Advanced Caspofungin Synthesis: High Yield Intermediates for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes for critical antifungal agents, and patent CN101305018A introduces a transformative approach to synthesizing caspofungin. This innovation centers on novel azacyclic hexapeptide intermediates that streamline the conversion of Pneumocandin B0 into the final active pharmaceutical ingredient. By leveraging a unique nitrile functionality within the intermediate structure, the process overcomes historical bottlenecks related to yield and purity that have long plagued echinocandin manufacturing. The strategic implementation of catalytic hydrogenation allows for highly selective reduction even in the presence of sensitive aminal groups, ensuring that the structural integrity of the macrocyclic core remains intact throughout the synthesis. This technological leap not only enhances the economic viability of production but also assures a consistent supply of high-quality material for global healthcare needs.
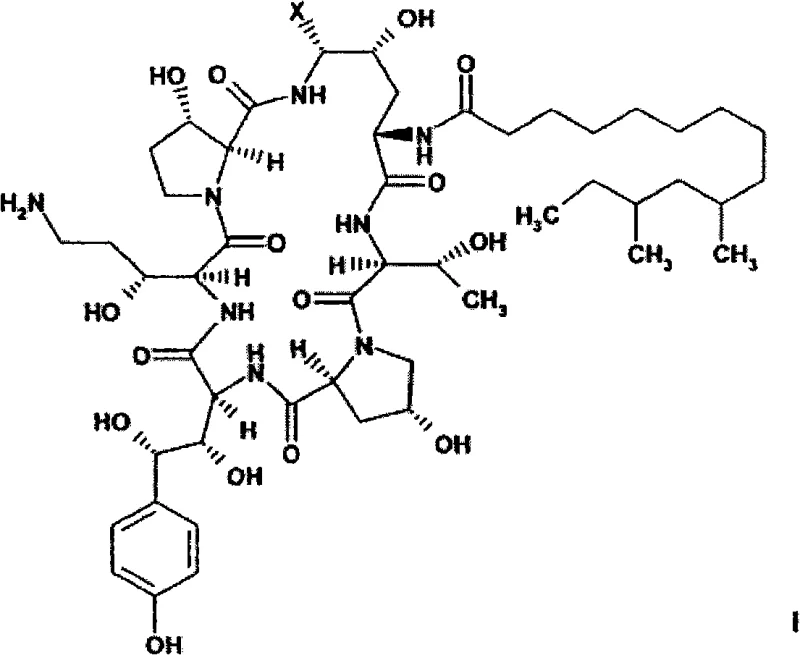
Conventional methods for producing caspofungin have historically relied on the reduction of primary amide functional groups, a transformation that is notoriously inefficient and prone to side reactions. Prior art techniques, such as those described in WO96/24613, often struggle to achieve yields exceeding 47%, necessitating extensive recycling of starting materials and generating significant chemical waste. Furthermore, these legacy processes frequently demand strictly anhydrous conditions and the use of molecular sieves, which complicates reactor operations and increases energy consumption. The reliance on multiple chromatographic purification steps to remove by-products further escalates costs and extends lead times, creating friction in the supply chain for manufacturers aiming to meet high-volume demand. These limitations underscore the urgent need for a more direct and efficient synthetic pathway that can operate under milder conditions.
The novel approach detailed in the patent data fundamentally reimagines the reduction step by utilizing a nitrile-containing intermediate rather than a direct amide reduction. This strategic modification enables the use of catalytic hydrogenation with noble metal catalysts such as Rh/Al2O3 or palladium on carbon, which drives the reaction to completion with remarkable efficiency. Reaction yields can reach approximately 82% by weight, a substantial improvement over the roughly 47% to 61% observed in previous methodologies. The process tolerates the presence of other functional groups without requiring extensive protecting group strategies, thereby simplifying the overall synthetic sequence. By eliminating the need for rigorous anhydrous environments and reducing the dependency on chromatography, this new route offers a clearer path to industrial scalability and cost optimization.
Mechanistic Insights into Catalytic Hydrogenation of Nitrile Intermediates
The core of this technological advancement lies in the highly selective catalytic hydrogenation of the nitrile group located at the C5-ornithine position of the azacyclic hexapeptide backbone. Unlike traditional reductions that might compromise the delicate aminal linkage within the macrocycle, this method employs specific catalyst systems that target the nitrile functionality exclusively. The use of hydrogen gas at pressures ranging from atmospheric pressure to 20 bar, preferably around 1 bar, in a solvent system of isopropanol and water, creates an ideal environment for this transformation. The addition of acids such as acetic acid during the reduction step further suppresses the formation of impurities, ensuring that the reaction proceeds with high chemoselectivity. This precision is critical for maintaining the stereochemical integrity of the molecule, which is essential for its biological activity as an antifungal agent.
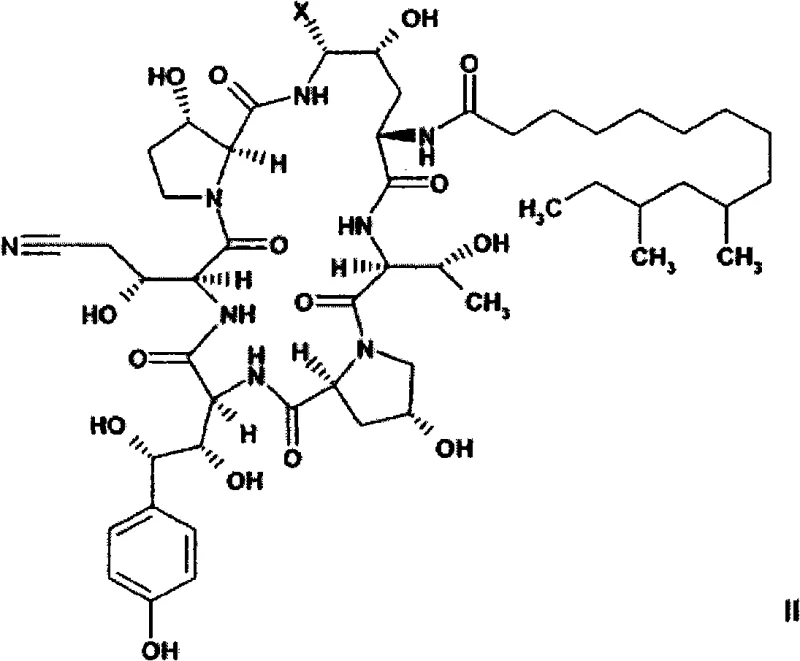
Impurity control is another pivotal aspect of this mechanism, particularly regarding the removal of the benzylic C-35 epimer which can arise during the synthesis. The novel intermediates, specifically the Formula VI compounds, possess physicochemical properties that allow for effective purification through crystallization rather than relying solely on chromatography. By isolating the intermediate as a monoacetate salt, manufacturers can exploit differences in solubility to precipitate the desired product while leaving epimeric impurities in the mother liquor. This capability is a significant advantage, as chromatographic removal of such closely related structural analogs is often economically prohibitive on a large scale. The ability to achieve high purity through crystallization not only reduces processing time but also minimizes solvent usage, aligning with modern green chemistry principles and regulatory expectations for impurity profiles.
How to Synthesize Caspofungin Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for converting Pneumocandin B0 into caspofungin through a series of well-defined chemical transformations. The process begins with the dehydration of the starting material to form a nitrile, followed by substitution reactions that introduce the necessary side chains before the final reduction step. Each stage is optimized to maximize conversion and minimize the formation of by-products, ensuring a smooth flow from raw materials to the final active ingredient. The detailed standardized synthesis steps below offer a comprehensive guide for technical teams looking to implement this methodology in their own facilities, highlighting critical parameters such as temperature control and reagent stoichiometry.
- Dehydrate Pneumocandin B0 using cyanuric chloride to form the nitrile intermediate.
- React the nitrile compound with thiophenol to introduce the thio-ether linkage.
- Perform catalytic hydrogenation on the ethylenediamine adduct to achieve high-yield reduction.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route addresses several critical pain points that typically impact the cost and reliability of pharmaceutical supply chains. The significant increase in reaction yield directly translates to reduced raw material consumption, lowering the overall cost of goods sold without compromising on quality standards. By simplifying the purification process and reducing the number of unit operations required, manufacturers can achieve faster batch cycle times and higher throughput in existing production facilities. These efficiencies contribute to a more resilient supply chain capable of responding swiftly to market fluctuations and unexpected demand surges. Furthermore, the robustness of the process under less stringent conditions reduces the risk of batch failures, ensuring greater consistency in supply continuity for downstream partners.
- Cost Reduction in Manufacturing: The elimination of expensive protecting groups and the reduction in chromatographic purification steps lead to substantial cost savings in reagent and solvent procurement. The higher yield means less starting material is wasted, optimizing the utilization of high-value precursors like Pneumocandin B0. Additionally, the ability to operate at atmospheric pressure for hydrogenation reduces energy costs associated with high-pressure reactors. These cumulative factors result in a more economical manufacturing process that enhances profit margins while maintaining competitive pricing structures for the final API.
- Enhanced Supply Chain Reliability: The use of readily available catalysts and solvents ensures that the supply chain is not vulnerable to shortages of specialized reagents. The stability of the crystalline intermediates allows for safer storage and transportation, reducing the risk of degradation during logistics. This robustness enables manufacturers to maintain strategic stockpiles of key intermediates, buffering against potential disruptions in the upstream supply of natural fermentation products. Consequently, partners can rely on a steady and predictable flow of materials, facilitating better planning and inventory management across the global network.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, avoiding conditions that are difficult to replicate in large-scale reactors such as strictly anhydrous environments. The reduction in solvent intensity and waste generation aligns with increasingly stringent environmental regulations, minimizing the burden on waste treatment facilities. This eco-friendly profile not only reduces compliance costs but also enhances the corporate sustainability image of the manufacturing entity. The straightforward workup procedures, involving simple filtration and crystallization, are easily adaptable to multi-ton production scales, ensuring that commercial demands can be met efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. They are derived from the specific beneficial effects and background technical challenges identified in the patent documentation. Understanding these aspects is crucial for stakeholders evaluating the feasibility of adopting this technology for their production lines. The answers provide clarity on yield expectations, impurity management, and the overall operational advantages that distinguish this method from conventional approaches.
Q: How does the new nitrile reduction method improve yield compared to prior art?
A: The novel catalytic hydrogenation method achieves yields of 80% to 90%, significantly surpassing the 47% to 61% yields observed in conventional amide reduction techniques.
Q: What are the stability advantages of the Formula VIa intermediate?
A: The monoacetate salt form (Formula VIa) offers enhanced crystallinity and stability, facilitating easier purification and removal of C-35 epimers without complex chromatography.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process eliminates strictly anhydrous conditions and reduces reliance on multiple chromatographic steps, making it highly scalable and cost-effective for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caspofungin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. We are committed to meeting stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify the identity and quality of every batch. By leveraging advanced process chemistry expertise, we can help you navigate the complexities of caspofungin manufacturing, delivering a reliable supply of high-quality intermediates and APIs that meet global regulatory standards.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain dynamics. Request a Customized Cost-Saving Analysis to understand the specific economic benefits applicable to your operation. Our experts are ready to provide specific COA data and conduct thorough route feasibility assessments tailored to your production capacity. Partnering with us means gaining access to a wealth of technical knowledge and a commitment to excellence that drives value across your entire organization. Let us collaborate to enhance your manufacturing efficiency and secure your position in the competitive antifungal market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
