Advanced Manufacturing of Entecavir Intermediates via Optimized Amino Protection and Oxidation Strategies
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiviral agents, and the recent disclosure in patent CN111732589A offers a transformative approach to the manufacturing of Entecavir, a potent nucleoside analogue used in the treatment of chronic hepatitis B. This technical breakthrough addresses longstanding inefficiencies in the synthesis of key intermediates, specifically focusing on the amino protection and oxidation stages which have historically plagued production lines with extended cycle times and suboptimal yields. By introducing a strategic batch-feeding protocol for the amino protecting group and rigorously optimizing reaction stoichiometry, the new methodology reduces the synthesis time for the amino-protected intermediate from over 10 hours to merely 50-70 minutes. This drastic reduction in processing time not only enhances throughput but also minimizes the exposure of sensitive intermediates to potentially degrading conditions, thereby securing a higher quality profile for the downstream API. For R&D directors and process chemists, this represents a significant leap forward in process intensification, offering a pathway to more sustainable and cost-effective manufacturing of high-purity pharmaceutical intermediates.
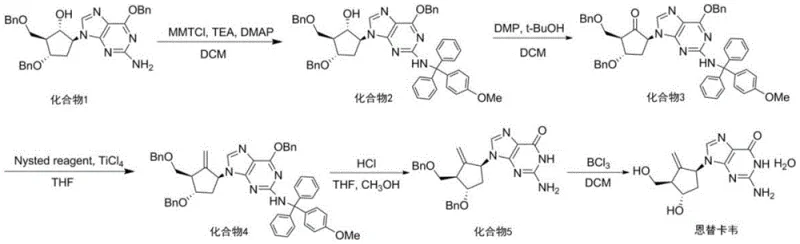
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Entecavir intermediates have long been hindered by kinetic inefficiencies and thermodynamic instabilities that compromise overall process economics. In the conventional synthesis of the amino-protected Compound 2, the standard practice involves a one-time addition of all reactants, including the bulky 4-methoxytriphenylchloromethane protecting group. This bulk addition often leads to localized high concentrations that can trigger exothermic spikes and promote side reactions, resulting in a prolonged reaction time exceeding 10 hours to reach completion. Furthermore, the subsequent oxidation step to generate Compound 3 is notoriously susceptible to keto-enol tautomerism, a phenomenon where the desired ketone product equilibrates with an enol form, leading to difficult-to-remove impurities and a significant drop in isolated yield. These bottlenecks create substantial challenges for supply chain managers, as the extended reactor occupancy times limit production capacity and increase the cost of goods sold due to higher solvent and energy consumption.
The Novel Approach
The innovative process detailed in the patent fundamentally re-engineers the reaction kinetics by implementing a multi-batch feeding strategy for the amino protection step. Instead of a single charge, the 4-methoxytriphenylchloromethane is introduced in five distinct portions over a controlled timeframe, maintaining an optimal concentration gradient that drives the reaction forward efficiently while mitigating thermal risks. This adjustment, coupled with a precisely tuned molar ratio of Compound 1 to protecting agent (1.0:1.4), allows the reaction to reach completion in just 60 minutes at mild temperatures of 20-30°C. Additionally, the oxidation stage is refined by controlling the post-reaction treatment temperature between 0-10°C, which effectively suppresses the keto-enol tautomerism that plagues traditional methods. The result is a streamlined, high-yielding pathway that delivers intermediates with superior purity profiles, setting a new benchmark for the commercial scale-up of complex antiviral intermediates.
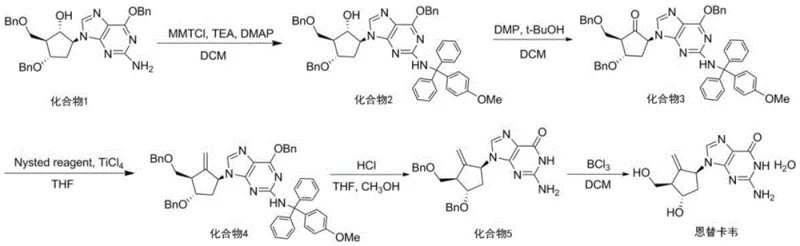
Mechanistic Insights into Optimized Amino Protection and Oxidation
The core of this technological advancement lies in the precise manipulation of reaction kinetics during the amino protection phase. By utilizing 4-dimethylaminopyridine (DMAP) as a nucleophilic catalyst alongside triethylamine, the process facilitates the formation of the trityl ether linkage with high specificity. The batch-feeding mechanism ensures that the concentration of the electrophilic chloromethane species remains low enough to prevent oligomerization or decomposition, yet high enough to maintain a favorable reaction rate. This balance is critical for preserving the stereochemical integrity of the cyclopentene ring system, which is essential for the biological activity of the final Entecavir molecule. The optimization of the molar ratio to 1.0:1.4:1.5:0.08 (Compound 1:MMTCl:TEA:DMAP) represents a sweet spot where reagent waste is minimized without sacrificing conversion efficiency, demonstrating a deep understanding of the underlying mechanistic requirements for high-fidelity synthesis.
Furthermore, the control of impurity profiles during the oxidation and methylenation steps is achieved through rigorous thermal management and stoichiometric precision. The use of Dess-Martin periodinane (DMP) for oxidation is well-known for its mildness, but the patent highlights that the workup conditions are equally vital; cooling the system to 0-10°C before quenching prevents the acid-catalyzed tautomerization that typically degrades product quality. In the subsequent methylenation step using the Nysted reagent and titanium tetrachloride, the molar ratios are tightly controlled (1:5.0:5.0) to ensure complete conversion of the ketone to the exocyclic methylene group without over-reacting or generating titanium-based sludge that complicates filtration. This holistic approach to impurity control ensures that the final deprotection steps using boron trichloride proceed cleanly, yielding Entecavir with a purity exceeding 99% after simple recrystallization.
How to Synthesize Entecavir Intermediates Efficiently
The synthesis of high-purity Entecavir intermediates requires a disciplined adherence to the optimized parameters regarding reagent addition and temperature control outlined in the patent data. The process begins with the preparation of the amino-protected scaffold, which serves as the foundation for the entire synthetic sequence, necessitating strict monitoring of TLC to determine the exact endpoint of the 60-minute reaction window. Following isolation, the oxidation and methylenation steps must be performed under inert atmosphere conditions to prevent moisture sensitivity issues associated with the titanium and boron reagents. The detailed standardized synthesis steps below provide a roadmap for replicating this high-efficiency pathway in a GMP-compliant environment, ensuring consistent quality and yield for commercial production runs.
- Perform amino protection on Compound 1 using 4-methoxytriphenylchloromethane added in multiple batches over 50-70 minutes to maximize yield.
- Execute oxidation using Dess-Martin periodinane with strict temperature control (0-10°C) to prevent keto-enol tautomerism.
- Complete methylenation with Nysted reagent and TiCl4, followed by sequential deprotection using HCl and BCl3 to obtain the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this improved synthesis process translates directly into enhanced operational agility and reduced manufacturing costs. The drastic reduction in reaction time for the amino protection step—from over 10 hours down to approximately one hour—significantly increases the asset utilization rate of existing reactor infrastructure, allowing for more batches to be produced within the same timeframe without requiring capital expenditure on new equipment. This efficiency gain is compounded by the improved yield and purity profiles, which reduce the volume of raw materials required per kilogram of final API and minimize the need for extensive purification processes such as repeated chromatography or recrystallization. Consequently, the overall cost of goods sold is lowered, providing a competitive edge in the pricing of generic antiviral medications while maintaining robust profit margins.
- Cost Reduction in Manufacturing: The elimination of extended reaction times and the optimization of reagent stoichiometry lead to substantial savings in energy consumption and solvent usage. By preventing the formation of difficult-to-remove isomers during the oxidation step, the process reduces the load on downstream purification units, lowering the cost associated with chromatographic media and waste disposal. This lean manufacturing approach ensures that the production of high-purity pharmaceutical intermediates remains economically viable even in fluctuating market conditions.
- Enhanced Supply Chain Reliability: The robustness of the new synthetic route, characterized by its tolerance to minor variations and high reproducibility, ensures a steady and reliable supply of critical intermediates. The simplified workflow reduces the risk of batch failures and production delays, which are common pain points in complex nucleoside synthesis. This reliability is crucial for maintaining continuity in the supply of hepatitis B treatments, safeguarding against shortages that could impact patient access to life-saving medications.
- Scalability and Environmental Compliance: The controlled addition of reagents and the mitigation of exothermic events make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduction in reaction time and improved selectivity also contribute to a smaller environmental footprint by decreasing the generation of chemical waste and hazardous byproducts. This aligns with global sustainability goals and regulatory pressures for greener pharmaceutical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this improved Entecavir synthesis process, derived directly from the experimental data and claims of the patent. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or process optimization projects. The answers provided reflect the specific advantages gained through the novel batch-feeding and temperature control strategies.
Q: How does the batch-feeding method improve the amino protection reaction?
A: By adding the protecting group in five portions rather than all at once, the reaction time is reduced from over 10 hours to approximately 60 minutes, significantly improving yield and preventing side reactions caused by high local concentrations.
Q: What specific measures prevent keto-enol tautomerism during oxidation?
A: The process strictly controls the post-reaction treatment temperature at 0-10°C and optimizes the concentration and duration of the reaction, effectively suppressing isomer formation and ensuring high purity of the oxidation product.
Q: What is the achieved purity of the final Entecavir product using this method?
A: Through the optimization of raw material ratios in the methylenation and deprotection steps, combined with recrystallization, the final Entecavir product achieves a purity exceeding 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the optimized Entecavir synthesis described in patent CN111732589A can be seamlessly translated into industrial reality. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to technical excellence ensures that every batch we produce adheres to the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage these advanced manufacturing techniques for your supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized processes can drive value and efficiency in your antiviral drug production portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
