Advanced Manufacturing of 2-Chloro-3-Amino-4-Methylpyridine via Streamlined Aminolysis Technology
Introduction to Advanced Nevirapine Intermediate Synthesis
The global demand for effective antiretroviral therapies continues to drive innovation in the manufacturing of key pharmaceutical intermediates, particularly for non-nucleoside reverse transcriptase inhibitors (NNRTIs) like Nevirapine. A pivotal breakthrough in this domain is detailed in patent CN101565399A, which outlines a highly optimized synthetic pathway for 2-chloro-3-amino-4-methylpyridine (CAPIC), the critical precursor for Nevirapine. This proprietary methodology addresses longstanding inefficiencies in heterocyclic chemistry by fundamentally restructuring the reaction sequence to enhance both economic viability and environmental sustainability. By leveraging a novel direct aminolysis strategy, the process successfully condenses what was traditionally a multi-stage conversion into a singular, high-yielding transformation. This technological leap not only elevates the overall molar yield from a historical baseline of 24% to an impressive 27% but also ensures final product purity levels surpassing 99%. For stakeholders in the fine chemical sector, this represents a paradigm shift towards more robust and scalable manufacturing protocols that align with modern green chemistry principles while maintaining rigorous quality standards required for HIV medication supply chains.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-chloro-4-methylnicotinamide from its ester precursor involved a cumbersome and resource-intensive three-step sequence that posed significant logistical and economic challenges for large-scale producers. The traditional route necessitated an initial alkaline hydrolysis of the ethyl ester to generate the corresponding carboxylic acid, followed by activation using thionyl chloride to form the reactive acid chloride intermediate, and finally, ammonolysis to yield the target amide. This tripartite approach inherently suffers from cumulative yield losses at each isolation and purification stage, alongside the generation of substantial quantities of acidic wastewater and sulfur-containing byproducts that require costly treatment protocols. Furthermore, the handling of thionyl chloride introduces severe safety hazards and corrosion risks to reactor infrastructure, complicating maintenance schedules and increasing the total cost of ownership for manufacturing facilities. The operational complexity of managing three distinct reaction environments, each requiring specific temperature controls and workup procedures, creates bottlenecks that limit throughput and extend production lead times, making the conventional method increasingly untenable in a competitive market focused on cost efficiency and rapid delivery.
The Novel Approach
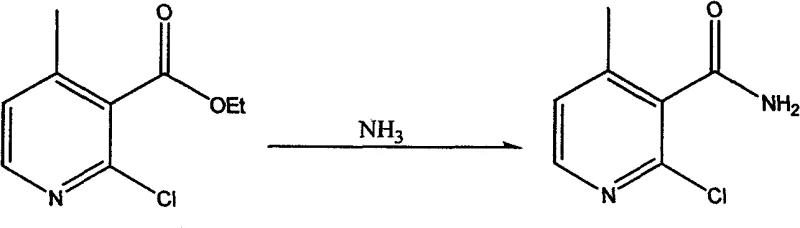
In stark contrast to the fragmented legacy processes, the innovative methodology disclosed in the patent streamlines the conversion of ethyl 2-chloro-4-methylnicotinate directly into the amide through a single-pot aminolysis reaction, effectively bypassing the need for intermediate isolation and activation. This consolidated approach utilizes ammonia gas as a potent nucleophile in high-boiling polar solvents such as ethylene glycol or methanol, facilitating a direct attack on the ester carbonyl under elevated thermal conditions around 120°C. By eliminating the hydrolysis and chlorination steps entirely, the new process drastically reduces the consumption of auxiliary reagents and solvents, thereby minimizing the environmental footprint and simplifying the downstream purification workflow. The result is a more linear and predictable production trajectory where material throughput is maximized, and the risk of cross-contamination between steps is virtually eliminated. This structural simplification of the synthetic route translates directly into enhanced operational agility, allowing manufacturers to respond more swiftly to market demands for Nevirapine intermediates while adhering to stricter regulatory guidelines regarding waste disposal and process safety.
Mechanistic Insights into Direct Ester Aminolysis
The core chemical innovation driving this process improvement lies in the mechanistic execution of the nucleophilic acyl substitution, where ammonia acts as the primary nucleophile against the electrophilic carbonyl carbon of the ester functionality. Unlike traditional methods that rely on converting the carboxyl group into a more reactive acid chloride to overcome the poor leaving group ability of the alkoxide, this method leverages high thermal energy and solvent effects to facilitate the direct displacement of the ethoxy group. The reaction proceeds through a tetrahedral intermediate where the nitrogen lone pair attacks the carbonyl, followed by the elimination of ethanol, driven forward by the excess concentration of ammonia and the removal of the volatile alcohol byproduct. This mechanism is particularly effective in polar protic solvents like ethylene glycol, which stabilize the transition state and enhance the solubility of the gaseous ammonia reactant, ensuring homogeneous reaction conditions that promote uniform conversion rates throughout the batch. Understanding this mechanistic nuance is critical for process chemists aiming to replicate these results, as precise control over ammonia saturation and temperature gradients is essential to prevent side reactions and ensure the integrity of the sensitive chloropyridine ring system during the transformation.
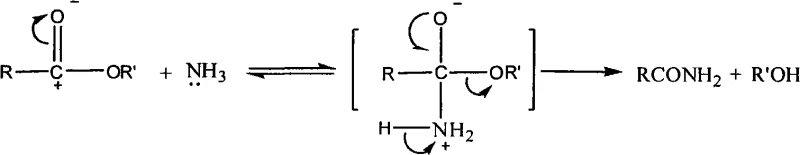
Furthermore, the impurity profile of the final product is significantly improved due to the absence of harsh chlorinating agents and strong bases typically employed in the hydrolysis-activation sequence. In conventional routes, the use of thionyl chloride can lead to the formation of chlorinated byproducts or over-chlorination species that are difficult to separate from the desired amide, often requiring extensive recrystallization or chromatographic purification that further erodes yield. The direct aminolysis pathway avoids these aggressive reagents entirely, resulting in a cleaner crude reaction mixture where the primary impurities are limited to unreacted starting material or minor hydrolysis products that are easily removed during the standard aqueous workup. This inherent selectivity of the new method ensures that the final 2-chloro-3-amino-4-methylpyridine meets stringent purity specifications of greater than 99% with minimal downstream processing, reducing the burden on quality control laboratories and accelerating the release of batches for subsequent coupling reactions in the Nevirapine synthesis pipeline.
How to Synthesize 2-Chloro-3-Amino-4-Methylpyridine Efficiently
The implementation of this streamlined synthesis protocol requires careful attention to reaction parameters, particularly during the aminolysis and cyclization stages, to maximize the benefits of the reduced step count. The process begins with the condensation of ethyl cyanoacetate and acetone, followed by enamine formation and cyclization to establish the pyridine core, before executing the critical direct aminolysis step that defines the efficiency of this route. Operators must ensure strict temperature control during the ammonia saturation phase to maintain optimal reaction kinetics without compromising the stability of the intermediate species. Detailed standard operating procedures regarding solvent selection, ammonia flow rates, and thermal ramping profiles are essential for reproducing the high yields reported in the patent literature. For a comprehensive breakdown of the specific experimental conditions and stoichiometric ratios required for each stage of this five-step sequence, please refer to the standardized synthesis guide provided below.
- Condense ethyl cyanoacetate with acetone using piperidine catalyst to form Condensate I.
- React Condensate I with N,N-dimethylformamide dimethyl acetal to generate the conjugated enamine (Condensate II).
- Cyclize Condensate II in ethanol/HCl to obtain ethyl 2-chloro-4-methylnicotinate.
- Perform direct aminolysis of the ester with ammonia gas at 120°C to yield 2-chloro-4-methylnicotinamide.
- Execute Hofmann degradation using bromine and sodium hydroxide to finalize the synthesis of 2-chloro-3-amino-4-methylpyridine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain management perspective, the adoption of this optimized synthetic route offers profound advantages that extend well beyond simple yield improvements, fundamentally altering the cost structure and reliability of the intermediate supply. By collapsing three discrete unit operations into a single reaction step, manufacturers can significantly reduce the capital expenditure associated with reactor occupancy time, labor hours, and utility consumption, leading to a more competitive pricing model for the finished intermediate. The elimination of thionyl chloride and the associated waste treatment protocols not only lowers compliance costs but also mitigates the supply risk associated with the procurement of hazardous regulated chemicals, ensuring a more resilient and uninterrupted production schedule. This process intensification allows for larger batch sizes to be processed within existing infrastructure, effectively increasing plant capacity without the need for major equipment upgrades, which is a critical factor for meeting the growing global demand for antiretroviral medications in developing markets.
- Cost Reduction in Manufacturing: The consolidation of the hydrolysis, chlorination, and ammoniation steps into a single direct aminolysis reaction eliminates the need for expensive activating agents like thionyl chloride and reduces the consumption of solvents and energy across multiple heating and cooling cycles. This reduction in material intensity and operational complexity translates into substantial cost savings per kilogram of produced intermediate, allowing for more aggressive pricing strategies in tender situations while maintaining healthy profit margins. Additionally, the higher overall yield means that less raw material is required to produce the same amount of final product, further driving down the variable cost of goods sold and enhancing the economic feasibility of large-scale production runs.
- Enhanced Supply Chain Reliability: Simplifying the manufacturing process reduces the number of potential failure points and quality hold instances, leading to more predictable lead times and consistent on-time delivery performance for downstream API manufacturers. The reliance on commodity chemicals like ammonia and common alcohols, rather than specialized or regulated reagents, minimizes the risk of supply disruptions caused by regulatory changes or vendor shortages, ensuring a stable and secure supply of this critical HIV drug intermediate. This reliability is paramount for pharmaceutical companies managing complex global supply chains where any delay in intermediate availability can impact the production of life-saving finished dosage forms.
- Scalability and Environmental Compliance: The streamlined nature of the process facilitates easier scale-up from pilot plant to commercial manufacturing scales, as fewer unit operations need to be validated and optimized for larger volumes. The significant reduction in hazardous waste generation, particularly the avoidance of sulfur-containing effluents and acidic wash streams, simplifies environmental permitting and reduces the long-term liability associated with waste disposal, aligning the production process with increasingly stringent global environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method for 2-chloro-3-amino-4-methylpyridine. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of why this technology represents a superior alternative to legacy manufacturing routes. Stakeholders evaluating this technology for potential licensing or procurement partnerships will find these answers critical for assessing the feasibility and value proposition of integrating this process into their existing supply networks.
Q: How does this new synthesis route improve upon conventional methods for CAPIC production?
A: The patented method reduces the synthetic sequence from seven steps to five steps by replacing a three-step hydrolysis-chlorination-ammoniation sequence with a single direct aminolysis reaction. This consolidation increases the total molar yield from 24% to 27% and achieves product purity exceeding 99%.
Q: What are the specific reaction conditions for the critical aminolysis step?
A: The direct aminolysis of ethyl 2-chloro-4-methylnicotinate is conducted at an elevated temperature of 120°C using solvents such as methanol, ethanol, or ethylene glycol. The reaction utilizes saturated ammonia gas, either in a sealed system or via continuous flow, monitored by TLC to ensure complete conversion.
Q: Why is this method considered more suitable for large-scale industrial production?
A: By eliminating the need for thionyl chloride and separate hydrolysis steps, the process significantly reduces hazardous waste generation and operational complexity. The simplified workflow enhances scalability, lowers raw material consumption, and ensures consistent high purity suitable for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Amino-4-Methylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against HIV, and we are committed to delivering 2-chloro-3-amino-4-methylpyridine that meets the highest standards of purity and consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthetic routes like the one described in CN101565399A are fully realized in practical, industrial settings. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch exceeds the 99% purity threshold required for API synthesis, providing our partners with the confidence they need to streamline their own regulatory filings and production schedules.
We invite pharmaceutical manufacturers and procurement leaders to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to our supply source, along with access to specific COA data and route feasibility assessments tailored to your volume needs. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of this essential Nevirapine intermediate, driving efficiency and success in your antiretroviral drug development programs.
