Advanced Catalytic Synthesis of p-Hydroxybenzaldehyde for Commercial Scale-up and High-Purity Applications
Introduction to Advanced p-Hydroxybenzaldehyde Manufacturing
The global demand for high-purity p-hydroxybenzaldehyde, a critical intermediate in the synthesis of pharmaceuticals such as esmolol and antimicrobial agents, as well as premium fragrances like vanillin and anisaldehyde, necessitates robust and efficient production methodologies. Patent CN113121323B introduces a transformative two-step catalytic process that addresses longstanding inefficiencies in traditional synthesis routes. This technology leverages a synergistic combination of Lewis acid catalysis in the condensation phase and heterogeneous spinel catalysis in the oxidation phase to achieve exceptional selectivity and yield. By utilizing inexpensive and readily available raw materials like phenol and glyoxylic acid, this method offers a compelling alternative to hazardous or cost-prohibitive legacy processes. The core innovation lies in the precise control of electronic effects during the initial condensation and the optimization of surface oxygen adsorption during the subsequent oxidation, ensuring a streamlined pathway to commercial-grade products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-hydroxybenzaldehyde has been plagued by significant technical and environmental hurdles associated with established routes such as the Reimer-Tiemann and Gattermann reactions. The Reimer-Tiemann process, while conceptually simple, suffers from poor regioselectivity, generating substantial quantities of the ortho-isomer alongside the desired para-product, which complicates purification and drastically reduces overall yield. Furthermore, the reliance on chloroform and strong bases leads to the formation of heavy tars and hazardous waste streams, imposing severe burdens on environmental compliance and waste treatment infrastructure. Similarly, the Gattermann reaction requires the handling of highly toxic hydrocyanic acid and corrosive hydrogen chloride, presenting unacceptable safety risks for large-scale operations. Alternative routes involving p-cresol oxidation often depend on expensive raw materials and volatile organic solvents like methanol, which introduce explosion hazards and inflate production costs, making them less attractive for cost-sensitive bulk chemical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN113121323B establishes a safer and more economically viable pathway by decoupling the synthesis into two distinct, optimized catalytic stages. The first stage employs aluminum or zinc salts to direct the electrophilic attack of glyoxylic acid exclusively to the para-position of the phenol ring, effectively suppressing the formation of ortho-impurities at the molecular level. This is followed by a highly efficient aerobic oxidation step utilizing a reusable copper ferrite catalyst, which operates under moderate temperatures and oxygen pressures. This novel approach eliminates the need for toxic cyanides and chlorinated solvents, significantly reducing the environmental footprint and operational risks. The result is a process that not only achieves total yields reaching up to 85.0% but also simplifies the downstream processing requirements, thereby enhancing the overall economic feasibility for industrial adoption.
Mechanistic Insights into Aluminum/Zinc Salt and Copper Ferrite Catalysis
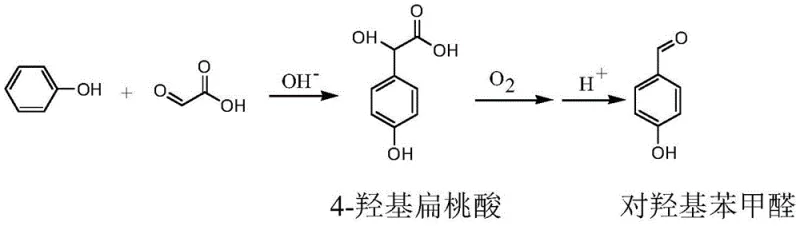
The exceptional performance of this synthesis route is rooted in the sophisticated manipulation of electronic density and surface chemistry. In the initial condensation step, the presence of aluminum or zinc salts plays a pivotal role in modulating the reactivity of the phenoxide anion formed under alkaline conditions. While the oxygen atom in phenol naturally donates electron density to the benzene ring via p-π conjugation, favoring ortho-attack, the coordination of aluminum or zinc ions specifically enhances the electron cloud density at the para-position relative to the ortho-position. This electronic differentiation ensures that the nucleophilic addition of glyoxylic acid occurs predominantly at the para-site, minimizing the generation of 2-hydroxymandelic acid (By-product 1) and 1,3-bis(2-hydroxyacetyl)-4-hydroxybenzene (By-product 2). This mechanistic control is critical for achieving the reported selectivity improvements from approximately 84% in uncatalyzed systems to over 94% in the presence of these metal salts.
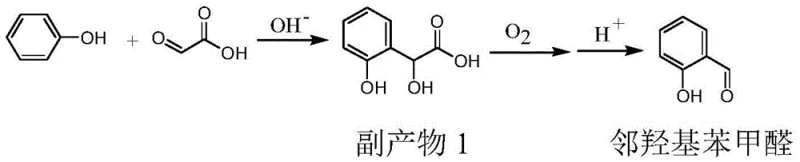
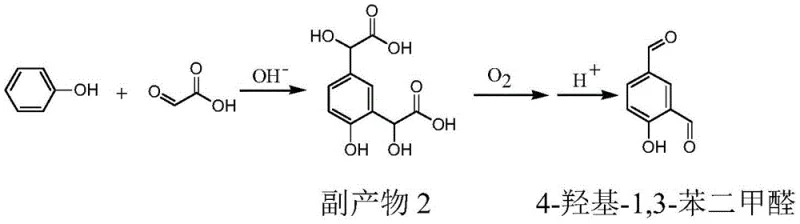
Following the condensation, the oxidation of 4-hydroxymandelic acid to p-hydroxybenzaldehyde is driven by the unique properties of the copper ferrite (CuFe2O4) catalyst. This catalyst features an inverse spinel structure where iron and copper cations exist in multiple valence states (Fe2+/Fe3+ and Cu+/Cu2+) on the surface. The electron transfer between these mixed-valence cations creates a metastable state characterized by lattice defects and low-coordination oxygen atoms. These structural features dramatically increase the capacity for chemical adsorption of both the organic substrate and molecular oxygen. Consequently, the activation energy for the oxidative decarboxylation is lowered, allowing the reaction to proceed rapidly at temperatures between 70°C and 110°C under relatively low oxygen pressures (0.10-0.30 MPa). This mechanism ensures near-quantitative conversion of the mandelic acid intermediate while maintaining 100% selectivity for the aldehyde product, preventing over-oxidation to carboxylic acids.
How to Synthesize p-Hydroxybenzaldehyde Efficiently
The implementation of this patented technology requires careful attention to reaction parameters to maximize the benefits of the dual-catalyst system. The process begins with the preparation of the copper ferrite catalyst via co-precipitation and calcination, followed by the sequential execution of the condensation and oxidation steps in controlled reactor environments. Operators must maintain strict pH control during the condensation phase to ensure the formation of the active phenoxide species without degrading the catalyst, and subsequently manage oxygen pressure and temperature during oxidation to optimize reaction kinetics. For a comprehensive understanding of the specific operational parameters, reagent ratios, and workup procedures required to replicate these high-yield results, please refer to the standardized synthesis protocol outlined below.
- Condense phenol and glyoxylic acid under alkaline conditions (pH 11-14) using aluminum or zinc salts as catalysts at 10-50°C to form 4-hydroxymandelic acid.
- Recover the catalyst by filtration at pH 8.0, then acidify the mother liquor to pH 3.0 to extract unreacted phenol, retaining the 4-hydroxymandelic acid solution.
- Oxidize the 4-hydroxymandelic acid solution using a copper ferrite (CuFe2O4) catalyst under oxygen pressure (0.10-0.30 MPa) at 70-110°C to yield p-hydroxybenzaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology represents a strategic opportunity to mitigate risk and optimize cost structures in the production of fine chemical intermediates. By shifting away from processes reliant on volatile, toxic, or imported specialty reagents, manufacturers can secure a more resilient supply chain that is less susceptible to regulatory crackdowns or raw material price fluctuations. The use of commodity chemicals like phenol and glyoxylic acid, combined with robust heterogeneous catalysts that can be recovered and reused, fundamentally alters the cost equation by reducing both material expenses and waste disposal liabilities. This transition supports a more sustainable manufacturing model that aligns with increasingly stringent global environmental standards while delivering consistent product quality.
- Cost Reduction in Manufacturing: The elimination of hazardous reagents such as hydrocyanic acid and chloroform removes the need for specialized containment systems and expensive neutralization protocols, leading to substantial operational savings. Furthermore, the high selectivity of the aluminum/zinc catalyzed condensation minimizes the loss of valuable phenol feedstock to ortho-isomers and polymeric by-products, directly improving material efficiency. The ability to recover and reuse the copper ferrite catalyst further decreases the recurring cost of catalytic materials, contributing to a lower overall cost of goods sold without compromising on reaction performance or throughput.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly strengthened by the reliance on phenol and glyoxylic acid, which are produced at massive scales globally for diverse industrial applications, ensuring stable availability and competitive pricing. Unlike routes dependent on p-cresol, which may face supply constraints due to its competing uses in antioxidants and polymers, this phenol-based route leverages a ubiquitous feedstock. Additionally, the mild reaction conditions reduce the wear and tear on production equipment, minimizing unplanned downtime and maintenance intervals, thereby guaranteeing more reliable delivery schedules to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard stainless steel reactors capable of handling mild alkaline conditions and moderate oxygen pressures without requiring exotic metallurgy. The reduction in hazardous waste generation, particularly the absence of chlorinated organic waste and cyanide residues, simplifies the permitting process and reduces the long-term liability associated with environmental compliance. This cleaner profile facilitates easier expansion of production capacity to meet growing market demand for high-purity pharmaceutical and fragrance intermediates while adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this catalytic synthesis route. These insights are derived directly from the experimental data and mechanistic explanations provided in the patent documentation, offering clarity on how this technology compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios.
Q: How does the aluminum/zinc salt catalyst improve selectivity in the condensation step?
A: The aluminum or zinc salts enhance the electron cloud density specifically at the para-position of the phenoxide anion. This electronic modulation significantly suppresses the formation of ortho-substituted by-products (2-hydroxymandelic acid) and bis-substituted impurities, raising the selectivity of 4-hydroxymandelic acid from roughly 84% to over 94%.
Q: What are the advantages of using copper ferrite (CuFe2O4) over traditional oxidation catalysts?
A: Copper ferrite possesses a specific inverse spinel structure with multivalent cations (Fe2+/Fe3+ and Cu+/Cu2+) on its surface. This creates lattice defects and low-coordination oxygen atoms that facilitate superior chemical adsorption of reactants and oxygen, leading to conversion rates as high as 99.5% with 100% selectivity under mild conditions.
Q: Is this process suitable for large-scale manufacturing compared to the Reimer-Tiemann reaction?
A: Yes, this process is significantly more scalable and safer. Unlike the Reimer-Tiemann reaction which uses hazardous chloroform and produces substantial tar, this method utilizes readily available phenol and glyoxylic acid under mild alkaline conditions, eliminating toxic reagents and simplifying downstream purification and waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Hydroxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN113121323B can be successfully translated into robust industrial operations. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of p-hydroxybenzaldehyde performs consistently in your downstream applications, whether for life-saving pharmaceuticals or high-value fragrances.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in your supply chain.
