Advanced Catalytic Hydrogenation for Commercial Scale Methoxyphenamine Hydrochloride Production
Advanced Catalytic Hydrogenation for Commercial Scale Methoxyphenamine Hydrochloride Production
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic viability, particularly for bronchodilator intermediates like Methoxyphenamine Hydrochloride. A pivotal advancement in this domain is documented in Chinese Patent CN101712623B, which discloses a highly efficient method for synthesizing this critical compound via catalytic hydrogenation. Unlike traditional methodologies that rely on toxic mercury amalgams or prohibitively expensive platinum catalysts, this innovation leverages palladium on carbon (Pd/C) to drive the reductive amination of o-methoxyphenylacetone. This technical breakthrough not only streamlines the reaction pathway but also addresses critical environmental and safety concerns associated with legacy processes. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for optimizing supply chains and ensuring the consistent availability of high-quality respiratory therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Methoxyphenamine Hydrochloride has been plagued by significant technical and economic hurdles that hinder large-scale production efficiency. Early literature, such as Japanese Patent JP31010222, described methods utilizing mercury-aluminum amalgam catalysts, which, while effective in yielding the product, introduced severe environmental liabilities due to mercury toxicity and disposal complexities. Furthermore, alternative routes reported by Heinzelman R.V. employed platinum oxide as a catalyst; while chemically effective, platinum group metals represent a substantial capital expenditure, drastically inflating the cost of goods sold (COGS) for the final active pharmaceutical ingredient. These conventional approaches often suffered from moderate yields, typically hovering around 81%, and required rigorous purification steps to remove heavy metal residues, thereby extending production lead times and complicating regulatory compliance for global markets.
The Novel Approach
In stark contrast, the methodology outlined in CN101712623B introduces a paradigm shift by utilizing palladium on carbon as a heterogeneous catalyst, offering a superior balance of activity, selectivity, and cost-effectiveness. This novel approach facilitates the direct reductive amination of o-methoxyphenylacetone with methylamine in an alcoholic solvent system under mild hydrogenation conditions. The transition to Pd/C eliminates the need for toxic mercury reagents and reduces reliance on ultra-expensive platinum variants, thereby fundamentally altering the economic landscape of the synthesis. As illustrated in the reaction scheme below, the process is straightforward yet highly potent, enabling yields exceeding 90% and purity levels greater than 99% after simple recrystallization.
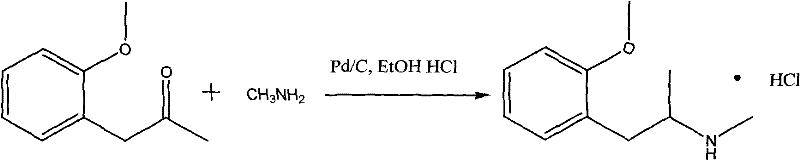
The operational simplicity of this new route cannot be overstated, as it proceeds efficiently at temperatures between 40-70°C and pressures as low as 0-3 atmospheres. This mild condition profile reduces energy consumption and minimizes the formation of unwanted by-products, which is a common issue in harsher reductive amination protocols. By filtering off the solid Pd/C catalyst post-reaction, the downstream processing is significantly simplified, avoiding the complex extraction procedures often required to remove soluble metal contaminants. This results in a cleaner crude product that requires less intensive purification, directly translating to higher throughput and reduced operational expenditures for manufacturing facilities.
Mechanistic Insights into Pd/C Catalyzed Reductive Amination
The core chemical transformation driving this synthesis is a catalytic reductive amination, a powerful tool in organic synthesis for constructing C-N bonds. In this specific mechanism, the carbonyl group of o-methoxyphenylacetone first undergoes nucleophilic attack by methylamine to form an imine or iminium ion intermediate in situ. The presence of the palladium on carbon catalyst is crucial at this stage, as it activates molecular hydrogen, facilitating its addition across the carbon-nitrogen double bond of the transient imine species. This hydrogenation step effectively reduces the imine to the corresponding secondary amine, methoxyphenamine, with high stereoselectivity and minimal over-reduction of the aromatic ring. The heterogeneous nature of the Pd/C catalyst ensures that the reaction occurs on the metal surface, allowing for easy separation of the catalyst from the reaction mixture once hydrogen uptake ceases.
From an impurity control perspective, this mechanism offers distinct advantages over competing pathways. The use of a controlled excess of methylamine (preferably 1.2 to 3 molar equivalents) drives the equilibrium towards imine formation, suppressing the potential for self-condensation of the ketone starting material. Furthermore, the mild acidic workup involving hydrochloric acid in ethanol serves a dual purpose: it converts the free base amine into its stable hydrochloride salt form and helps precipitate the product while leaving soluble organic impurities in the mother liquor. The patent data indicates that this specific combination of catalytic hydrogenation followed by salt formation and recrystallization is highly effective at rejecting structurally related impurities, ensuring the final API intermediate meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Methoxyphenamine Hydrochloride Efficiently
Implementing this synthesis on an industrial scale requires precise adherence to the optimized parameters defined in the patent to maximize yield and safety. The process begins with the preparation of the reaction mixture, where o-methoxyphenylacetone is combined with a methylamine alcohol solution, preferably ethanol or methanol, in a hydrogenation vessel. The concentration of methylamine is a critical variable, with the patent suggesting a range of 5-15% to ensure sufficient reactivity without excessive solvent load. Once the catalyst is added, the system is pressurized with hydrogen and heated to the optimal range of 40-70°C. Detailed standardized operating procedures for scaling this reaction from laboratory to commercial production are provided in the technical guide below.
- Charge o-methoxyphenylacetone and methylamine alcohol solution into a hydrogenation reactor, then add 5-10% Pd/C catalyst.
- Conduct catalytic hydrogenation at 40-70°C under 0-3 atm pressure until hydrogen absorption ceases.
- Filter the catalyst, distill off solvents, adjust pH to 2 with hydrochloric acid ethanol solution, and recrystallize to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Pd/C catalyzed route represents a strategic opportunity to optimize costs and mitigate supply risks associated with Methoxyphenamine Hydrochloride manufacturing. The shift away from mercury-based and platinum-based catalysts addresses two major pain points: regulatory compliance regarding heavy metal residues and the volatility of precious metal prices. By utilizing a more abundant and cost-effective catalyst system, manufacturers can stabilize their raw material costs and reduce the financial exposure linked to fluctuating platinum market rates. Additionally, the simplified workup procedure reduces the consumption of auxiliary chemicals and solvents, contributing to a leaner and more sustainable production model.
- Cost Reduction in Manufacturing: The elimination of expensive platinum oxide and toxic mercury-aluminum amalgam leads to a substantial decrease in direct material costs. Since the Pd/C catalyst is heterogeneous, it can be filtered and potentially regenerated or recovered, further enhancing the economic efficiency of the process. The high yield (>90%) means less starting material is wasted, maximizing the output per batch and lowering the unit cost of the final API intermediate significantly compared to older 81% yield methods.
- Enhanced Supply Chain Reliability: The robustness of this synthesis under mild conditions (low pressure and temperature) makes it highly adaptable to various manufacturing setups, reducing the risk of production bottlenecks caused by equipment limitations. The use of common solvents like ethanol and readily available reagents ensures that the supply chain is not dependent on niche or hard-to-source specialty chemicals. This accessibility translates to shorter lead times and a more resilient supply network capable of meeting sudden spikes in demand for respiratory medications.
- Scalability and Environmental Compliance: From an environmental standpoint, removing mercury from the process flow drastically reduces the burden of hazardous waste treatment and disposal, aligning with modern green chemistry principles and strict environmental regulations. The process generates minimal pollution, as noted in the patent, which simplifies the permitting process for new production lines and reduces long-term environmental liability. The ease of scale-up, demonstrated by the successful execution of 100kg batch sizes in the patent examples, confirms its viability for multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Methoxyphenamine Hydrochloride using the patented Pd/C method. These insights are derived directly from the experimental data and claims presented in CN101712623B, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of using Pd/C over Platinum Oxide for this synthesis?
A: The use of Palladium on Carbon (Pd/C) significantly reduces raw material costs compared to expensive Platinum Oxide catalysts. Furthermore, Pd/C allows for easier filtration and recovery, simplifying the downstream processing and reducing heavy metal contamination risks in the final API.
Q: What purity levels can be achieved with this patented hydrogenation method?
A: According to patent CN101712623B, this method consistently achieves product content exceeding 99% after recrystallization, with isolated yields typically surpassing 90%, making it highly suitable for stringent pharmaceutical quality standards.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is designed for easy industrialization. It operates under mild conditions (0-3 atm pressure, 40-70°C) and utilizes standard hydrogenation equipment, eliminating the need for complex mercury-aluminum amalgam handling or high-pressure specialized reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methoxyphenamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for high-value pharmaceutical intermediates like Methoxyphenamine Hydrochloride. Our technical team has extensively analyzed the Pd/C catalyzed pathway and possesses the expertise to implement this technology at scale, ensuring consistent quality and supply continuity. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, supported by our state-of-the-art facilities and rigorous QC labs. Our commitment to maintaining stringent purity specifications ensures that every batch of Methoxyphenamine Hydrochloride we deliver meets the exacting standards required by global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to leverage these process improvements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your production of bronchodilator APIs remains competitive, compliant, and uninterrupted in an evolving market landscape.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
