Advanced Manufacturing of Saxagliptin Intermediate A: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing of Saxagliptin Intermediate A: Technical Breakthroughs and Commercial Scalability
The global demand for Dipeptidyl Peptidase-4 (DPP-4) inhibitors continues to surge as type 2 diabetes prevalence rises, placing immense pressure on the supply chain for high-quality active pharmaceutical ingredients (APIs) and their precursors. Patent CN111170927B discloses a significant technological advancement in the preparation of Saxagliptin Intermediate A, a critical building block for this blockbuster drug class. This novel methodology addresses long-standing inefficiencies in traditional synthetic routes by introducing a streamlined, two-step process that utilizes ethyl dichlorophosphate for cyanation and chlorinating agents for peptide coupling. For R&D directors and procurement specialists seeking a reliable saxagliptin intermediate supplier, understanding the mechanistic superiority and economic implications of this patent is essential for securing a competitive edge in the pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
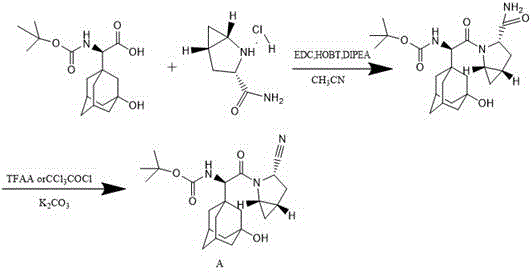
Historically, the synthesis of Saxagliptin Intermediate A has relied on protocols such as those described in WO2004052850, which present substantial operational and economic drawbacks for large-scale manufacturing. The conventional route typically employs carbodiimide-based coupling reagents, specifically EDC (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide) and HOBT (Hydroxybenzotriazole), to facilitate the amide bond formation. While effective on a small laboratory scale, these reagents are prohibitively expensive for industrial application and generate urea byproducts that are notoriously difficult to remove, complicating downstream purification. Furthermore, the subsequent cyanation step in prior art often necessitates the use of excessive trifluoroacetic anhydride (TFAA) under harsh conditions. This aggressive reagent profile frequently leads to unwanted side reactions, most notably the esterification of the sensitive hydroxyl group located on the 3-position of the adamantyl ring, resulting in a complex impurity profile and reduced overall yield.
The Novel Approach
In stark contrast, the methodology outlined in CN111170927B offers a robust alternative that fundamentally reengineers the synthetic strategy to enhance both efficiency and purity. The new process decouples the synthesis into two distinct, highly optimized stages: first, the conversion of a primary amide to a nitrile using ethyl dichlorophosphate, and second, the condensation of this nitrile with the adamantyl-glycine acid component using activated chlorinating agents. By shifting away from TFAA for the dehydration step, the new route operates under significantly milder conditions, preserving the integrity of the hydroxyl functionality and preventing ester formation. Additionally, the replacement of EDC/HOBT with reagents like trichloro-s-triazine or oxalyl chloride for the coupling reaction not only lowers raw material costs but also simplifies the workup procedure, as the byproducts are more easily separated from the desired product. This strategic pivot represents a major leap forward in cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Ethyl Dichlorophosphate-Mediated Cyanation and Coupling
The first critical transformation in this novel pathway involves the dehydration of the primary amide (Compound a) to the corresponding nitrile (Compound b). Mechanistically, ethyl dichlorophosphate acts as a potent dehydrating agent that activates the amide oxygen, facilitating the elimination of water to form the carbon-nitrogen triple bond. Unlike traditional dehydrating agents that may require elevated temperatures or strictly anhydrous conditions, this reaction proceeds efficiently at room temperature (25±2°C) in a mixed solvent system of dichloromethane and DMF or DBU. The choice of solvent plays a pivotal role in stabilizing the transition state and solubilizing the polar intermediates, ensuring high conversion rates. Experimental data from the patent indicates that optimizing the molar ratio of ethyl dichlorophosphate to the substrate (preferably 1:1 to 1:3) allows for precise control over the reaction kinetics, minimizing the formation of unreacted starting material while avoiding the degradation of the sensitive bicyclic amine scaffold.
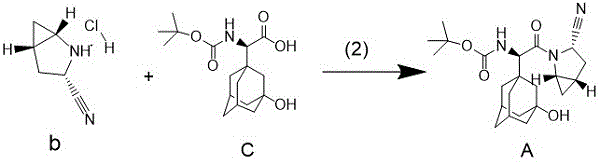
Following the successful generation of the nitrile intermediate, the process moves to the peptide coupling stage, which is the cornerstone of constructing the Saxagliptin backbone. In this step, the carboxylic acid component (Compound c), which contains the bulky 3-hydroxy-1-adamantyl group, is activated in situ using a chlorinating agent such as trichloro-s-triazine. This activation generates a highly reactive acyl chloride or mixed anhydride species that is immediately susceptible to nucleophilic attack by the secondary amine of the nitrile intermediate (Compound b). The presence of a base, such as sodium bicarbonate or triethylamine, is crucial here to scavenge the generated hydrogen chloride and drive the equilibrium toward product formation. This approach effectively circumvents the racemization risks often associated with carbodiimide coupling, ensuring the stereochemical fidelity of the (2S)-configuration is maintained. The result is a high-purity intermediate with minimal epimerization, a critical parameter for regulatory compliance in API synthesis.
How to Synthesize Saxagliptin Intermediate A Efficiently
The implementation of this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process is designed to be operationally simple, avoiding the need for cryogenic cooling or high-pressure reactors, which makes it particularly attractive for contract development and manufacturing organizations (CDMOs). The detailed standardized synthesis steps involve precise control of stoichiometry, solvent ratios, and reaction times to mitigate the formation of specific impurities such as the adamantyl ester. For technical teams looking to adopt this technology, the following guide outlines the critical operational phases derived directly from the patent examples, serving as a foundational protocol for process validation and scale-up activities.
- Cyanation of Compound a: React the primary amide precursor with ethyl dichlorophosphate in a mixed solvent system (e.g., DCM/DMF) at room temperature to form the nitrile intermediate (Compound b).
- Activation of Acid Component: Treat N-Boc-protected adamantyl-glycine (Compound c) with a chlorinating agent such as trichloro-s-triazine or oxalyl chloride in dichloromethane.
- Coupling Reaction: Add the activated acid solution to a mixture containing the nitrile intermediate (Compound b) and a base (e.g., sodium bicarbonate) to form the final Intermediate A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial optimization of the bill of materials (BOM). By eliminating the reliance on premium-priced coupling reagents like EDC and HOBT, which are subject to market volatility and supply constraints, manufacturers can achieve significant cost stabilization. Furthermore, the substitution of trifluoroacetic anhydride with ethyl dichlorophosphate reduces the consumption of fluorinated reagents, which are increasingly scrutinized under environmental regulations. This shift not only lowers direct material costs but also reduces the burden on waste treatment facilities, as the effluent profile becomes less hazardous and easier to manage. Consequently, the overall cost of goods sold (COGS) for the intermediate is drastically improved, allowing for more competitive pricing in the final API market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive, non-recyclable reagents with commodity chemicals. Ethyl dichlorophosphate and trichloro-s-triazine are widely available bulk chemicals with stable pricing structures, unlike the specialized peptide coupling agents used in legacy processes. Additionally, the simplified purification workflow—relying on standard extraction and crystallization rather than complex chromatography—reduces solvent consumption and processing time. This lean manufacturing approach translates directly into lower operational expenditures (OPEX) and higher throughput capacity per batch, enabling suppliers to offer more attractive commercial terms to downstream API producers without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of robust, commercially mature reagents. The dependency on niche reagents that may face shortages is minimized, ensuring a continuous and predictable production schedule. The mild reaction conditions (room temperature) further reduce the risk of batch failures due to equipment malfunction or temperature excursions, which are common causes of supply disruption in exothermic or cryogenic processes. By adopting this method, partners can secure a more reliable saxagliptin intermediate supplier relationship, characterized by consistent lead times and the ability to ramp up production volume rapidly in response to market demand fluctuations for diabetes medications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is inherently greener and more scalable. The avoidance of excessive TFAA eliminates the generation of trifluoroacetic acid waste, a persistent environmental pollutant. The process utilizes common solvents like dichloromethane and toluene, which have well-established recovery and recycling protocols in modern chemical plants. The high yields reported in the patent examples (exceeding 90% in optimized steps) indicate excellent atom economy, meaning less raw material is wasted as byproduct. This efficiency facilitates seamless commercial scale-up of complex pharmaceutical intermediates from pilot plant quantities to multi-ton annual production, meeting the rigorous sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Saxagliptin Intermediate A using the patented method. These insights are derived from a deep analysis of the reaction mechanisms and process data provided in CN111170927B, aimed at clarifying the operational benefits for potential partners. Understanding these nuances is vital for making informed sourcing decisions and ensuring that the selected manufacturing partner possesses the requisite technical expertise to handle the specific challenges of this chemistry.
Q: What are the key advantages of this new synthesis route over the prior art?
A: The new method replaces expensive and difficult-to-remove coupling agents like EDC/HOBT with more economical chlorinating agents. It also avoids the use of excessive trifluoroacetic anhydride (TFAA), thereby eliminating side reactions such as the esterification of the hydroxyl group on the adamantyl ring, leading to higher purity and yield.
Q: What represents the critical quality attribute for this intermediate?
A: The critical quality attributes include the stereochemical integrity of the chiral centers (1S,3S,5S and 2S configurations) and the absence of ester impurities at the 3-hydroxy position of the adamantyl group. The described process achieves HPLC purity exceeding 98% through optimized reaction conditions and recrystallization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates under mild conditions (room temperature, 25±2°C) and utilizes common organic solvents like dichloromethane and toluene. The workup involves standard extraction and crystallization techniques, making it highly scalable from kilogram to multi-ton production without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Saxagliptin Intermediate A Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medications like Saxagliptin depends on a supply chain built on technical excellence and reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Saxagliptin Intermediate A meets the highest international regulatory standards. Our facility is equipped to handle the specific solvent systems and reagent handling requirements of this novel process, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how our optimized manufacturing capabilities can drive value and efficiency in your DPP-4 inhibitor projects.
