Advanced Metalloporphyrin Catalysis for Commercial Scale-up of Complex Pharmaceutical Intermediates
Introduction to Breakthrough Oxidation Technology
The chemical industry is constantly seeking more efficient pathways for producing high-value intermediates, and patent CN101759536A presents a significant advancement in the synthesis of p-hydroxybenzaldehyde. This technology leverages the unique electronic properties of metalloporphyrin complexes to catalyze the oxidation of p-cresol, offering a distinct alternative to traditional, resource-intensive methods. By utilizing specific mononuclear and binuclear metalloporphyrin structures, the process achieves high selectivity under remarkably mild conditions, typically ranging from 50°C to 90°C at atmospheric pressure. This innovation addresses critical pain points in fine chemical manufacturing, including excessive catalyst consumption, complex separation protocols, and the generation of hazardous waste streams. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is essential for evaluating its potential integration into existing supply chains for pharmaceutical and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of p-hydroxybenzaldehyde has relied on methods such as the Reimer-Tiemann reaction, Gattermann synthesis, or oxidation using manganese trioxide. These conventional pathways are fraught with inefficiencies that pose significant challenges for commercial scale-up and environmental compliance. A primary drawback is the reliance on supported catalysts, such as cobalt phthalocyanine loaded on activated carbon, which necessitates complex preparation steps and high energy consumption during the loading process. Furthermore, these traditional methods often require substantial amounts of co-catalysts, including transition metal and rare earth inorganic salts, which complicate the downstream purification process. The separation and recovery of these supported catalysts are not only costly but also generate significant volumes of saline wastewater and solid waste, creating a heavy burden on waste treatment facilities and increasing the overall carbon footprint of the manufacturing operation.
The Novel Approach
In stark contrast, the metalloporphyrin-catalyzed oxidation method described in the patent data introduces a streamlined, homogeneous catalytic system that eliminates the need for solid supports and co-catalysts. This novel approach utilizes highly active mononuclear or binuclear metalloporphyrins at extremely low loadings, typically between 0.001% and 0.08% of the raw material weight, which is a fraction of the catalyst quantity required by prior art. The ability to operate without separating or recovering the catalyst post-reaction is a game-changer, as the metalloporphyrins are designed to be biodegradable, thereby preventing secondary pollution. Additionally, the flexibility to use aqueous alcohol solvents instead of strictly anhydrous systems reduces raw material costs and simplifies solvent management. This shift from heterogeneous, high-waste processes to a efficient, low-loading homogeneous system represents a substantial leap forward in green chemistry for fine chemical intermediates.
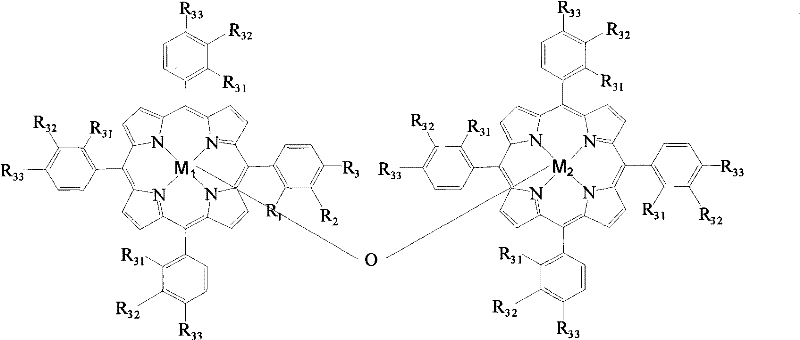
Mechanistic Insights into Metalloporphyrin-Catalyzed Oxidation
The core of this technological advancement lies in the specific structural configuration of the metalloporphyrin catalysts, which facilitate the activation of molecular oxygen for the selective oxidation of the methyl group in p-cresol. The patent details three primary structural formulas (I, II, and III), where the central metal ions (M1, M2) can be iron, cobalt, manganese, copper, zinc, chromium, or nickel. These metal centers within the porphyrin macrocycle act as active sites for oxygen binding and electron transfer, enabling the reaction to proceed efficiently at normal pressure. The presence of specific substituents (R groups) on the phenyl rings of the porphyrin structure further tunes the electronic environment of the metal center, optimizing the catalytic activity and stability during the reaction cycle. This precise molecular engineering allows for high conversion rates and selectivity without the need for harsh oxidizing agents or extreme thermal conditions.
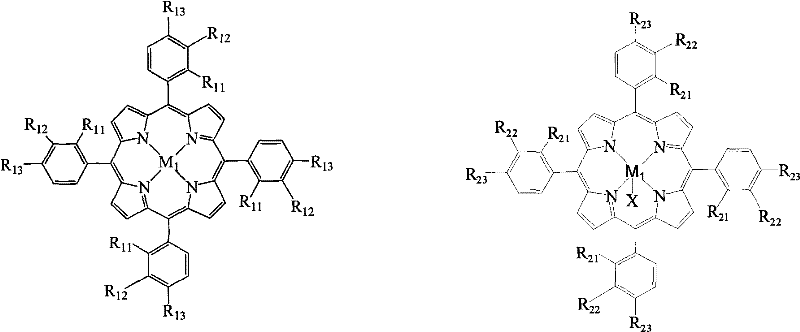
From an impurity control perspective, the mechanism of this catalytic system inherently minimizes the formation of by-products associated with over-oxidation or ring degradation. Traditional methods often struggle with controlling the oxidation state, leading to the formation of p-hydroxybenzoic acid or other polymeric by-products that are difficult to separate. The metalloporphyrin catalyst, however, demonstrates a high degree of chemoselectivity, favoring the transformation of the methyl group to an aldehyde while preserving the aromatic ring and the hydroxyl group. The absence of inorganic salt co-catalysts further reduces the risk of metal contamination in the final product, which is a critical quality attribute for pharmaceutical intermediates. This clean reaction profile simplifies the purification workflow, reducing the need for extensive chromatography or recrystallization steps, and ultimately leading to a higher quality final product with a cleaner impurity profile.
How to Synthesize p-Hydroxybenzaldehyde Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction mixture and the control of oxidation parameters to maximize yield and selectivity. The process begins with the dissolution of p-cresol in a solvent system that can range from pure monohydric alcohols like methanol or ethanol to mixtures containing water, providing flexibility based on solvent availability and cost. The addition of the metalloporphyrin catalyst is critical, and it must be introduced at the specified low concentration alongside sodium hydroxide, which acts as a promoter for the oxidation reaction. Detailed standardized synthesis steps, including specific molar ratios, temperature ramping profiles, and oxygen flow rates, are essential for reproducibility and safety during scale-up. The following guide outlines the critical operational parameters derived from the patent examples to ensure successful implementation of this technology in a pilot or commercial setting.
- Prepare the reaction mixture by dissolving p-cresol in a solvent system comprising monohydric alcohol, dihydric alcohol, or a mixture of alcohol and water.
- Introduce the metalloporphyrin catalyst (Formula I, II, or III) at a loading of 0.001% to 0.08% relative to the raw material weight, along with sodium hydroxide.
- Maintain the reaction temperature between 50°C and 90°C while introducing oxygen or air at normal pressure for 6 to 13 hours to achieve oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metalloporphyrin catalysis technology offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational resilience. The most significant advantage is the drastic reduction in catalyst consumption, which translates to lower raw material costs and reduced dependency on expensive transition metals. Since the catalyst does not require recovery or regeneration, the operational expenditure associated with filtration, washing, and reactivation of solid catalysts is completely eliminated. This simplification of the process flow also reduces the overall cycle time per batch, allowing for increased throughput without the need for additional capital investment in separation equipment. Furthermore, the ability to use aqueous solvents reduces the fire hazard profile of the plant and lowers the cost of solvent procurement and disposal, contributing to a safer and more cost-effective manufacturing environment.
- Cost Reduction in Manufacturing: The elimination of supported catalyst preparation and the removal of co-catalyst requirements lead to substantial cost savings in raw material procurement. By avoiding the use of activated carbon supports and rare earth metal salts, the process reduces the complexity of the supply chain for reagents. The low loading of the primary catalyst means that even if the metalloporphyrin itself is specialized, the total cost per kilogram of product remains highly competitive. Additionally, the reduction in sodium hydroxide usage decreases the consumption of acid for neutralization in the workup phase, further lowering chemical costs and waste treatment fees associated with saline effluent.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system under mild conditions (50-90°C) and atmospheric pressure reduces the risk of equipment failure and safety incidents, ensuring more consistent production schedules. The flexibility in solvent choice, allowing for the use of water-mixed alcohols, mitigates the risk of supply disruptions for strictly anhydrous solvents. Moreover, the biodegradable nature of the catalyst removes the logistical burden of managing hazardous spent catalyst waste, simplifying compliance with environmental regulations and ensuring uninterrupted operations. This reliability is crucial for maintaining long-term contracts with downstream pharmaceutical customers who require consistent quality and delivery.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the homogeneous nature of the reaction, which ensures uniform heat and mass transfer in large reactors. The significant reduction in three wastes (waste gas, waste water, and solid waste) aligns with increasingly stringent global environmental standards, reducing the risk of regulatory shutdowns. The lower salinity of the wastewater simplifies treatment processes, making it easier to meet discharge limits and reducing the environmental tax burden. This eco-friendly profile enhances the corporate sustainability image, which is becoming a key factor in supplier selection for major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metalloporphyrin oxidation technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams. Understanding these details is vital for assessing the feasibility of adopting this method for your specific production needs. The responses cover catalyst handling, solvent flexibility, and environmental impact, providing a comprehensive overview of the operational expectations.
Q: What are the primary advantages of using metalloporphyrin catalysts over traditional cobalt chelates?
A: Metalloporphyrin catalysts require significantly lower loading (0.001-0.08% vs 0.2-5.1%), eliminate the need for complex support materials like activated carbon, and do not require separation or recovery post-reaction due to their biodegradable nature.
Q: Does this process require anhydrous solvents?
A: No, unlike prior art methods that strictly require anhydrous monohydric alcohols, this invention allows for the use of aqueous alcohol solutions (60-99% volume), which simplifies solvent handling and reduces drying costs.
Q: How does this method impact environmental compliance in large-scale manufacturing?
A: The process significantly reduces saline wastewater generation by lowering sodium hydroxide consumption (37.5-80% vs 55.6-222%) and eliminates heavy metal salt co-catalysts, thereby simplifying waste treatment and reducing environmental pollution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Hydroxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the metalloporphyrin oxidation method for producing high-purity p-hydroxybenzaldehyde. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of analyzing complex impurity profiles to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging such green chemistry advancements to deliver products that are not only cost-effective but also environmentally sustainable.
We invite you to collaborate with us to optimize your supply chain for p-hydroxybenzaldehyde and other critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality specifications. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance data. By partnering with us, you gain access to a reliable network of chemical expertise dedicated to driving efficiency and innovation in your manufacturing processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
