Advanced AE-Active Ester Production: A Sustainable Route for Cephalosporin Intermediates
Advanced AE-Active Ester Production: A Sustainable Route for Cephalosporin Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for critical beta-lactam intermediates. Patent CN1709880A introduces a transformative chemical synthesis method for AE-active ester, a pivotal precursor in the manufacture of cephalosporin analog antibiotics. This technology distinguishes itself by integrating a sophisticated byproduct recycling loop directly into the production workflow. Unlike conventional linear syntheses that discard valuable phosphorus and sulfur-containing species, this novel approach employs di(trichloromethyl) carbonate to chemically regenerate key reagents from the reaction waste stream. For R&D directors and procurement strategists, this represents a significant leap forward in process chemistry, offering a pathway to reduce raw material consumption while simultaneously addressing stringent environmental compliance standards regarding hazardous waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of AE-active ester has been plagued by inefficient atom economy and substantial environmental burdens. Traditional routes typically rely on the activation of cefotaxime acid derivatives using coupling agents that generate stoichiometric amounts of difficult-to-remove byproducts. Specifically, the formation of triphenylphosphine oxide and 2-mercaptobenzothiazole is unavoidable in standard activation protocols. In legacy processes, these compounds are often treated as waste residues, necessitating costly incineration or complex purification steps to prevent contamination of the final API. This not only inflates the cost of goods sold (COGS) due to the loss of expensive phosphine reagents but also creates significant logistical challenges in waste management. Furthermore, the accumulation of these impurities can complicate downstream crystallization, potentially compromising the purity profile required for parenteral antibiotic formulations.
The Novel Approach
The methodology disclosed in CN1709880A fundamentally reengineers the reaction landscape by converting waste liabilities into valuable assets. By introducing a secondary reaction stage utilizing di(trichloromethyl) carbonate (triphosgene), the process effectively reverses the degradation of the activating agents. The triphenylphosphine oxide and mercaptobenzothiazole generated in the primary esterification are chemically transformed back into bisbenzothiazole thioether and triphenylphosphine. This closed-loop strategy ensures that the expensive activating components are not consumed but rather circulated within the system. For a reliable pharmaceutical intermediate supplier, this translates to a drastic reduction in the net consumption of raw materials per batch. The process operates under mild conditions, typically between 20°C and 60°C, utilizing common solvents like dichloromethane, which simplifies the engineering requirements for commercial scale-up of complex pharmaceutical intermediates while maintaining a high safety profile.
Mechanistic Insights into Triphenylphosphine-Mediated Activation and Recycling
The core of this synthesis relies on the nucleophilic activation of the carboxylic acid group on cefotaxime acetate. In the presence of a base catalyst such as pyridine or triethylamine, triphenylphosphine reacts with bisbenzothiazole thioether to generate a highly reactive phosphonium intermediate in situ. This activated species facilitates the nucleophilic attack by the carboxylate anion, leading to the formation of the AE-active ester bond. The elegance of this mechanism lies in the leaving group ability of the benzothiazole moiety, which stabilizes the transition state and drives the reaction to completion with high stereochemical integrity. Maintaining the reaction temperature between 20°C and 25°C is critical during this phase to prevent the decomposition of the sensitive beta-lactam ring, ensuring that the final product meets the rigorous high-purity pharmaceutical intermediate specifications demanded by regulatory bodies.
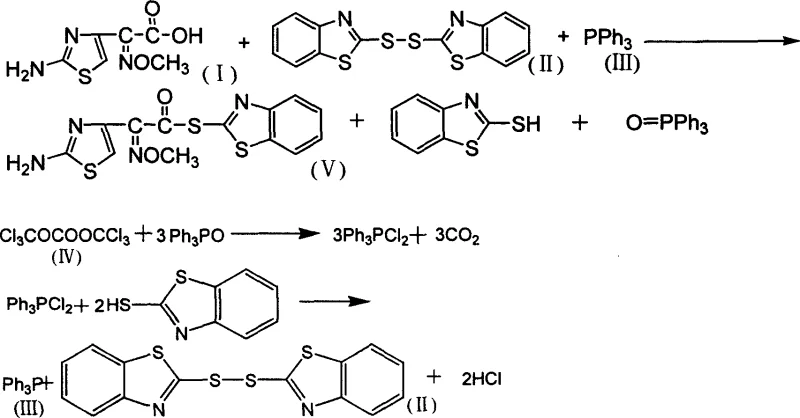
Following the isolation of the target ester, the mechanistic ingenuity of the patent becomes most apparent in the treatment of the mother liquor. The filtrate contains the oxidized phosphine and the thiol byproduct. Upon the addition of di(trichloromethyl) carbonate, a chlorination and coupling sequence occurs. The carbonate acts as a dehydrating and chlorinating agent, facilitating the reformation of the disulfide bond in bisbenzothiazole thioether and the reduction of the phosphine oxide back to triphenylphosphine. This regeneration step is not merely a cleanup operation but a synthetic reconstruction of the reagents. By recovering these materials with yields exceeding 70% in experimental embodiments, the process effectively decouples the cost of the activating system from the volume of production. This mechanism provides a robust solution for cost reduction in pharmaceutical intermediates manufacturing, as the effective lifetime of the phosphine and disulfide reagents is extended indefinitely through continuous recycling.
How to Synthesize AE-Active Ester Efficiently
The operational protocol for this synthesis is designed for reproducibility and ease of handling in a GMP environment. The process begins with the dissolution of cefotaxime acetate and the recycled bisbenzothiazole thioether in an anhydrous organic solvent, preferably dichloromethane, under an inert atmosphere. A catalytic amount of pyridine is introduced to scavenge protons and facilitate the activation cycle. The triphenylphosphine solution is then added dropwise to control the exotherm and ensure uniform mixing. Detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure consistent quality across batches.
- React cefotaxime acetate with bisbenzothiazole thioether and triphenylphosphine in dichloromethane with pyridine catalyst at 20-25°C to form the active ester.
- Filter the reaction mixture to isolate the AE-active ester product, washing the filter cake with methanol and drying under vacuum.
- Treat the filtrate containing byproducts with di(trichloromethyl) carbonate to regenerate bisbenzothiazole thioether and triphenylphosphine for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the implementation of this recycling technology offers profound strategic advantages beyond simple yield improvements. The primary value driver is the substantial mitigation of raw material volatility. Triphenylphosphine and specialized benzothiazole derivatives are subject to market price fluctuations and supply chain disruptions. By internalizing the regeneration of these key inputs, manufacturers can insulate their production costs from external market shocks. This self-sufficiency enhances supply chain reliability, ensuring that production schedules for critical cephalosporin antibiotics are not compromised by the delayed delivery of activating agents. Furthermore, the reduction in waste volume directly correlates to lower disposal fees and reduced environmental compliance overhead, contributing to a leaner and more sustainable operational expenditure model.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of single-use reagent costs. In traditional synthesis, the stoichiometric consumption of triphenylphosphine represents a significant portion of the variable cost. By recovering and reusing over seventy percent of this material, the effective cost per kilogram of the active ester is drastically lowered. Additionally, the valorization of what was previously hazardous waste into usable feedstock removes the financial burden of waste treatment. This structural cost advantage allows for more competitive pricing strategies in the global market for pharmaceutical intermediates, providing a buffer against margin compression.
- Enhanced Supply Chain Reliability: Dependence on external suppliers for every batch of reagents introduces latency and risk. This integrated recycling loop reduces the frequency of raw material orders, thereby simplifying inventory management and reducing working capital requirements. The ability to regenerate critical reagents on-site means that production continuity is less vulnerable to logistics bottlenecks or geopolitical supply constraints. For a reliable pharmaceutical intermediate supplier, this resilience is a key differentiator, guaranteeing consistent delivery performance to downstream API manufacturers even during periods of global supply chain stress.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process aligns perfectly with modern green chemistry principles. The conversion of toxic byproducts into harmless or reusable materials significantly reduces the facility's environmental footprint. This simplifies the permitting process for capacity expansion and minimizes the risk of regulatory penalties associated with hazardous waste discharge. The use of standard solvents like dichloromethane, which are easily recovered and recycled via distillation, further enhances the sustainability profile. This makes the technology ideal for reducing lead time for high-purity pharmaceutical intermediates while adhering to strict international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this AE-active ester synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and benefits. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: How does this process reduce waste compared to traditional AE-active ester synthesis?
A: Traditional methods generate substantial amounts of triphenylphosphine oxide and 2-mercaptobenzothiazole as waste. This patented process utilizes di(trichloromethyl) carbonate to chemically convert these byproducts back into the starting materials (bisbenzothiazole thioether and triphenylphosphine), creating a closed-loop system that drastically minimizes hazardous waste discharge.
Q: What are the optimal reaction conditions for maximizing yield in this synthesis?
A: According to the patent data, the optimal conditions involve using dichloromethane as the solvent and pyridine as the catalyst. The reaction should be maintained at a controlled temperature range of 20°C to 25°C for approximately 3 to 4 hours during the initial activation step, followed by a similar duration for the recycling step, achieving yields exceeding 83%.
Q: Is this synthesis method scalable for industrial production of cephalosporin intermediates?
A: Yes, the process is designed for industrial scalability. It utilizes common organic solvents like dichloromethane and readily available catalysts. The ability to recycle expensive reagents like triphenylphosphine significantly lowers the variable costs per kilogram, making it highly suitable for large-scale commercial manufacturing of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable AE-Active Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with advanced reactor systems capable of handling the precise temperature controls and inert atmospheres required for this sensitive phosphine-mediated chemistry. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of AE-active ester meets the exacting standards necessary for cephalosporin antibiotic synthesis. Our commitment to quality assurance guarantees that the theoretical benefits of this recycling process are fully realized in the final commercial product.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic benefits of this recycling route. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you secure a supply of high-quality intermediates backed by a process that prioritizes both economic efficiency and environmental stewardship.
