Advanced Green Synthesis of Diphenylacetylene Compounds for Commercial Scale-up
The global demand for high-performance liquid crystal materials continues to surge, driving the need for more sustainable and efficient synthetic routes for key intermediates like diphenylacetylene compounds. Patent CN101811918A introduces a groundbreaking methodology that leverages deep eutectic ionic liquids to facilitate the Sonogashira coupling reaction under remarkably mild conditions. This technological advancement addresses critical pain points in traditional manufacturing, such as high energy consumption and the reliance on volatile organic solvents, offering a pathway to greener chemical production. By utilizing a quaternary ammonium salt-imidazole system, this process not only achieves high yields but also simplifies downstream processing, making it an attractive option for large-scale industrial adoption. For R&D and procurement leaders, understanding this shift is vital for maintaining competitiveness in the display and optoelectronic materials sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of diphenylacetylene derivatives relies heavily on the Sonogashira reaction conducted in volatile organic solvents such as toluene, tetrahydrofuran (THF), or dimethylformamide (DMF). These conventional processes typically necessitate elevated reaction temperatures to overcome activation energy barriers, which significantly increases energy costs and poses safety risks associated with handling flammable solvents at high heat. Furthermore, the removal of these solvents post-reaction requires energy-intensive distillation processes, and the disposal of solvent waste creates substantial environmental liabilities. The reliance on homogeneous catalysis in these systems often leads to difficulties in catalyst recovery, resulting in the loss of precious palladium metals and increased contamination of the final product with heavy metal residues. These factors collectively contribute to a higher cost of goods sold and a larger carbon footprint, which are increasingly scrutinized by modern supply chain auditors and regulatory bodies.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a quaternary ammonium salt-imidazole deep eutectic ionic liquid as both the solvent and a promoter for the reaction. This system allows the coupling of iodoarenes and arylacetylenes to proceed efficiently at temperatures ranging from 20-60°C, drastically reducing thermal energy requirements. The unique physicochemical properties of the ionic liquid stabilize the catalytic species, enabling the reaction to reach completion within 4-12 hours with yields consistently between 72% and 80%. This method eliminates the need for hazardous volatile organic compounds, thereby simplifying the work-up procedure to simple water washing and extraction. The ability to operate at near-room temperature not only enhances safety but also preserves the integrity of temperature-sensitive functional groups on the aromatic rings, expanding the scope of synthesizable derivatives for advanced material applications.
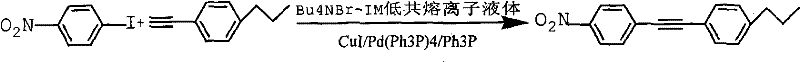
Mechanistic Insights into CuI/Pd-Catalyzed Coupling in Ionic Media
The core of this technological breakthrough lies in the synergistic interaction between the palladium-copper catalytic cycle and the deep eutectic solvent environment. The reaction initiates with the oxidative addition of the aryl iodide to the zero-valent palladium species, forming an aryl-palladium(II) intermediate. The presence of the ionic liquid likely facilitates the stabilization of this charged intermediate through electrostatic interactions, lowering the activation energy for subsequent steps. Copper(I) iodide acts as a co-catalyst, activating the terminal alkyne to form a copper-acetylide species, which then undergoes transmetallation with the palladium complex. This step is crucial for forming the carbon-carbon triple bond that characterizes the diphenylacetylene structure. Finally, reductive elimination releases the desired product and regenerates the active palladium catalyst, ready for another cycle. The ionic medium plays a pivotal role in maintaining the solubility of these catalytic species while preventing their aggregation into inactive palladium black.
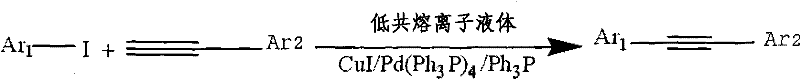
Impurity control is inherently improved in this system due to the selective solvation properties of the deep eutectic ionic liquid. Unlike traditional solvents that may dissolve a wide range of side products, the ionic phase can be tuned to preferentially solvate the catalyst and reactants while allowing the product to separate more easily upon quenching. The mild reaction conditions minimize thermal degradation pathways that often lead to complex impurity profiles in high-temperature syntheses. Additionally, the absence of protic impurities in the dried ionic liquid prevents premature protonation of the acetylide intermediate, ensuring high chemoselectivity. For quality control teams, this translates to a cleaner crude product profile, reducing the burden on purification columns and crystallization steps. The result is a final API intermediate or electronic chemical with superior purity specifications, meeting the stringent requirements of downstream liquid crystal manufacturers without extensive reprocessing.
How to Synthesize Diphenylacetylene Efficiently
To implement this synthesis effectively, operators must first prepare the deep eutectic solvent by precisely mixing quaternary ammonium salts with imidazole under controlled heating and vacuum drying conditions to remove moisture. The reaction is then set up under an inert nitrogen atmosphere to prevent oxidation of the sensitive phosphine ligands and palladium catalyst. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the deep eutectic ionic liquid by mixing quaternary ammonium salts and imidazole at a molar ratio of 1: (2-3), heating until molten, and drying under vacuum.
- Combine iodoarenes and arylacetylenes in the ionic liquid with CuI/Pd(Ph3P)4/Ph3P catalyst under nitrogen protection.
- React at mild temperatures between 20-60°C for 4-12 hours, then separate the product via water washing and organic extraction.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology offers substantial opportunities for cost optimization and supply chain resilience. The transition to deep eutectic ionic liquids removes the dependency on fluctuating markets for volatile organic solvents, which are subject to strict environmental regulations and price volatility. By operating at lower temperatures, manufacturing facilities can significantly reduce their energy consumption, leading to direct utility cost savings over the lifespan of the production campaign. The recyclability of the catalyst system further diminishes the requirement for fresh palladium inputs, which are among the most expensive raw materials in fine chemical synthesis. These efficiencies combine to create a more robust cost structure that can withstand market pressures while maintaining healthy margins for suppliers and buyers alike.
- Cost Reduction in Manufacturing: The elimination of volatile organic solvents fundamentally alters the cost structure by removing the need for complex solvent recovery distillation columns and associated energy inputs. Furthermore, the ability to recycle the catalyst system reduces the consumption of high-value palladium reagents, leading to substantial raw material savings. The simplified work-up procedure involving water washing instead of extensive chromatographic purification lowers labor and consumable costs significantly. Overall, these process intensifications drive down the total cost of ownership for the manufacturing process without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing widely available quaternary ammonium salts and imidazole as solvent components mitigates the risk of supply disruptions associated with specialized petrochemical solvents. The mild reaction conditions reduce the safety hazards associated with high-pressure and high-temperature operations, minimizing the risk of unplanned plant shutdowns due to safety incidents. This operational stability ensures consistent delivery schedules for downstream clients who rely on just-in-time inventory models for their display panel production lines. A more stable process inherently leads to a more reliable supply chain partner.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with increasingly stringent global environmental regulations regarding VOC emissions and heavy metal waste. Scaling this process does not require exponential increases in waste treatment capacity, as the ionic liquid can be reused multiple times. This scalability ensures that production can be ramped up to meet surging demand for electronic materials without facing regulatory bottlenecks. Companies adopting this technology position themselves as leaders in sustainable manufacturing, enhancing their brand reputation among environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid-based synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide clarity for potential adopters.
Q: What are the primary advantages of using deep eutectic ionic liquids over traditional solvents?
A: Deep eutectic ionic liquids significantly reduce reaction temperatures to 20-60°C compared to traditional high-temperature methods, minimize volatile organic compound emissions, and allow for catalyst recycling, thereby enhancing environmental compliance and operational safety.
Q: Can the catalyst system be reused in this synthesis method?
A: Yes, the patent data indicates that the CuI/Pd(Ph3P)4/Ph3P catalyst system within the ionic liquid medium exhibits excellent stability and can be recycled, which reduces the consumption of expensive palladium resources and lowers overall material costs.
Q: What is the typical yield range for this green synthesis process?
A: Experimental data from the patent demonstrates consistent yields ranging from 72% to 80% across various substituted diphenylacetylene derivatives, indicating a robust and reliable process suitable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenylacetylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green synthesis technologies like the one described in CN101811918A for the production of high-value electronic chemicals. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications required for liquid crystal intermediates, guaranteeing that every batch performs consistently in your final applications. We are committed to leveraging such advanced methodologies to deliver superior value to our global clientele.
We invite you to collaborate with us to optimize your supply chain for diphenylacetylene derivatives and related fine chemicals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a sustainable and cost-effective source for your critical raw materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
