Industrial Scale-Up of Non-Phosgene Toltrazuril Biuret Intermediate for Global Veterinary Supply Chains
The global veterinary pharmaceutical sector is increasingly demanding safer, more efficient synthetic routes for critical anticoccidial agents like toltrazuril. Patent CN102731351B introduces a transformative preparation method for 1-methyl-5-[3-methyl-4-(4-trifluoromethylthio-phenoxy)-phenyl]-biuret, a pivotal intermediate in the toltrazuril value chain. This technology addresses long-standing safety and efficiency bottlenecks by replacing hazardous phosgene chemistry with a mild cyanate-mediated process. For R&D directors and supply chain leaders, this patent represents a strategic opportunity to optimize manufacturing protocols while adhering to stricter environmental and safety regulations. The disclosed method not only simplifies the operational workflow but also significantly enhances the overall purity profile of the final active pharmaceutical ingredient, ensuring consistent quality for sensitive veterinary applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of biuret intermediates has relied heavily on isocyanate chemistry derived from phosgene, as detailed in legacy patents like US4874860. This conventional approach necessitates the use of hypertoxic phosgene gas, which imposes severe safety constraints and requires expensive, specialized containment infrastructure to prevent leakage and exposure. Furthermore, the subsequent condensation step often utilizes N-methylurea (MU), a reagent known for its hygroscopic nature and tendency to absorb moisture from the environment. This water sensitivity triggers hydrolysis side reactions that degrade product quality and lower overall yields, often capping total conversion rates at approximately 72%. The reliance on chlorinated solvents and hazardous reagents creates a complex waste management burden, increasing the operational expenditure for compliance and disposal.
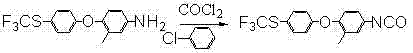
The Novel Approach
In stark contrast, the methodology disclosed in CN102731351B circumvents these critical vulnerabilities by employing an acid-catalyzed reaction between arylamine and cyanate. This innovative pathway completely eliminates the need for gaseous phosgene, thereby removing the associated high-risk safety protocols and reducing the capital expenditure required for gas scrubbing systems. Instead of moisture-sensitive MU, the process utilizes methylcarbamyl chloride in a controlled organic solvent system, which offers superior stability and reactivity. The reaction conditions are remarkably mild, operating effectively between 0°C and 90°C, which reduces energy consumption compared to high-temperature alternatives. This shift not only improves the safety profile for plant operators but also streamlines the purification process, resulting in a cleaner crude product that requires less intensive downstream processing.
Mechanistic Insights into Cyanate-Mediated Urea Synthesis
The core chemical innovation lies in the nucleophilic attack of the arylamine nitrogen on the electrophilic carbon of the cyanate ion, facilitated by an acidic environment. In the presence of acids such as hydrochloric or acetic acid, the cyanate species is activated, allowing for a smooth conversion to the N-arylurea intermediate without the formation of toxic isocyanate byproducts. This mechanism ensures that the reaction proceeds with high atom economy, as the cyanate group is efficiently incorporated into the urea backbone. The subsequent reaction with methylcarbamyl chloride involves a nucleophilic substitution where the urea nitrogen attacks the carbonyl carbon of the chloride, releasing HCl which is neutralized by the reaction medium. This two-step sequence is kinetically favorable and thermodynamically stable, minimizing the formation of oligomeric impurities that often plague phosgene-based routes.
Impurity control is inherently superior in this new system due to the absence of water-sensitive reagents and the use of biphasic or easily separable solvent systems. In conventional methods, trace moisture leads to the hydrolysis of isocyanates into amines, which then react further to create difficult-to-remove urea dimers. The cyanate route, however, tolerates aqueous conditions in the first step, allowing for a robust workup where inorganic salts are washed away with water. The organic layer, containing the high-purity arylurea, can be directly recycled or used in the next step without rigorous drying. This inherent resistance to moisture-induced side reactions ensures a consistent impurity profile, which is critical for meeting the stringent specifications required by regulatory bodies for veterinary drug substances.
How to Synthesize 1-methyl-5-[3-methyl-4-(4-trifluoromethylthio-phenoxy)-phenyl]-biuret Efficiently
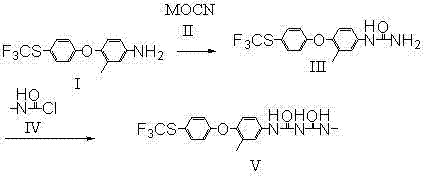
The synthesis protocol outlined in the patent provides a clear, scalable pathway for producing high-purity biuret intermediates suitable for commercial manufacturing. The process begins with the careful addition of cyanate to an arylamine solution under acidic conditions, followed by a controlled temperature ramp to ensure complete conversion to the urea intermediate. The second stage involves the reaction of this isolated or in-situ urea with methylcarbamyl chloride in solvents like toluene or ethyl acetate. Detailed standardized synthesis steps see the guide below.
- React 3-methyl-4-(4-trifluoromethylthio-phenoxy)-aniline with cyanate in the presence of acid to form N-[3-methyl-4-(4-trifluoromethylthio-phenoxy)-phenyl]-urea.
- React the resulting aryl urea with methylcarbamyl chloride in an organic solvent at 40°C to 90°C.
- Filter and dry the final 1-methyl-5-[3-methyl-4-(4-trifluoromethylthio-phenoxy)-phenyl]-biuret product for high-purity applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this phosgene-free technology offers substantial strategic benefits beyond mere chemical efficiency. The elimination of phosgene removes a major bottleneck in raw material sourcing, as cyanate salts are commodity chemicals with stable pricing and abundant global availability. This shift reduces the dependency on specialized chemical suppliers who handle hazardous gases, thereby diversifying the supply base and mitigating the risk of disruption due to safety incidents or regulatory shutdowns at vendor sites. Furthermore, the simplified waste profile means lower costs for environmental compliance and waste disposal, directly impacting the bottom line of the manufacturing operation.
- Cost Reduction in Manufacturing: By removing the need for expensive phosgene handling infrastructure and specialized scrubbing systems, manufacturers can achieve significant capital and operational cost savings. The use of cheaper, non-hygroscopic reagents like cyanate instead of MU reduces raw material costs and minimizes batch failures caused by reagent degradation. Additionally, the higher yields observed in this process mean less raw material is wasted per kilogram of final product, further driving down the cost of goods sold without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the cyanate route against moisture and its use of common organic solvents ensure consistent batch-to-batch performance. This reliability translates to predictable production schedules and reduced lead times, as there is less need for re-processing or troubleshooting failed batches. The availability of raw materials like potassium cyanate and methylcarbamyl chloride is high, ensuring that production can be scaled up rapidly to meet market demand without facing supply constraints typical of hazardous specialty gases.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup capabilities make this process highly scalable from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation aligns with green chemistry principles, facilitating easier permitting and compliance with increasingly strict environmental regulations. This environmental advantage also enhances the brand reputation of the manufacturer as a sustainable partner, which is a growing requirement for multinational pharmaceutical and agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: Why is the cyanate route preferred over the phosgene method for toltrazuril intermediates?
A: The cyanate route eliminates the use of hypertoxic phosgene gas, significantly reducing safety risks and regulatory compliance costs associated with hazardous gas handling and scrubbing systems.
Q: What are the yield advantages of the new biuret synthesis method?
A: The novel method achieves transformation efficiencies exceeding 96% in the urea formation step and over 97% in the final biuret cyclization, surpassing the 72% total conversion of conventional phosgene-based routes.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process utilizes mild reaction conditions (0°C to 90°C) and common organic solvents like toluene or ethyl acetate, making it highly suitable for large-scale commercial manufacturing without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Toltrazuril Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic technologies to deliver superior veterinary pharmaceutical intermediates. Our CDMO expertise allows us to seamlessly translate patent innovations like CN102731351B into robust commercial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this phosgene-free route are realized at an industrial level. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 1-methyl-5-[3-methyl-4-(4-trifluoromethylthio-phenoxy)-phenyl]-biuret meets the highest global standards.
We invite procurement leaders to collaborate with us on optimizing their supply chains for toltrazuril production. By leveraging our technical capabilities, you can secure a stable, cost-effective source of high-quality intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this technology can enhance your operational efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
