and crystallization techniques that are easily scalable. For the critical final step, maintaining the pH above 7.0 during the workup is essential to ensure the complete precipitation of the nitrile product while keeping inorganic salts in solution. The detailed standardized synthesis steps see guide below.
- React 3-bromo-5-methoxypyridine with hydroxylamine-O-sulfonic acid and water at 70-100°C, followed by base-catalyzed cycloaddition with alkyl propiolate to form esters 2a/2b.
- Hydrolyze and decarboxylate esters 2a/2b using aqueous HBr at 70-90°C to yield pyrazolo[1,5-a]pyridines 3a/3b.
- Perform Vilsmeier-Haack formylation on compounds 3a/3b using POCl3 in DMF at 0-25°C to generate aldehydes 4a/4b.
- Convert aldehydes 4a/4b directly to nitriles 5a/5b in a single step using hydroxylamine-O-sulfonic acid in aqueous amide solvent at ambient temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend far beyond simple chemistry. The primary value driver is the substantial reduction in raw material expenditure. By replacing high-cost specialty reagents like BocNHOH and tosyl chlorides with commodity chemicals like hydroxylamine-O-sulfonic acid, the direct material cost is significantly lowered. This cost structure provides a buffer against market volatility and allows for more competitive pricing in the final API market. Furthermore, the elimination of unstable intermediates reduces the risk of batch failures and inventory write-offs, ensuring a more predictable and reliable supply of critical starting materials for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the simplification of the process flow. Eliminating the two-step nitrile formation removes an entire unit operation, reducing labor, energy, and solvent consumption. The avoidance of acetic anhydride not only saves on reagent costs but also eliminates the generation of acetic acid waste, which requires neutralization and disposal. The high yields observed in the Vilsmeier-Haack step (up to 98%) minimize material loss, ensuring that expensive brominated precursors are efficiently converted into valuable product. These cumulative efficiencies translate into a drastically simplified cost model for the production of these complex heterocycles.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-proprietary reagents. Unlike specialized sulfonyl derivatives that may have limited suppliers and long lead times, hydroxylamine-O-sulfonic acid and alkyl propiolates are accessible from multiple global vendors. This diversification of the supply base mitigates the risk of shortages. Additionally, the robustness of the reaction conditions—specifically the tolerance for moisture and moderate temperatures—means that the process is less susceptible to variations in environmental conditions or minor deviations in operating parameters, leading to consistent batch quality and on-time delivery performance.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior. The removal of acetic anhydride reduces the hazard profile of the process, as it is a corrosive and lachrymatory agent. The aqueous workup conditions simplify wastewater treatment, as the effluent contains fewer organic contaminants compared to the traditional organic-solvent-heavy dehydration methods. The ability to produce white solids directly reduces the need for energy-intensive recrystallization or charcoal treatment steps. These factors collectively lower the environmental footprint of the manufacturing process, aligning with increasingly stringent global regulatory standards for green chemistry and sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms existing standards in terms of efficiency and product quality.
Q: How does this new route improve upon the prior art US20180134702?
A: The new route eliminates the need for expensive and unstable reagents like BocNHOH and 2,4,6-trimethylbenzenesulfonyl chloride. It replaces the traditional two-step dehydration of oximes with acetic anhydride with a direct, one-step conversion using hydroxylamine-O-sulfonic acid, significantly simplifying waste treatment and improving product color.
Q: What are the critical reaction conditions for the final nitrile formation?
A: The conversion of aldehydes 4a/4b to nitriles 5a/5b is conducted in a mixture of amide (DMF/DMAc) and water at mild temperatures (15-35°C). This avoids the harsh anhydrous conditions and high temperatures required by conventional acetic anhydride dehydration methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials like hydroxylamine-O-sulfonic acid and avoids moisture-sensitive steps. The high yields (up to 98% in formylation) and simplified workup procedures make it highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable RET Kinase Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. We have extensively evaluated the synthetic route described in CN111548349A and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this chemistry, including the safe handling of hydroxylamine derivatives and the precise temperature control needed for the Vilsmeier-Haack reaction. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of intermediates 5a and 5b meets the exacting standards required for oncology drug development.
We invite pharmaceutical companies and contract research organizations to collaborate with us to optimize their supply chains for RET kinase inhibitors. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this novel route can impact your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of these critical building blocks, ensuring that your development timelines remain on track and your commercial launch is successful.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
