Advanced Lithium Halide Catalysis for Scalable Efinaconazole Intermediate Manufacturing
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex antifungal agents, and patent CN107759565B represents a significant technological leap in the production of efinaconazole intermediates. This specific intellectual property discloses a novel preparation method for 1-triazole-2-butanol derivatives that fundamentally alters the economic and operational landscape of manufacturing this critical active pharmaceutical ingredient (API) precursor. By leveraging the unique catalytic properties of lithium or magnesium halides, the process achieves complete reaction conversion and exceptional selectivity, resulting in product purity exceeding 99% through simple crystallization rather than labor-intensive chromatographic purification. For R&D directors and procurement managers alike, this innovation addresses the perennial challenges of yield loss and high processing costs associated with traditional synthetic routes. The ability to utilize cost-effective starting materials like 4-methylenepiperidine hydrochloride while maintaining high yields marks a distinct departure from prior art that often required more expensive salt forms or suffered from poor scalability. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic advancements is crucial for securing a stable and cost-efficient supply chain for downstream antifungal drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of efinaconazole has been plagued by inefficiencies that severely impact both cost and throughput in commercial manufacturing settings. The pioneering method described in WO1994026734, for instance, relies on liberating the piperidine free base using 50% NaOH in diethyl ether, followed by reaction with the chiral epoxide in an ethanol-water solvent system at 85°C. While this established the foundational chemistry, it suffers from significant drawbacks including low overall yields and the generation of numerous impurities that necessitate purification via column chromatography. Furthermore, subsequent improvements like those in CN103080100A showed that while certain salts like hydrobromides offered better yields, the widely available and cheaper hydrochloride salts still resulted in suboptimal conversion rates, particularly when scaling beyond gram-level batches. The absolute requirement for column chromatography in these legacy processes introduces a massive bottleneck, consuming vast quantities of silica gel and organic solvents while drastically extending production lead times. This dependency on chromatographic purification not only inflates the cost of goods sold but also complicates waste management and environmental compliance, making it a less attractive option for large-scale industrial application.
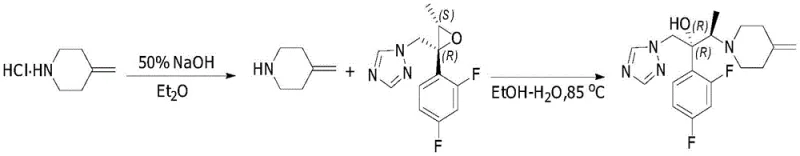
The Novel Approach
In stark contrast, the methodology outlined in CN107759565B introduces a paradigm shift by incorporating lithium or magnesium halides as essential promoters in an acetonitrile solvent system. This new approach enables the direct use of 4-methylenepiperidine acid addition salts, including the economically favorable hydrochloride form, without sacrificing yield or purity. The reaction proceeds efficiently at temperatures between 80°C and 90°C, where the presence of halides like lithium iodide or bromide dramatically accelerates the ring-opening of the epoxide. Crucially, the crude product obtained from this reaction is of such high quality that it bypasses the need for column chromatography entirely, relying instead on a straightforward ethanol-water recrystallization to achieve purity levels greater than 99%. This simplification of the downstream processing workflow represents a major breakthrough in cost reduction in API manufacturing, as it removes one of the most resource-intensive steps from the production line. Moreover, the robustness of this catalytic system ensures consistent performance regardless of the specific acid salt used, providing supply chain teams with greater flexibility in raw material procurement and inventory management.
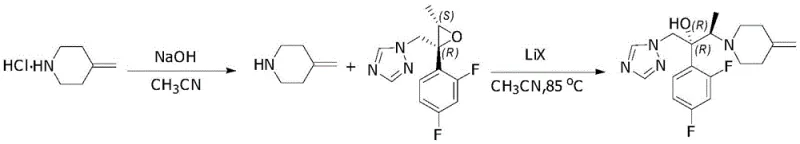
Mechanistic Insights into Lithium Halide-Catalyzed Epoxide Ring-Opening
The core innovation of this patent lies in the specific role played by lithium or magnesium cations in facilitating the nucleophilic attack on the chiral epoxide ring. In the absence of these Lewis acidic metal ions, the nucleophilicity of the piperidine nitrogen is insufficient to drive the reaction to completion efficiently, especially when competing with steric hindrance around the epoxide center. The lithium ion coordinates strongly with the oxygen atom of the oxirane ring, polarizing the C-O bond and rendering the adjacent carbon atoms more electrophilic and susceptible to attack. This coordination effect lowers the activation energy barrier for the ring-opening event, allowing the reaction to proceed rapidly even with less reactive amine salts. Additionally, the specific choice of halide counterions, particularly iodide or bromide, may further enhance the reaction kinetics through soft-soft interactions or by stabilizing transition states, although the primary driver is the Lewis acidity of the metal cation. This mechanistic understanding allows chemists to fine-tune reaction conditions, such as selecting acetonitrile as a solvent which effectively solvates the ionic species without interfering with the catalytic cycle.
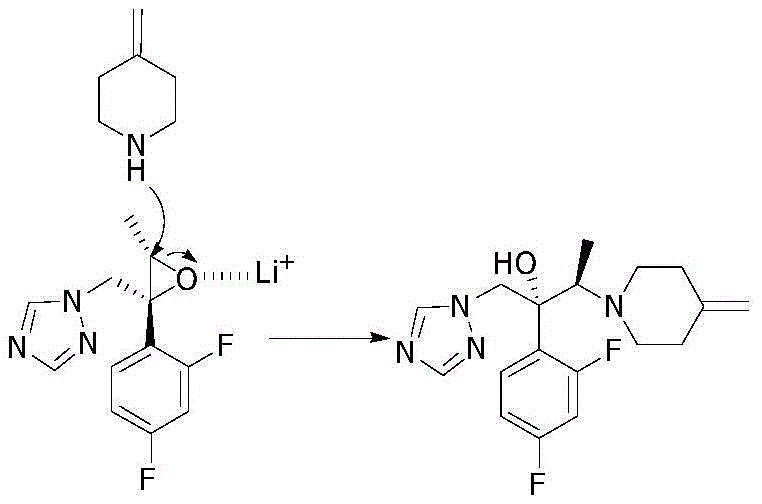
From an impurity control perspective, the lithium halide-mediated pathway offers superior regioselectivity and stereoselectivity compared to base-catalyzed alternatives. Traditional methods often suffer from non-specific ring opening or elimination side reactions that generate difficult-to-remove byproducts, necessitating the aforementioned chromatographic cleanup. By contrast, the directed nature of the metal-coordinated attack ensures that the piperidine moiety attaches at the correct position with retention of the desired stereochemical configuration. This high degree of selectivity minimizes the formation of structural isomers and degradation products, directly contributing to the ability to purify the final product via simple crystallization. For quality assurance teams, this means a cleaner impurity profile and reduced risk of genotoxic impurities carrying over into the final API. The elimination of silica gel chromatography also removes a potential source of metal contamination and particulate matter, further enhancing the overall quality and safety of the pharmaceutical intermediate.
How to Synthesize Efinaconazole Intermediate Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the lithium halide catalysis. The process begins with the in situ generation of the free amine base, followed by the precise addition of the halide promoter and the chiral epoxide substrate under controlled heating conditions. Detailed operational parameters, including specific molar ratios and crystallization protocols, are critical for reproducing the high yields and purity reported in the patent examples. The following guide outlines the standardized steps derived from the patent data to ensure successful technology transfer and scale-up.
- Generate the free base of 4-methylenepiperidine by reacting its acid addition salt with a base such as NaOH in acetonitrile at 25°C.
- Add lithium halide (e.g., LiBr or LiI) and the chiral epoxide substrate to the mixture, then heat to 85-90°C to facilitate ring-opening.
- Perform post-reaction workup involving filtration and solvent removal, followed by ethanol/water recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lithium halide-catalyzed process translates into tangible strategic advantages that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic simplification of the manufacturing workflow, which eliminates the need for expensive chromatographic media and the associated solvent recovery infrastructure. This reduction in processing complexity directly correlates to lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the ability to use generic and widely available 4-methylenepiperidine hydrochloride instead of specialized or expensive salt forms provides significant leverage in raw material negotiations. Suppliers can source starting materials from a broader vendor base without being locked into proprietary or high-cost specifications, thereby enhancing supply security and reducing the risk of shortages. The robustness of the reaction conditions also implies a more forgiving process window, which reduces batch failure rates and ensures consistent delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The elimination of column chromatography is the single most significant cost-saving factor in this new process, as it removes the need for large volumes of silica gel and elution solvents which are both expensive to purchase and dispose of. Additionally, the high yield achieved with inexpensive hydrochloride salts means that raw material costs are minimized compared to methods requiring hydrobromide or hydroiodide salts. The simplified workup procedure also reduces labor hours and energy consumption associated with extended purification steps, leading to a leaner and more cost-effective production model. These cumulative savings allow for a more competitive pricing structure for the final intermediate, benefiting the entire value chain from manufacturer to end-user.
- Enhanced Supply Chain Reliability: By decoupling the process performance from specific, hard-to-source acid salts, manufacturers gain substantial flexibility in their supply chain logistics. The method's tolerance for various salt forms means that if one supplier of 4-methylenepiperidine hydrochloride faces disruption, alternative sources can be qualified quickly without revalidating the entire synthetic route. This resilience is critical for maintaining continuous production schedules and meeting the rigorous delivery commitments expected by global pharmaceutical clients. Moreover, the use of common solvents like acetonitrile and ethanol ensures that solvent supply risks are minimized, as these are commodity chemicals with stable global availability.
- Scalability and Environmental Compliance: Transitioning from batch chromatography to crystallization-based purification is a key enabler for commercial scale-up, as crystallization is inherently easier to control and automate in large reactors. This scalability ensures that the process can meet increasing market demand for efinaconazole without the bottlenecks typically associated with preparative chromatography. From an environmental standpoint, the reduction in solvent usage and the elimination of silica gel waste significantly lower the E-factor of the process, facilitating compliance with increasingly stringent environmental regulations. This sustainability profile is becoming a key differentiator for suppliers seeking to partner with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These answers are derived directly from the experimental data and claims presented in patent CN107759565B, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating technology transfer and procurement officers assessing supplier capabilities.
Q: Why are lithium halides critical for this epoxide ring-opening reaction?
A: Lithium ions coordinate with the epoxide oxygen, significantly lowering the activation energy for nucleophilic attack by the piperidine nitrogen, thereby accelerating reaction rates and improving selectivity compared to traditional base-only methods.
Q: Can this method achieve high purity without column chromatography?
A: Yes, the patent demonstrates that using lithium halides in acetonitrile allows for simple ethanol/water recrystallization to achieve purity levels greater than 99%, completely eliminating the costly and time-consuming column chromatography step required in older methods.
Q: Is the process sensitive to the specific acid salt of the piperidine starting material?
A: No, the method is highly robust and works effectively with various acid addition salts including hydrochloride, hydrobromide, and hydroiodide, providing significant flexibility in raw material sourcing and cost optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Efinaconazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes like the one described in CN107759565B requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this lithium halide-catalyzed route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the absence of chromatographic impurities and confirm stereochemical integrity. Our commitment to quality ensures that every batch of efinaconazole intermediate meets the exacting standards required for final API synthesis, providing our partners with peace of mind and regulatory confidence.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your volume requirements and current sourcing strategy. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and competitiveness in your antifungal drug portfolio.
