Advanced Manufacturing of Efinaconazole Intermediates via Purified Amine Salt Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antifungal agents, and Patent CN103080100A presents a transformative approach to synthesizing Efinaconazole intermediates. This specific intellectual property addresses the longstanding challenges associated with the ring-opening addition of amines to epoxytriazoles, a key step in constructing the complex molecular architecture of this active pharmaceutical ingredient. By shifting from traditional aqueous amine solutions to the utilization of purified 4-methylenepiperidine acid addition salts, the disclosed methodology achieves high yields under significantly milder reaction conditions. This technical advancement is not merely a laboratory curiosity but represents a scalable solution for the commercial production of high-purity pharmaceutical intermediates, directly impacting the reliability of the global supply chain for antifungal medications.
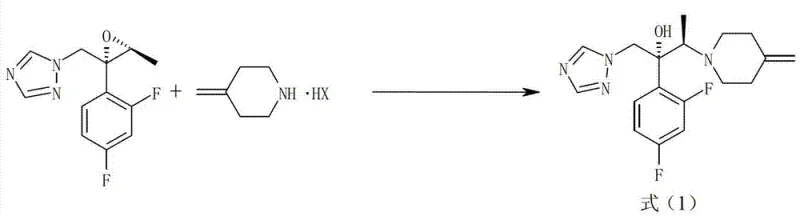
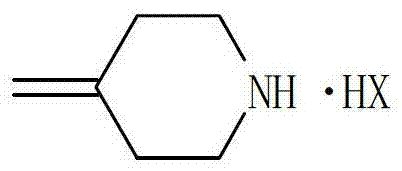
The core innovation lies in the strategic manipulation of the amine reagent's physical state and purity profile prior to the coupling reaction. Conventional wisdom often dictated the use of free amines or their aqueous solutions, which introduced significant variability due to water content and thermal instability. The patent elucidates a process where the amine is first converted into a stable solid salt, such as the hydrobromide or hydroiodide salt, which can be rigorously purified via crystallization. This pre-purification step effectively removes heat-generated impurities that typically plague the final drug substance, thereby enhancing the overall impurity profile of the Efinaconazole intermediate and ensuring compliance with stringent regulatory standards for pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminoalcohols via the ring-opening addition of amines to epoxides has been fraught with inefficiencies that hinder industrial scalability. Traditional protocols frequently necessitate the use of a large excess of the amine reagent, often in the form of an aqueous solution, to drive the reaction to completion. This requirement creates a substantial burden on downstream processing, as the recovery and recycling of the unreacted amine become mandatory yet costly operations. Furthermore, the reliance on aqueous amine solutions introduces water into the reaction system, which can interfere with reaction kinetics and complicate solvent removal. Perhaps most critically, the free amine, particularly 4-methylenepiperidine, is susceptible to thermal degradation during distillation processes used for purification, leading to the formation of difficult-to-remove impurities that compromise the quality of the final API.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages the unique properties of 4-methylenepiperidine acid addition salts to overcome these thermodynamic and kinetic barriers. By employing the amine in its salt form, manufacturers can utilize a high-purity solid reagent that is stable at room temperature and free from the thermal degradation products associated with distilling the free base. The reaction proceeds efficiently with only a stoichiometric amount of the amine salt, typically between 1 to 1.5 equivalents relative to the epoxytriazole, eliminating the need for massive excesses. This shift not only simplifies the reaction workup by removing the amine recovery step but also ensures that the nucleophile is introduced into the system in a controlled manner, facilitated by the in situ generation of the free amine via a mild base.
Mechanistic Insights into Base-Promoted Epoxide Ring-Opening
The mechanistic elegance of this process relies on the precise interplay between the acid addition salt and the selected metal hydroxide base within the reaction solvent. When the 4-methylenepiperidine acid addition salt is mixed with a base such as lithium hydroxide or calcium hydroxide, an acid-base neutralization occurs that liberates the free amine nucleophile in situ. This generated free amine then attacks the electrophilic carbon of the epoxytriazole ring, specifically targeting the less hindered position to open the epoxide and form the desired beta-amino alcohol structure. The presence of the base serves a dual purpose: it activates the nucleophile by deprotonating the ammonium salt and simultaneously neutralizes the acid byproduct (HX) released during the ring-opening event, driving the equilibrium forward without requiring harsh conditions or excessive reagent loading.
From an impurity control perspective, this mechanism offers superior selectivity compared to direct heating of free amines. The controlled release of the amine prevents localized high concentrations that could lead to polymerization or side reactions with the sensitive triazole moiety. Furthermore, the use of specific solvents like acetonitrile or cyclopentyl methyl ether provides an optimal dielectric environment that stabilizes the transition state of the nucleophilic attack. The result is a reaction profile that minimizes the formation of regioisomers and over-alkylation byproducts, which are common pitfalls in epoxide chemistry. This high level of control is essential for maintaining the stereochemical integrity of the (2R,3R) configuration required for the biological activity of the final antifungal agent.
How to Synthesize Efinaconazole Efficiently
The implementation of this synthesis route requires careful attention to the preparation of the amine salt and the selection of the base-solvent system to maximize yield and purity. The process begins with the conversion of crude 4-methylenepiperidine into a crystalline salt, which acts as a purification checkpoint before the critical coupling step. Following this, the reaction is conducted under reflux conditions in a polar aprotic or alcoholic solvent, allowing for efficient heat transfer and mixing. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction mixture by combining 4-methylenepiperidine acid addition salt (e.g., hydrobromide) and an alkali metal hydroxide (e.g., LiOH) in a suitable solvent like acetonitrile.
- Add the epoxytriazole starting material, (2R,3S)-2-(2,4-difluorophenyl)-3-methyl-2-[(1H-1,2,4-triazol-1-yl)methyl]oxirane, to the stirred mixture under controlled temperature conditions.
- Heat the reaction to reflux (e.g., 100°C) for approximately 14 hours, then isolate the product through crystallization or extraction followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation strategies. The ability to source stable, solid amine salts rather than volatile aqueous amine solutions significantly reduces the hazards associated with transportation and storage, thereby lowering insurance and logistics costs. Moreover, the elimination of the amine recovery unit operation simplifies the plant infrastructure requirements, allowing for faster batch turnover and reduced utility consumption. This streamlined process flow enhances the overall agility of the manufacturing site, enabling it to respond more rapidly to fluctuations in market demand for antifungal intermediates without the bottleneck of complex solvent and reagent recycling loops.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in raw material waste and the simplification of the purification train. By avoiding the use of large excesses of expensive amine reagents, the direct material cost per kilogram of product is significantly lowered. Additionally, the removal of the amine recovery step eliminates the energy-intensive distillation processes traditionally required to separate and recycle unreacted amine, leading to substantial savings in steam and electricity usage. The high purity of the starting salt also reduces the load on final crystallization steps, minimizing solvent usage and increasing the overall throughput of the production facility.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of solid acid addition salts, which possess superior shelf-life stability compared to their liquid counterparts. This stability reduces the risk of reagent degradation during long-term storage or international shipping, ensuring consistent quality upon arrival at the manufacturing site. Furthermore, the robustness of the reaction conditions allows for the use of a wider range of commercially available solvents, reducing dependency on single-source specialty chemicals. This flexibility empowers procurement teams to negotiate better terms with multiple vendors and mitigates the risk of supply disruptions caused by raw material shortages.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns perfectly with green chemistry principles by minimizing waste generation and hazardous emissions. The stoichiometric use of reagents means less chemical waste requires treatment or disposal, lowering the environmental footprint of the manufacturing process. The mild reaction conditions and the absence of toxic Lewis acids or perchlorates further enhance the safety profile of the operation, facilitating easier regulatory approval for commercial scale-up. This compliance advantage accelerates the timeline from pilot plant validation to full-scale commercial production, ensuring a steady supply of high-quality intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and background analysis of the patent literature. Understanding these nuances is critical for R&D teams planning to integrate this technology into their existing manufacturing workflows. The answers provided reflect the specific conditions and findings documented in the intellectual property to ensure accurate technical guidance.
Q: Why is the acid addition salt of 4-methylenepiperidine preferred over the free amine?
A: Using the acid addition salt allows for the removal of impurities generated during the distillation of the free amine. It enables the use of high-purity solid reagents instead of unstable aqueous solutions, significantly reducing by-product formation.
Q: What bases are suitable for this ring-opening reaction?
A: The process utilizes alkali metal or alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, calcium hydroxide, or strontium hydroxide. Lithium hydroxide is particularly preferred for optimal yield and reaction kinetics.
Q: Does this method require a large excess of the amine reagent?
A: No, unlike conventional methods that require large excesses of amine to drive the reaction, this novel approach operates efficiently with stoichiometric amounts (1 to 1.5 equivalents), simplifying downstream processing and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Efinaconazole Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this patented laboratory methodology into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot batches to industrial volumes is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch of Efinaconazole intermediate we produce.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this salt-based methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our commitment to delivering high-value pharmaceutical intermediates with unmatched reliability and quality assurance.
