Advanced Synthesis of 2-Chloro-3-Amino-4-Methylpyridine for Commercial API Manufacturing
Advanced Synthesis of 2-Chloro-3-Amino-4-Methylpyridine for Commercial API Manufacturing
Introduction to Patent CN100358871C and Strategic Value
The global pharmaceutical landscape continuously demands more efficient and sustainable pathways for producing critical antiretroviral intermediates, specifically for drugs like Nevirapine which remain essential in HIV treatment protocols. Patent CN100358871C introduces a transformative synthetic methodology for 2-chloro-3-amino-4-methylpyridine, utilizing cyanoacetamide and acetone as primary starting materials to achieve superior selectivity and yield. This technical breakthrough addresses long-standing inefficiencies in heterocyclic chemistry by streamlining the reaction sequence into four distinct, high-yielding stages that eliminate the need for hazardous nitration or expensive noble metal catalysts. For R&D Directors and Supply Chain Heads, this patent represents a viable route to secure high-purity pharmaceutical intermediates with reduced environmental impact and optimized cost structures. The process demonstrates a robust overall yield of approximately 37% with final product purity exceeding 99%, establishing a new benchmark for industrial feasibility in the synthesis of complex pyridine derivatives. By leveraging this proprietary knowledge, manufacturers can significantly enhance their supply chain resilience while meeting the rigorous quality standards required for global regulatory compliance in the antiretroviral sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-3-amino-4-methylpyridine has been plagued by complex multi-step routes that suffer from poor selectivity and excessive waste generation, creating substantial bottlenecks for commercial procurement teams. Prior art methods, such as those described in US6399781, rely on expensive starting materials like 2-chloro-3-nitro-4-picoline, which drastically inflate the cost of goods sold and limit scalability for large-volume production. Other documented approaches involve harsh nitration and chlorination sequences that generate significant quantities of waste acid and wastewater, posing severe environmental compliance challenges and increasing disposal costs for manufacturing facilities. Furthermore, routes utilizing palladium-catalyzed hydrogenation, as seen in US5668287, introduce dependency on precious metals that are subject to volatile market pricing and require complex removal steps to meet residual metal specifications in API intermediates. These conventional pathways often result in low overall yields and difficult purification processes, making them economically unviable for competitive generic drug manufacturing where margin compression is a constant pressure. The cumulative effect of these technical deficiencies is a supply chain that is fragile, expensive, and environmentally unsustainable, necessitating a paradigm shift towards more atom-economical and operationally simple synthetic strategies.
The Novel Approach
The innovative methodology outlined in Patent CN100358871C circumvents these historical constraints by employing a condensation-cyclization-degradation strategy that utilizes readily available and cost-effective raw materials like cyanoacetamide and acetone. This novel approach simplifies the synthetic tree by avoiding hazardous nitration steps entirely, thereby reducing the safety risks associated with handling energetic intermediates and minimizing the generation of toxic byproducts. The reaction conditions are notably mild, operating at manageable temperatures and pressures that do not require specialized high-pressure equipment, thus lowering capital expenditure requirements for facility upgrades. By replacing expensive palladium catalysts with common organic bases and halogens, the process achieves a drastic reduction in raw material costs while simplifying the downstream purification workflow. The selectivity of the reaction is inherently high, leading to fewer side products and a cleaner crude profile that facilitates easier isolation of the target molecule. This strategic redesign of the synthetic route not only improves the economic metrics of production but also aligns with modern green chemistry principles, offering a compelling value proposition for procurement managers seeking to optimize their supply chain for both cost and sustainability.
Mechanistic Insights into Cyanoacetamide Condensation and Cyclization
The initial phase of this synthesis involves the condensation of cyanoacetamide with acetone under the catalytic influence of piperidine, a reaction that forms the critical carbon-carbon bond necessary for building the pyridine skeleton. This step proceeds through a dehydration mechanism where the active methylene group of cyanoacetamide attacks the carbonyl carbon of acetone, resulting in the formation of Condensate I with high efficiency. The use of aromatic solvents such as benzene or toluene facilitates the azeotropic removal of water, driving the equilibrium towards product formation and ensuring high conversion rates without the need for excessive reagent excess. Following this, the intermediate undergoes a vinylogous amidine formation reaction with N,N-dimethylformamide dimethyl acetal (DMF-DMA) to generate the conjugated enamine Condensate II. This transformation is crucial as it activates the molecule for the subsequent ring-closing step, introducing the necessary nitrogen functionality and extending the conjugated system required for cyclization. The precise control of temperature and stoichiometry during this stage is vital to prevent polymerization or decomposition, ensuring that the reactive enamine species remains stable prior to the cyclization event.
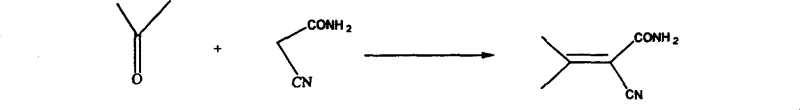
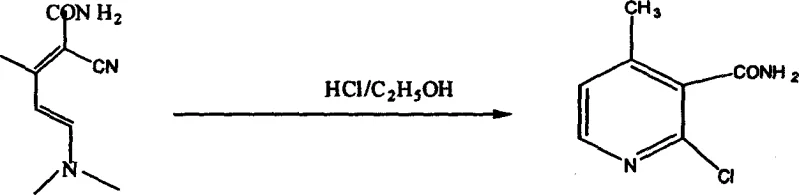
The cyclization step represents the core structural formation of the pyridine ring, achieved by treating the conjugated enamine with hydrochloric acid in ethanol, which promotes intramolecular nucleophilic attack and ring closure. This acid-catalyzed process effectively constructs the heterocyclic core, yielding a chlorinated pyridine amide intermediate that retains the necessary functional groups for the final transformation. The final stage involves a Hofmann degradation reaction using bromine and sodium hydroxide, which converts the amide functionality into the primary amine, completing the synthesis of 2-chloro-3-amino-4-methylpyridine. This degradation step is highly selective, preserving the chloro and methyl substituents on the pyridine ring while efficiently excising the carbonyl carbon. The mechanistic elegance of this route lies in its step-economy and the use of robust, well-understood chemical transformations that are easily controlled on a large scale. Each intermediate can be isolated and purified if necessary, providing multiple quality control checkpoints that ensure the final API intermediate meets stringent purity specifications required by regulatory bodies.
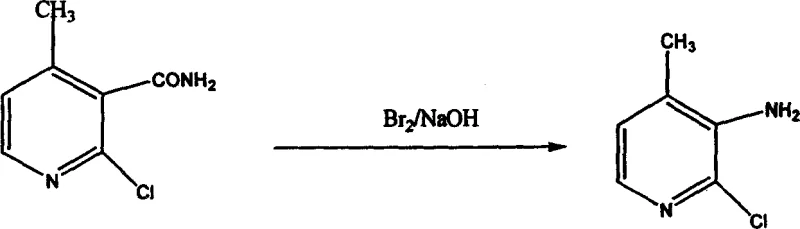
How to Synthesize 2-Chloro-3-Amino-4-Methylpyridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters and workup procedures to maximize yield and purity at every stage of the process. The detailed standardized synthetic steps involve precise temperature control during the condensation and enamine formation phases, followed by rigorous pH adjustments during the isolation of intermediates to ensure optimal recovery. Operators must adhere to strict safety protocols when handling bromine and strong acids during the degradation and cyclization steps, utilizing appropriate containment and ventilation systems to mitigate exposure risks. The purification strategy typically involves crystallization or extraction techniques that leverage the solubility differences between the product and impurities, ensuring a high-quality final solid. For a complete breakdown of the operational parameters, reagent ratios, and specific workup instructions, please refer to the standardized guide below.
- Condense cyanoacetamide with acetone using piperidine catalyst to form Condensate I.
- React Condensate I with DMF-DMA to generate the conjugated enamine Condensate II.
- Perform acid-catalyzed cyclization followed by Hofmann degradation to yield the final pyridine amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages for procurement managers and supply chain heads who are tasked with reducing costs and ensuring continuity of supply for critical pharmaceutical ingredients. The elimination of expensive noble metal catalysts and hazardous nitration reagents translates directly into a significantly reduced cost of goods, allowing for more competitive pricing in the global market without compromising on quality. The simplified process flow reduces the number of unit operations required, which in turn lowers energy consumption and labor costs associated with manufacturing, contributing to substantial overall cost savings. Furthermore, the use of common, commercially available solvents and raw materials mitigates the risk of supply disruptions caused by geopolitical issues or raw material shortages, enhancing the reliability of the supply chain. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the burden on quality control laboratories and shortening the release time for batches. This operational efficiency allows manufacturers to respond more quickly to market demand fluctuations, ensuring that downstream API producers have access to the intermediates they need without lengthy lead times.
- Cost Reduction in Manufacturing: The strategic removal of palladium catalysts and complex nitration steps eliminates the need for expensive metal scavenging and hazardous waste treatment, leading to drastic cost optimization in the production budget. By utilizing commodity chemicals like acetone and cyanoacetamide, the raw material cost base is significantly lowered compared to routes relying on specialized nitro-pyridines. The reduced number of synthetic steps also decreases solvent consumption and energy usage, further compounding the financial benefits for large-scale production facilities. These cumulative efficiencies allow for a more robust margin structure, enabling suppliers to offer competitive pricing while maintaining high profitability standards.
- Enhanced Supply Chain Reliability: Sourcing common organic solvents and bulk chemicals ensures that production is not vulnerable to the supply constraints often associated with specialized reagents or imported catalysts. The robustness of the reaction conditions means that manufacturing can be sustained across different geographic locations without requiring highly specialized infrastructure, diversifying supply risk. This flexibility is crucial for maintaining continuity of supply for essential HIV medications, where interruptions can have severe public health consequences. The simplified logistics of raw material procurement also streamline the supply chain, reducing lead times and inventory holding costs for manufacturers.
- Scalability and Environmental Compliance: The avoidance of heavy nitration waste streams simplifies environmental compliance and reduces the cost of wastewater treatment, making the process more sustainable and easier to permit in regulated jurisdictions. The mild reaction conditions facilitate safe scale-up from pilot plant to commercial production volumes without encountering the thermal runaway risks associated with exothermic nitration reactions. This scalability ensures that the supply can grow in tandem with market demand for antiretroviral therapies, supporting global health initiatives. The greener profile of the synthesis also aligns with corporate sustainability goals, enhancing the brand value of the manufacturer in an increasingly eco-conscious market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of 2-chloro-3-amino-4-methylpyridine, based on the detailed patent specifications and industrial best practices. These answers are designed to provide clarity on the feasibility, quality, and logistical aspects of adopting this synthetic route for commercial manufacturing. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their supply chain.
Q: What are the key advantages of this synthesis route over traditional nitration methods?
A: This route avoids hazardous nitration steps and expensive palladium catalysts, significantly reducing environmental waste and raw material costs while maintaining high selectivity.
Q: What is the expected purity level for the final 2-chloro-3-amino-4-methylpyridine product?
A: The patented process consistently achieves product purity exceeding 99%, meeting stringent specifications required for antiretroviral API manufacturing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes common solvents and mild reaction conditions, making it highly adaptable for commercial scale-up from 100 kgs to 100 MT annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Amino-4-Methylpyridine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging deep technical expertise to translate complex patent routes like CN100358871C into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-chloro-3-amino-4-methylpyridine meets the exacting standards required for Nevirapine synthesis. Our commitment to technical excellence means we can navigate the nuances of this specific chemistry, optimizing yields and minimizing impurities to deliver a superior product.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our advanced process capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing prowess can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
