Advanced Synthesis of 2-Substituted-5-((1-Alkylthio)Alkyl)Pyridines for Global Agrochemical Applications
Advanced Synthesis of 2-Substituted-5-((1-Alkylthio)Alkyl)Pyridines for Global Agrochemical Applications
The development of efficient synthetic routes for heterocyclic compounds remains a cornerstone of modern agrochemical innovation. Patent CN102532012B introduces a robust and high-yielding methodology for the preparation of 2-substituted-5-((1-alkylthio)alkyl)pyridines, a class of molecules serving as vital precursors for next-generation pesticides. This technology addresses the critical industry demand for scalable processes that minimize waste while maximizing purity, offering a strategic advantage for manufacturers aiming to secure a reliable agrochemical intermediate supplier partnership. The core innovation lies in a streamlined three-step sequence that avoids the pitfalls of traditional halogenation or multi-step substitution reactions, instead leveraging a condensation-cyclization strategy that is both atom-economical and operationally simple.
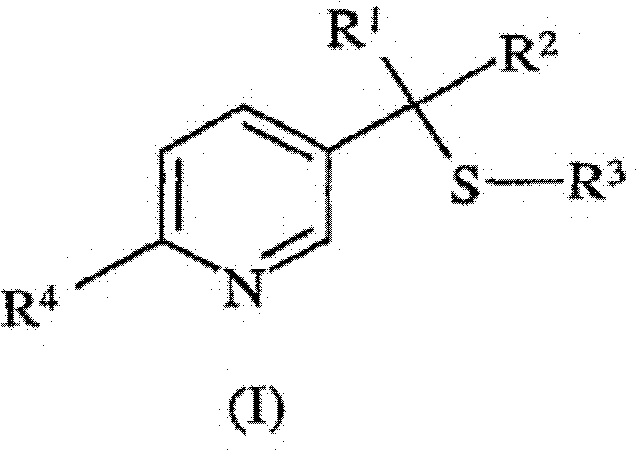
For R&D directors evaluating process feasibility, the versatility of this method is paramount. The patent details a pathway where substituents such as trifluoromethyl groups can be introduced with high fidelity, a feature essential for enhancing the biological activity of the final pesticide active ingredients. By utilizing readily accessible starting materials like substituted enones and enamines, the process reduces dependency on exotic reagents, thereby stabilizing the supply chain against market volatility. Furthermore, the ability to conduct the reaction in common polar solvents such as acetonitrile or dimethylformamide ensures that the transition from laboratory bench to commercial scale-up of complex agrochemical intermediates can be achieved with minimal capital expenditure on specialized equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyridines has often relied on methodologies that suffer from significant drawbacks regarding yield and environmental impact. Traditional approaches frequently involve harsh halogenation steps followed by nucleophilic substitution, which can generate substantial quantities of hazardous waste and require rigorous purification protocols to remove residual halides. These legacy methods often struggle with regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate, ultimately driving up the cost of goods sold. Additionally, the use of heavy metal catalysts in some conventional cross-coupling reactions necessitates expensive removal steps to meet the stringent purity specifications required for agricultural applications, creating bottlenecks in production throughput.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data utilizes a condensation reaction between a substituted enone and an enamine, followed by an ammonia-mediated cyclization. This strategy bypasses the need for pre-functionalized halogenated intermediates, effectively eliminating the associated waste streams and safety hazards. The reaction conditions are mild to moderate, typically ranging from 0°C to 80°C for the initial steps, which preserves the integrity of sensitive functional groups like the alkylthio moiety. This gentle yet effective chemistry ensures that the final product profile is clean, with fewer by-products, thereby simplifying downstream processing and significantly reducing the overall manufacturing footprint.
Mechanistic Insights into Enamine-Enone Condensation and Cyclization
The mechanistic pathway begins with the nucleophilic attack of the enamine on the electrophilic beta-carbon of the substituted enone. This Michael-type addition forms a reactive intermediate that subsequently undergoes intramolecular cyclization in the presence of an ammonia source. The patent specifies that ammonium salts, such as ammonium acetate or ammonium formate, act as effective ammonia donors, facilitating the formation of the dihydropyridine ring which then aromatizes to the pyridine system. This mechanism is particularly advantageous because it allows for the precise installation of the sulfur-containing side chain at the 5-position of the pyridine ring without scrambling or degradation, a common issue in high-temperature syntheses.
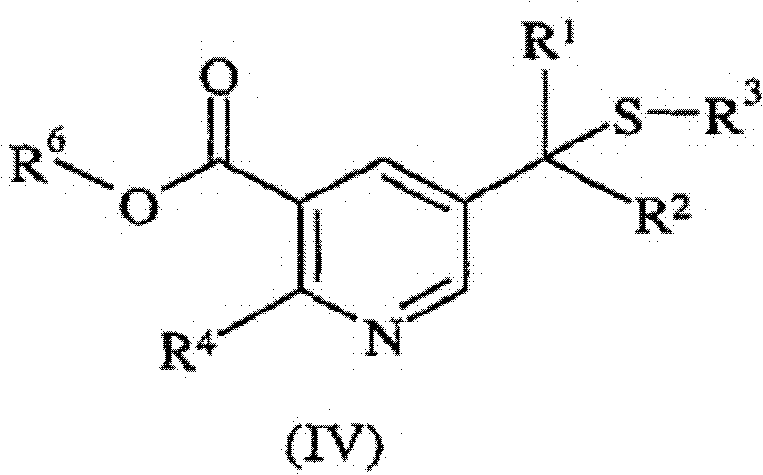
Impurity control is inherently built into this mechanism due to the specificity of the enamine-enone coupling. Unlike radical-based substitutions which can lead to random functionalization, this ionic pathway is highly directed. The subsequent saponification and decarboxylation steps are performed under controlled thermal conditions, typically between 150°C and 250°C in high-boiling solvents like DowTherm A. The inclusion of copper powder during decarboxylation acts as a catalyst to lower the activation energy, ensuring complete conversion while minimizing the formation of tarry by-products. This level of mechanistic control translates directly to a superior impurity profile, meeting the rigorous quality standards demanded by global regulatory bodies for pesticide registration.
How to Synthesize 2-Substituted-5-((1-Alkylthio)Alkyl)Pyridines Efficiently
The synthesis protocol described offers a clear roadmap for production teams, starting from the preparation of the enamine precursor via the reaction of a substituted aldehyde with a secondary amine in the presence of a drying agent. The subsequent condensation with the enone is exothermic and requires careful temperature management, ideally maintained between -5°C and 20°C initially, before heating to drive the cyclization. Detailed standardized operating procedures for each unit operation, including workup and purification via silica gel chromatography or vacuum distillation, are essential to replicate the high yields reported in the patent examples. For a comprehensive breakdown of the specific reaction parameters and stoichiometry, please refer to the technical guide below.
- Condense a substituted enone (II) with an enamine (III) in a polar solvent at temperatures between -20°C and 150°C.
- Cyclize the reaction mixture using ammonia or an ammonium salt (e.g., ammonium acetate) to form the 2,3,5-substituted pyridine intermediate (IV).
- Perform saponification followed by thermal decarboxylation at 150°C to 250°C to yield the final 2-substituted-5-((1-alkylthio)alkyl)pyridine (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers transformative potential for cost reduction in pesticide manufacturing. By eliminating the need for expensive transition metal catalysts in the primary bond-forming steps, the process removes a significant cost center associated with both reagent purchase and heavy metal scavenging technologies. The reliance on bulk commodity chemicals such as acetoacetates, orthoformates, and simple amines ensures that raw material costs remain stable and predictable, shielding the project from the volatility often seen with specialized organometallic reagents. This stability allows for more accurate long-term budgeting and pricing strategies for the final agrochemical active ingredient.
- Cost Reduction in Manufacturing: The streamlined nature of the process significantly lowers operational expenditures by reducing the number of isolation steps required. Since the reaction proceeds with high conversion and selectivity, the need for extensive chromatographic purification is minimized, allowing for simpler crystallization or distillation techniques that are far more economical at scale. Furthermore, the avoidance of cryogenic conditions for the main reaction steps reduces energy consumption, contributing to a lower carbon footprint and reduced utility costs for the manufacturing facility.
- Enhanced Supply Chain Reliability: The starting materials identified in this process, such as substituted propionaldehydes and pyrrolidine, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base ensures continuity of supply even during market disruptions, a critical factor for maintaining production schedules for high-volume pesticide campaigns. The robustness of the chemistry also means that slight variations in raw material quality can be accommodated without compromising the final product specification, adding another layer of resilience to the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-gram to kilogram scales in the patent examples without loss of efficiency. The use of recyclable solvents like acetonitrile and the generation of benign by-products such as water and simple organic acids align with modern green chemistry principles. This alignment facilitates easier regulatory approval and reduces the burden on waste treatment facilities, ensuring that the manufacturing site remains compliant with increasingly strict environmental regulations regarding effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is crucial for process engineers and quality assurance teams when adapting this chemistry to their specific production environments. The answers provided reflect the optimal conditions and scope defined by the inventors to ensure successful replication.
Q: What are the key advantages of this pyridine synthesis method?
A: The process described in CN102532012B offers high yields and utilizes commercially available starting materials like acetoacetates and orthoformates, significantly simplifying the supply chain for agrochemical intermediates.
Q: Is this method suitable for trifluoromethyl-substituted pyridines?
A: Yes, the patent explicitly highlights that the method is particularly well-suited for preparing compounds where the R4 group is a trifluoromethyl (CF3) group, which is critical for modern pesticide efficacy.
Q: What conditions are required for the decarboxylation step?
A: The final step involves heating the pyridine carboxylate in a high-boiling solvent like DowTherm A, optionally with copper powder, at temperatures ranging from 150°C to 250°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted-5-((1-Alkylthio)Alkyl)Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of novel crop protection solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-substituted-5-((1-alkylthio)alkyl)pyridine meets the exacting standards required for downstream pesticide synthesis.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your time-to-market while optimizing your overall production costs.
