Advanced Manufacturing of Selpercatinib Intermediate: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of Selpercatinib Intermediate: A Technical Breakthrough for Global Supply Chains
The pharmaceutical landscape for oncology treatments is continuously evolving, driven by the urgent need for potent and selective kinase inhibitors. A pivotal development in this domain is detailed in Chinese Patent CN113816955B, which discloses a novel and highly efficient preparation method for a critical intermediate of the RET kinase inhibitor, Selpercatinib (also known as LOXO-292). This patent addresses significant bottlenecks in the existing synthetic routes, offering a pathway that promises superior purity and operational simplicity. For R&D directors and procurement strategists alike, understanding the nuances of this technology is essential for securing a reliable supply of high-quality active pharmaceutical ingredients. The intermediate, identified as Compound 7 in the patent documentation, serves as the cornerstone for the final assembly of Selpercatinib, a drug vital for treating cancers with RET gene fusions or mutations. By leveraging the innovative chemistry outlined in this intellectual property, manufacturers can overcome historical challenges related to isomer contamination and low overall yields.
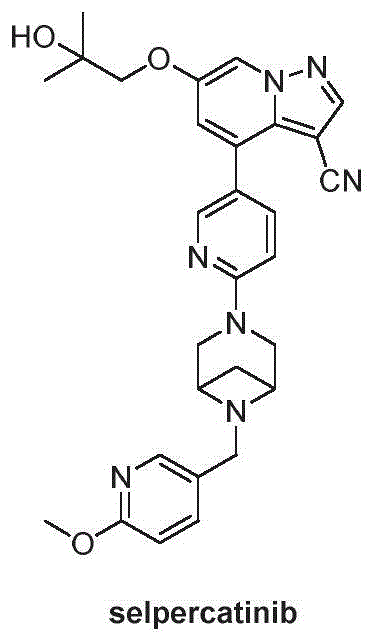
Selpercatinib represents a class of small molecule inhibitors designed to target the rearrangement-during-transfection (RET) proto-oncogene. The clinical efficacy of this drug relies heavily on the structural integrity and purity of its precursors. The patent explicitly highlights that while previous methods, such as those reported in U.S. Pat. No. US20180134702, could theoretically access the target molecule, they suffered from practical industrial limitations. Specifically, the legacy routes often resulted in the co-formation of isomers that were chemically similar and notoriously difficult to separate. These impurities not only complicated the purification process but also posed risks to the safety profile of the final drug substance. The new methodology presented in CN113816955B circumvents these issues through a redesigned synthetic logic that prioritizes selectivity and ease of isolation, marking a substantial advancement in the field of medicinal chemistry for oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations disclosed in CN113816955B, the synthesis of the key intermediate Compound 7 was fraught with inefficiencies that hindered large-scale production. As illustrated in the background section of the patent, traditional routes often involved multi-step sequences that lacked robustness. A primary technical hurdle was the generation of structural isomers during the construction of the pyrazolo[1,5-a]pyridine core. These isomers possessed physical properties nearly identical to the desired product, rendering standard purification techniques like crystallization ineffective. Consequently, manufacturers were forced to employ resource-intensive methods such as preparative chromatography, which is economically unviable for commercial API production. Furthermore, the cumulative yield of these older pathways was suboptimal, leading to excessive waste generation and inflated raw material costs. The inability to completely convert starting materials or intermediates meant that residual impurities would carry through to the final Selpercatinib synthesis, potentially compromising regulatory compliance and patient safety.
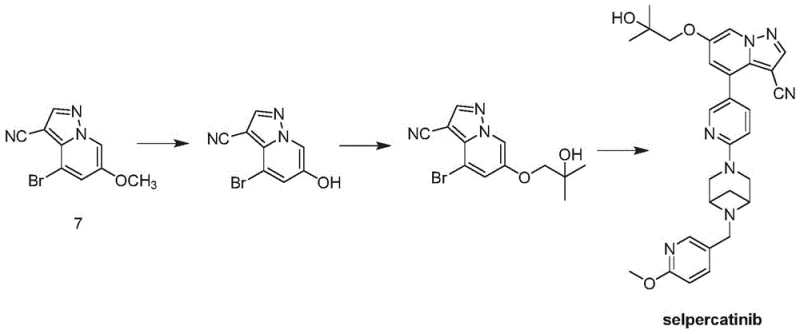
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach introduced in this patent offers a streamlined and high-yielding alternative. The core innovation lies in a strategic disconnection that utilizes a specific hydrazine salt (Compound 5) reacting with a halogenated acrylonitrile. This reaction sequence, facilitated by an organic base and an oxidant, constructs the heterocyclic framework with exceptional regioselectivity. By carefully controlling the reaction conditions, specifically the choice of base such as DBU and oxidant like DDQ, the process effectively suppresses the formation of unwanted isomers. The subsequent reduction step is equally elegant, utilizing zinc powder in a protic solvent to cleanly convert the intermediate nitrile species into the final target Compound 7. This two-step transformation from Compound 5 to Compound 7 is not only shorter but also operationally simpler, allowing for direct isolation of the product via filtration. The result is a process that delivers the intermediate with a purity exceeding 99.5% without the need for further recrystallization, a feat that dramatically simplifies the downstream processing workflow.
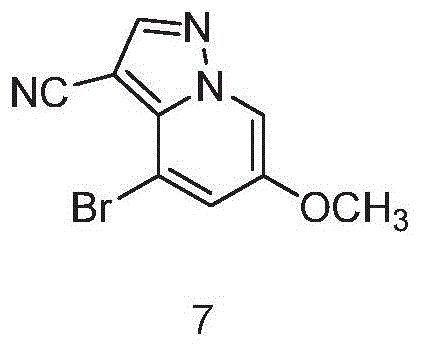
Mechanistic Insights into Oxidative Cyclization and Reduction
The chemical elegance of this new route is underpinned by a sophisticated understanding of heterocyclic formation mechanisms. The first critical transformation involves the reaction of the hydrazinium salt (Compound 5) with a halogenated acrylonitrile, such as chloroacrylonitrile. In the presence of a strong non-nucleophilic base like 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), the hydrazine moiety acts as a nucleophile, attacking the electron-deficient alkene. This initial addition is followed by an intramolecular cyclization, driven by the oxidation potential of reagents like 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ). The oxidant plays a dual role: it facilitates the aromatization of the newly formed ring system and ensures the complete conversion of the dihydro-intermediate into the stable pyrazolo[1,5-a]pyridine structure (Compound 6). The choice of solvent, typically a polar aprotic medium like acetonitrile, is crucial for solubilizing the ionic intermediates while maintaining the stability of the reactive species. This mechanistic pathway is highly specific, effectively blocking alternative cyclization modes that would lead to the problematic isomers seen in prior art.
Following the construction of the core scaffold, the second phase of the synthesis involves a chemoselective reduction. The intermediate Compound 6 contains a nitrile group and a bromine substituent that must remain intact while a specific double bond or functionality is reduced. The patent specifies the use of zinc powder in conjunction with ammonium chloride in a methanol-water mixture. This reagent system generates nascent hydrogen in situ, which is mild enough to reduce the target functionality without affecting the sensitive bromine atom or the nitrile group. The mechanism likely proceeds via a single-electron transfer process typical of metal-mediated reductions in protic media. The beauty of this step lies in its workup; upon completion, the zinc salts are water-soluble, and the organic product precipitates or can be easily extracted. This eliminates the need for complex aqueous washes to remove heavy metal catalysts often found in hydrogenation reactions, thereby reducing the environmental footprint and simplifying the purification protocol significantly.
How to Synthesize Selpercatinib Intermediate Efficiently
Implementing this synthesis in a GMP environment requires strict adherence to the optimized parameters outlined in the patent examples. The process begins with the preparation of the key hydrazine salt precursor, followed by the oxidative cyclization and final reduction. Operators must monitor reaction progress closely using High Performance Liquid Chromatography (HPLC) to ensure that starting material consumption is complete, typically defined as less than 0.5% residual starting material. The temperature control during the exothermic addition of reagents is critical to maintaining selectivity. For a detailed breakdown of the specific molar ratios, solvent volumes, and isolation techniques that guarantee the reported 99.5% purity, please refer to the standardized operating procedures below.
- React Compound 5 with halogenated acrylonitrile in an organic base (e.g., DBU) and oxidant (e.g., DDQ) to form Compound 6.
- Perform a reduction reaction on Compound 6 using Zinc and ammonium chloride in a protic solvent like methanol.
- Isolate the final high-purity Compound 7 through simple filtration and washing, achieving over 99% purity without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits beyond mere technical superiority. The elimination of complex purification steps directly impacts the cost of goods sold (COGS) by reducing solvent usage, energy consumption, and labor hours. Traditional methods that rely on column chromatography or multiple recrystallizations are not only expensive but also introduce significant variability in batch-to-batch turnaround times. By switching to a process where the product precipitates directly from the reaction mixture or requires only simple extraction, manufacturers can drastically shorten the production cycle. This efficiency gain allows for more flexible scheduling and faster response to market demand fluctuations, which is critical in the fast-paced oncology sector. Furthermore, the robustness of the chemistry reduces the risk of batch failures, ensuring a more consistent and reliable supply of the intermediate for downstream API synthesis.
- Cost Reduction in Manufacturing: The most significant economic advantage of this process is the removal of expensive and time-consuming purification technologies. By avoiding column chromatography and minimizing the need for repeated recrystallization, the facility saves substantially on silica gel, specialized solvents, and filtration equipment. The reagents used, such as zinc powder and DBU, are commodity chemicals with stable pricing, unlike some specialized catalysts that might be subject to supply volatility. Additionally, the high yield of each step means that less raw material is wasted, maximizing the atom economy of the process. These factors combine to lower the overall manufacturing cost per kilogram, providing a competitive edge in pricing negotiations with API customers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the reaction conditions. The process operates at mild temperatures, often between 10°C and 30°C, which reduces the energy load on the facility and minimizes the risk of thermal runaways or safety incidents. The use of readily available starting materials ensures that the supply chain is not dependent on single-source vendors for exotic reagents. Moreover, the solid nature of the intermediates facilitates easier handling, storage, and transportation compared to unstable oils or liquids. This stability allows for the strategic stocking of key intermediates, creating a buffer against potential disruptions in the global logistics network and ensuring continuous production flow.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for industrial scale-up. The avoidance of heavy metal catalysts (other than zinc, which is easily managed) simplifies wastewater treatment and reduces the burden on effluent processing systems. The reduction in solvent volume and the elimination of silica waste from chromatography columns significantly lower the facility's hazardous waste generation. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals. The process has been demonstrated to work effectively on multi-gram scales in the patent examples, and the linear nature of the chemistry suggests that translation to hundred-kilogram or ton-scale reactors can be achieved with minimal engineering modifications, ensuring a smooth path from pilot plant to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in CN113816955B, providing clarity on how this method compares to industry standards. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams assessing supplier capabilities.
Q: How does this new route improve the purity of Selpercatinib Intermediate?
A: The novel pathway described in CN113816955B avoids the formation of difficult-to-separate isomers common in prior art, achieving 99.5% purity directly after workup.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes robust reagents like Zinc and DBU and avoids complex chromatography, making it highly suitable for multi-kilogram to ton-scale manufacturing.
Q: What are the key cost drivers eliminated in this new process?
A: The method eliminates the need for repeated recrystallization and column chromatography, significantly reducing solvent consumption and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selpercatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our technical team has thoroughly analyzed the breakthroughs presented in CN113816955B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot batches to full-scale manufacturing is seamless and compliant with international GMP standards. Our state-of-the-art facilities are designed to handle complex heterocyclic chemistry with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Selpercatinib intermediate meets the highest quality benchmarks required by global regulatory bodies.
We invite pharmaceutical partners and contract research organizations to collaborate with us on optimizing this synthesis for their specific supply chain needs. By leveraging our expertise in process chemistry and scale-up engineering, we can help you realize the full cost and efficiency potential of this novel route. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project volume. We are ready to provide specific COA data, route feasibility assessments, and sample quantities to support your development timelines and secure your position in the competitive RET inhibitor market.
