Advanced Synthesis and Isolation of Anastrozole Succinimide Impurity for Global Pharmaceutical Quality Control
Advanced Synthesis and Isolation of Anastrozole Succinimide Impurity for Global Pharmaceutical Quality Control
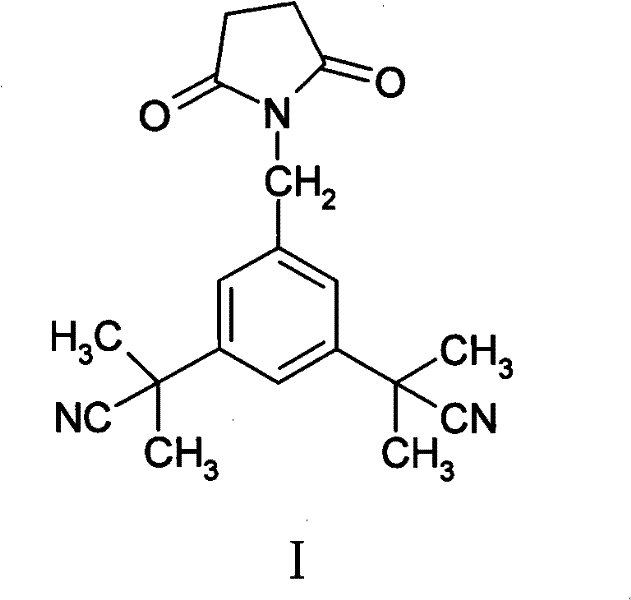
The pharmaceutical industry's relentless pursuit of patient safety mandates rigorous control over the impurity profiles of Active Pharmaceutical Ingredients (APIs). In the context of oncology therapeutics, specifically for the potent aromatase inhibitor Anastrozole, the identification and quantification of trace organic impurities are not merely regulatory checkboxes but critical safety imperatives. Patent CN102050775A introduces a groundbreaking methodology for the preparation of α,α,α′,α′-tetramethyl-5-(succinimide-1-methyl)-1,3-benzenediacetonitrile, a structurally complex impurity that arises during the industrial synthesis of Anastrozole. This technical disclosure provides a robust pathway for generating high-purity reference standards, addressing a significant gap in the supply chain for analytical controls. By leveraging a clever recovery strategy from the main synthesis stream, this innovation offers a reliable pharmaceutical intermediate supplier solution that enhances both the economic and technical feasibility of producing certified reference materials (CRMs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of specific process-related impurities for API quality control has been a bottleneck for pharmaceutical manufacturers. Conventional approaches often rely on purchasing small quantities of custom-synthesized standards from niche vendors, which frequently results in exorbitant costs, long lead times, and inconsistent purity profiles. Furthermore, attempting to isolate these impurities from pilot plant batches without a dedicated protocol is often inefficient, as the impurities exist in trace amounts and are chemically similar to the API, making separation via standard chromatography difficult and costly. The lack of a standardized, scalable method for generating the succinimide-based impurity of Anastrozole has historically complicated the validation of analytical methods, forcing quality control laboratories to rely on less specific detection methods that may fail to identify critical genotoxic alerts or process deviations.
The Novel Approach
The methodology outlined in Patent CN102050775A represents a paradigm shift by integrating the production of the impurity standard directly into the understanding of the Anastrozole synthesis mechanism. Rather than treating the succinimide derivative solely as an unwanted contaminant to be discarded, this approach systematically isolates it from the mother liquor and washing fractions of the main reaction. This "waste-to-value" strategy ensures a consistent supply of the reference material without requiring a separate, dedicated synthesis line. By optimizing the workup conditions—specifically the pH adjustments and solvent extraction sequences—the process maximizes the recovery of the target impurity while maintaining the integrity of the main API yield. This dual-output capability significantly streamlines the manufacturing workflow, offering a distinct advantage in cost reduction in pharmaceutical intermediate manufacturing by monetizing what was previously considered process waste.
Mechanistic Insights into NBS-Mediated Benzylic Functionalization
The formation of the target impurity is rooted in the mechanistic nuances of the benzylic bromination step using N-bromosuccinimide (NBS). In the primary synthesis of Anastrozole, the starting material, 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene, undergoes radical bromination to generate the reactive benzyl bromide intermediate. However, NBS is a dual-function reagent; while it serves as a source of bromine radicals, it also generates succinimide as a byproduct. Under the specific thermal and radical conditions employed (reflux in carbon tetrachloride with benzoyl peroxide initiation), a competitive nucleophilic substitution or radical coupling can occur where the succinimide anion attacks the benzylic position. This side reaction installs the succinimide ring onto the central aromatic core, creating the structural scaffold of the impurity. Understanding this mechanistic pathway is crucial for high-purity pharmaceutical intermediates development, as it allows chemists to predict and control the levels of this specific byproduct.
Following the formation of the bromo-intermediate, the subsequent reaction with 1,2,4-triazole to form Anastrozole creates a complex mixture. The impurity, lacking the triazole ring but possessing the bulky succinimide group, exhibits different solubility characteristics, particularly in acidic versus basic media. The patent exploits these differences during the workup phase. By carefully managing the acid-base extraction—dissolving the crude mixture in hydrochloric acid and washing with anhydrous ether—the succinimide impurity partitions into the organic ether layer, while the basic triazole-containing API remains in the aqueous phase as a salt. This elegant separation technique avoids the need for preparative HPLC, demonstrating a sophisticated grasp of physical organic chemistry to achieve commercial scale-up of complex pharmaceutical intermediates with high efficiency.
How to Synthesize Anastrozole Succinimide Impurity Efficiently
The synthesis protocol described in the patent is designed for reproducibility and scalability, making it ideal for industrial reference standard production. The process begins with the controlled radical bromination of the toluene derivative, followed by the triazole substitution reaction. The critical innovation lies not in the reaction itself, but in the downstream processing where the impurity is harvested from the ether washings of the crude API. Detailed operational parameters, including temperature controls during concentration (maintaining internal temperatures between 30°C and 60°C) and specific solvent ratios for recrystallization, are essential to achieving the reported purity of >98%. For a comprehensive guide on executing this procedure with GMP-compliant documentation, please refer to the standardized operating procedures below.
- Perform radical bromination of 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene using N-bromosuccinimide (NBS) and benzoyl peroxide in carbon tetrachloride to generate the bromo-intermediate.
- React the bromo-intermediate with 1,2,4-triazole under basic conditions to form Anastrozole, while simultaneously generating the succinimide impurity as a byproduct.
- Isolate the target impurity from the anhydrous ether washing liquids obtained during the acidic workup of the crude Anastrozole, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the implementation of this patented isolation method offers tangible strategic benefits beyond mere technical compliance. The ability to internally generate or reliably source this specific impurity standard mitigates the risk of supply disruptions associated with external specialty chemical vendors. It transforms a potential liability (process impurity) into a valuable asset (QC standard), thereby optimizing the overall cost structure of the API production facility. This approach aligns perfectly with lean manufacturing principles, reducing the total cost of ownership for quality assurance operations.
- Cost Reduction in Manufacturing: By recovering the impurity from the existing process stream, the need for purchasing expensive external reference standards is eliminated. This internal generation capability drastically reduces the direct material costs associated with quality control testing. Furthermore, the utilization of standard industrial solvents and reagents (such as NBS and common organic acids/bases) ensures that the marginal cost of producing the standard is negligible compared to the value it adds in batch release testing. This efficiency drives significant operational expenditure (OpEx) savings over the lifecycle of the drug product.
- Enhanced Supply Chain Reliability: Dependence on third-party suppliers for critical analytical standards introduces vulnerability regarding lead times and certificate of analysis (COA) availability. Adopting this in-house or partnered recovery method ensures a continuous, on-demand supply of the reference material. This reliability is paramount for maintaining uninterrupted production schedules and meeting strict regulatory submission deadlines. It effectively decouples the QC laboratory's readiness from the volatility of the external fine chemicals market, ensuring that batch release testing never becomes a bottleneck.
- Scalability and Environmental Compliance: The process utilizes established unit operations such as reflux, filtration, and recrystallization, which are easily scalable from gram to kilogram quantities without requiring specialized equipment. Additionally, by capturing and purifying a byproduct that would otherwise contribute to the waste stream, the process contributes to a lower environmental footprint. This "green chemistry" aspect of waste valorization supports corporate sustainability goals and simplifies the regulatory narrative regarding waste management and process mass intensity (PMI).
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this Anastrozole impurity standard. These insights are derived directly from the experimental data and claims within Patent CN102050775A, providing a factual basis for decision-making regarding quality control strategies.
Q: What is the primary application of the succinimide-based Anastrozole impurity?
A: This specific compound serves as a critical reference standard for HPLC and GC analysis, allowing manufacturers to accurately quantify trace impurities in Anastrozole API batches to meet ICH Q3A/B guidelines.
Q: How does this patent improve the availability of the reference standard?
A: Instead of complex de novo synthesis, the method recovers the impurity from the process stream of the main API synthesis, turning a potential waste product into a high-value analytical tool.
Q: What is the purity level achievable with this isolation method?
A: The patent demonstrates that through specific solvent washing and recrystallization techniques using ethyl acetate and n-hexane, purity levels exceeding 98% can be consistently achieved.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anastrozole Impurity Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your final drug product depends on the precision of your analytical controls. As a leading CDMO and fine chemical manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of characterizing complex impurities to stringent purity specifications, ensuring that every reference standard we provide meets the highest global regulatory requirements. We are committed to supporting your R&D and QA teams with materials that facilitate accurate, compliant, and efficient drug development.
We invite you to collaborate with our technical team to explore how this innovative isolation technology can be integrated into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. Let us provide you with specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in securing the quality and continuity of your Anastrozole supply chain.
