Advanced Pd/C Catalytic Hydrogenation for Commercial Scale Methoxyphenamine Hydrochloride Production
Advanced Pd/C Catalytic Hydrogenation for Commercial Scale Methoxyphenamine Hydrochloride Production
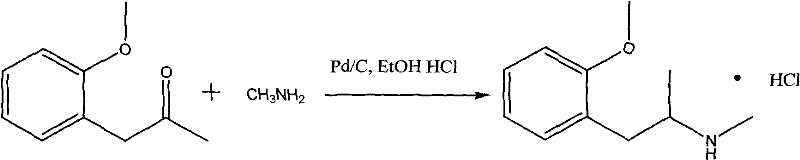
The pharmaceutical industry continuously seeks robust, scalable, and economically viable synthetic routes for critical bronchodilator intermediates. Patent CN101712623A introduces a transformative methodology for the synthesis of Methoxyphenamine Hydrochloride, leveraging palladium on carbon (Pd/C) catalysis to overcome historical limitations in yield and safety. This innovation represents a significant leap forward for manufacturers aiming to optimize their supply chains for respiratory therapeutic agents. By replacing toxic mercury-based systems and costly platinum catalysts with a highly efficient Pd/C system, the patent outlines a pathway that achieves yields exceeding 90% with product purity greater than 99%. For R&D directors and procurement strategists, this technology offers a compelling value proposition: a greener, safer, and more cost-effective manufacturing process that aligns perfectly with modern Good Manufacturing Practice (GMP) standards. The ability to operate under relatively mild hydrogenation conditions further underscores the industrial feasibility of this approach, making it an ideal candidate for large-scale commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Methoxyphenamine Hydrochloride has been plagued by significant technical and economic hurdles that hindered efficient commercial scale-up. Early methodologies, such as those utilizing Mercury-Aluminum amalgams, presented severe environmental and safety liabilities due to the inherent toxicity of mercury, complicating waste disposal and increasing regulatory compliance costs. Alternative routes employing Platinum Oxide or Platinum Dichloride, while effective in laboratory settings, imposed prohibitive raw material costs that eroded profit margins in high-volume manufacturing scenarios. Furthermore, these traditional catalytic systems often required rigorous reaction conditions, including higher pressures or extended reaction times, which increased energy consumption and equipment wear. The presence of heavy metal residues in the final product also necessitated complex purification steps to meet pharmaceutical purity specifications, adding further time and expense to the production cycle. These cumulative inefficiencies created a pressing need for a catalytic system that could balance high performance with economic and environmental sustainability.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this synthesis by utilizing Palladium on Carbon (Pd/C) as the primary catalyst for the reductive amination of o-methoxy phenyl acetone. This method streamlines the reaction workflow by enabling the direct use of methylamine alcohol solutions under mild hydrogenation pressures ranging from 0 to 3 atmospheres. The transition to Pd/C not only drastically reduces catalyst costs compared to platinum group metals but also simplifies the post-reaction workup through straightforward filtration. The process demonstrates exceptional selectivity, minimizing the formation of side products and ensuring a crude product quality that facilitates high-yield recrystallization. By operating at moderate temperatures between 40°C and 70°C, the method reduces thermal stress on the equipment and lowers energy requirements. This strategic shift in catalytic technology effectively resolves the cost and toxicity issues of legacy methods, establishing a new benchmark for efficiency in the production of this vital pharmaceutical intermediate.
Mechanistic Insights into Pd/C-Catalyzed Reductive Amination
The core of this synthetic breakthrough lies in the efficient mechanism of heterogeneous catalytic hydrogenation facilitated by the Pd/C surface. The reaction initiates with the condensation of o-methoxy phenyl acetone and methylamine to form an imine or iminium intermediate in situ within the alcoholic solvent medium. The palladium nanoparticles dispersed on the carbon support provide active sites for the dissociative adsorption of molecular hydrogen, generating reactive atomic hydrogen species. These species subsequently attack the carbon-nitrogen double bond of the imine intermediate, reducing it to the corresponding secondary amine with high stereoselectivity and chemoselectivity. The porous structure of the carbon support ensures excellent mass transfer properties, allowing the reactants to access the catalytic sites rapidly while preventing the aggregation of palladium particles. This mechanistic pathway is crucial for maintaining the integrity of the methoxy group on the aromatic ring, preventing unwanted hydrogenolysis that could compromise the molecular structure. The precise control over hydrogen uptake serves as a real-time indicator of reaction completion, allowing operators to terminate the process exactly when conversion is maximized, thereby preventing over-reduction or degradation.
Impurity control is inherently built into this catalytic system through the specific choice of solvent and reaction parameters. The use of ethanol or methanol as the reaction medium not only solubilizes the organic reactants but also stabilizes the intermediate species, reducing the likelihood of polymerization or oligomerization side reactions. The mild acidic workup involving the addition of hydrochloric acid ethanol solution to adjust the pH to 2 ensures the quantitative conversion of the free amine into its stable hydrochloride salt form. This salt formation step is critical for crystallization, as it allows for the effective exclusion of non-basic impurities and residual starting materials from the crystal lattice. The subsequent recrystallization from ethanol further purifies the solid, leveraging solubility differences to remove trace organic byproducts. This multi-stage purification strategy, driven by the clean nature of the Pd/C catalysis, ensures that the final API intermediate meets the stringent purity requirements necessary for downstream pharmaceutical formulation, minimizing the risk of genotoxic impurities.
How to Synthesize Methoxyphenamine Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to catalyst loading and hydrogen management to ensure optimal safety and yield. The process begins by charging the ketone substrate and methylamine solution into a pressure-rated hydrogenation vessel, followed by the addition of the Pd/C catalyst. Operators must monitor hydrogen pressure and temperature closely, maintaining the system within the preferred 0-3 atm and 40-70°C ranges to maximize reaction kinetics without compromising safety. Once hydrogen absorption ceases, indicating full conversion, the catalyst is removed via filtration, and the solvent is recovered through vacuum distillation. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Charge o-methoxy phenyl acetone and methylamine alcohol solution into a hydrogenation reactor, then add Pd/C catalyst (5-10% Pd content) at 1-10% weight ratio.
- Conduct catalytic hydrogenation at 40-70°C and 0-3 atm pressure until hydrogen uptake ceases, indicating complete reduction of the imine intermediate.
- Filter the catalyst, distill off solvents under vacuum, dissolve residue in ethanol, adjust pH to 2 with hydrochloric acid, and recrystallize to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this Pd/C catalytic route offers substantial strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic reduction of raw material costs associated with catalyst procurement. By substituting expensive platinum-based catalysts with commercially abundant and cheaper palladium on carbon, manufacturers can significantly lower the variable cost per kilogram of the final product. This cost structure improvement provides a competitive edge in pricing negotiations with downstream API manufacturers, allowing for better margin retention or more aggressive market positioning. Additionally, the elimination of mercury-based reagents removes the complex logistical and financial burdens associated with hazardous waste disposal and environmental compliance reporting. This simplification of the regulatory landscape accelerates the time-to-market for new batches and reduces the risk of production stoppages due to environmental audits.
- Cost Reduction in Manufacturing: The economic impact of switching to Pd/C is profound, driven by both lower catalyst acquisition costs and improved process efficiency. The high activity of the palladium catalyst allows for lower catalyst loadings relative to the substrate, further driving down material expenses. Moreover, the mild reaction conditions reduce energy consumption for heating and pressurization, contributing to lower utility costs per batch. The high yield (>90%) minimizes the loss of valuable starting materials, ensuring that nearly all input mass is converted into saleable product. These factors combine to create a leaner manufacturing cost structure that enhances overall profitability and resilience against raw material price fluctuations in the global chemical market.
- Enhanced Supply Chain Reliability: The reliance on widely available reagents such as o-methoxy phenyl acetone, methylamine, and standard Pd/C catalysts ensures a robust and resilient supply chain. Unlike specialized or proprietary catalysts that may have long lead times or single-source dependencies, Pd/C is a commodity chemical available from multiple global suppliers. This diversity of supply sources mitigates the risk of production delays caused by vendor shortages or geopolitical disruptions. Furthermore, the simplicity of the reaction setup means that production can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without requiring specialized equipment modifications. This flexibility guarantees consistent supply continuity for customers relying on this critical intermediate for their own asthma medication production lines.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been demonstrated effectively at the 100 kg scale in the patent examples with clear pathways to tonnage production. The absence of toxic mercury and the use of common organic solvents simplify the waste treatment process, making it easier to meet increasingly strict environmental regulations. The closed-system hydrogenation approach minimizes solvent emissions and exposure risks for plant personnel, aligning with modern occupational health and safety standards. This environmental compatibility not only protects the company's reputation but also future-proofs the manufacturing asset against tightening global environmental laws, ensuring long-term operational viability without the need for costly retrofits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pd/C catalytic synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on process capabilities and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios. The responses highlight the balance between high-performance chemistry and practical industrial application.
Q: What are the primary advantages of using Pd/C over Platinum catalysts for this synthesis?
A: The use of Palladium on Carbon (Pd/C) significantly reduces raw material costs compared to expensive Platinum Oxide or Platinum Dichloride catalysts. Furthermore, Pd/C operates effectively under milder pressures (0-3 atm) and offers easier filtration and recovery, enhancing overall process safety and economic viability.
Q: How does this method address environmental and toxicity concerns associated with older routes?
A: Traditional methods often utilized Mercury-Aluminum amalgams, which pose severe toxicity and disposal challenges. The Pd/C catalytic hydrogenation route described in CN101712623A eliminates heavy metal contamination risks, resulting in a cleaner process with reduced hazardous waste generation and simpler compliance with environmental regulations.
Q: What purity levels can be achieved with the described recrystallization process?
A: By employing a specific workup involving vacuum distillation followed by ethanol recrystallization after pH adjustment, the process consistently achieves product content exceeding 99%. This high purity is critical for pharmaceutical intermediates to meet stringent regulatory standards for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methoxyphenamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for high-value pharmaceutical intermediates like Methoxyphenamine Hydrochloride. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the Pd/C catalytic method are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global regulatory bodies. Our commitment to technological excellence allows us to offer a supply partner relationship that is both reliable and technically sophisticated, capable of adapting to the evolving needs of the respiratory therapy market.
We invite procurement directors and supply chain heads to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized Pd/C process, we can help you achieve significant reductions in total cost of ownership while securing a stable supply of high-quality intermediates. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can become a strategic asset to your supply chain, driving value and efficiency for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
