Advanced Titanium Catalysis for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the synthesis of chiral building blocks, particularly optically active alpha-aminonitriles which serve as critical precursors for alpha-amino acids. Patent CN101848916A introduces a groundbreaking titanium catalyst system designed for the asymmetric cyanation of imines, offering a significant departure from traditional methods that often rely on expensive ligands or harsh reaction conditions. This technology leverages a partially hydrolyzed titanium alkoxide complexed with a chiral N-salicyl-beta-amino alcohol ligand to achieve exceptional stereocontrol. For R&D Directors and Procurement Managers, this represents a viable pathway to enhance the purity and cost-efficiency of API intermediate manufacturing. The ability to operate under mild conditions while maintaining high enantiomeric excess addresses key pain points in process development and supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the catalytic asymmetric hydrocyanation of imines has been plagued by significant operational challenges that hinder large-scale adoption in commercial settings. Many existing catalyst systems necessitate the use of highly expensive chiral ligands that require multi-step synthesis, driving up the raw material costs substantially. Furthermore, these traditional processes often demand严苛 reaction conditions, such as cryogenic temperatures below -78°C, to maintain acceptable levels of enantioselectivity. Such requirements impose a heavy burden on energy consumption and specialized equipment, making the cost reduction in API manufacturing difficult to achieve. Additionally, the sensitivity of many organometallic catalysts to moisture and oxygen requires strictly anhydrous and inert atmospheres, complicating the workflow and increasing the risk of batch failure due to environmental exposure.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes a titanium-based catalyst system that operates efficiently under ambient conditions, fundamentally altering the economic landscape of this transformation. By employing a partially hydrolyzed titanium alkoxide precursor, the method eliminates the need for extreme cooling, allowing reactions to proceed at room temperature or slightly above without compromising yield or selectivity. This shift not only reduces energy costs but also simplifies the reactor setup, making it more accessible for standard chemical manufacturing facilities. The use of inexpensive and readily available building blocks for the chiral ligands further enhances the economic viability, providing a reliable pharmaceutical intermediate supplier with a competitive edge. The robustness of this system against moisture also streamlines the operational protocol, reducing the complexity associated with solvent drying and inert gas purging.
Mechanistic Insights into Titanium-Catalyzed Asymmetric Cyanation
The core of this technological advancement lies in the unique activation mechanism of the titanium catalyst, which involves the controlled hydrolysis of titanium alkoxides. When water is introduced to the titanium alkoxide monomer, it forms a partially hydrolyzed species that acts as a pre-catalyst. This species then coordinates with the chiral N-salicyl-beta-amino alcohol ligand, creating an active catalytic center capable of distinguishing between enantiotopic faces of the imine substrate. The presence of water is not merely tolerated but is integral to the formation of the active catalytic structure, which is a significant deviation from traditional moisture-sensitive Lewis acid catalysts. This mechanistic feature ensures that the catalyst remains stable and active even in the presence of residual moisture in the solvent, thereby enhancing the reproducibility of the reaction across different batches and scales.
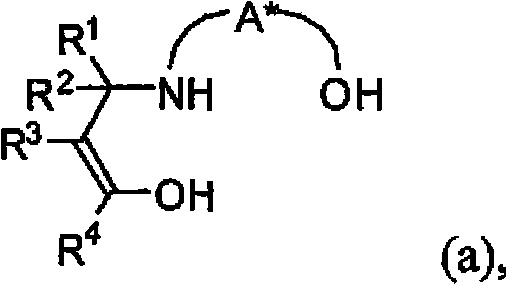
Furthermore, the stereochemical outcome of the reaction is meticulously controlled by the chiral environment provided by the ligand framework. The specific arrangement of the hydroxyl and amino groups in the ligand creates a rigid coordination sphere around the titanium center, directing the approach of the cyanating agent to the imine bond with high precision. This results in the formation of optically active alpha-aminonitriles with enantiomeric excess values reaching up to 98% ee, as demonstrated in various embodiments within the patent. The ability to achieve such high optical purity is crucial for pharmaceutical applications where impurity profiles are strictly regulated. By minimizing the formation of unwanted enantiomers, this process reduces the need for downstream purification steps, thereby improving the overall efficiency and yield of the synthesis pathway for high-purity chiral aminonitriles.
How to Synthesize Optically Active Alpha-Aminonitriles Efficiently
The synthesis of these valuable chiral intermediates follows a streamlined protocol that begins with the in situ preparation of the titanium catalyst. This involves mixing a titanium alkoxide with a controlled amount of water or a water source, such as an inorganic hydrate, in an organic solvent like toluene. Once the partially hydrolyzed species is formed, the chiral ligand is added to generate the active catalyst. This mixture is then combined with the imine substrate and a cyanating agent, such as trimethylsilylnitrile or hydrogen cyanide. The reaction proceeds smoothly at room temperature, often reaching completion within 15 minutes to 2 hours. The detailed standardized synthesis steps see the guide below.
- Prepare the titanium catalyst by contacting a titanium alkoxide with water to form a partially hydrolyzed species, then mix with a chiral N-salicyl-beta-amino alcohol ligand.
- Combine the prepared catalyst with an imine substrate and a cyanating agent such as trimethylsilylnitrile or hydrogen cyanide in an organic solvent.
- Stir the reaction mixture at room temperature for 15 minutes to 2 hours to achieve high conversion and enantioselectivity without strict inert atmosphere requirements.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this titanium-catalyzed process offers substantial strategic benefits that extend beyond mere technical performance. The primary advantage lies in the significant cost savings derived from the use of inexpensive reagents and the elimination of energy-intensive cooling requirements. Traditional asymmetric cyanation often relies on cryogenic conditions, which demand specialized equipment and high energy consumption, whereas this method operates effectively at ambient temperatures. This reduction in operational complexity translates directly into lower manufacturing costs, allowing for more competitive pricing in the global market. Additionally, the robustness of the catalyst against moisture reduces the need for expensive drying agents and strict inert atmosphere controls, further lowering the cost of goods sold.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the use of commercially available titanium alkoxides and simple chiral ligands that do not require complex multi-step synthesis. By avoiding the need for precious metal catalysts or exotic ligands, the raw material costs are significantly minimized. Furthermore, the ability to run reactions at room temperature eliminates the capital and operational expenditures associated with cryogenic cooling systems. The high conversion rates and yields observed in the patent data mean that less raw material is wasted, contributing to a more sustainable and cost-effective production model. These factors collectively ensure a substantial cost reduction in API manufacturing without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized reagents or conditions that are difficult to maintain at scale. This titanium catalyst system mitigates such risks by utilizing reagents that are readily available from multiple global suppliers. The tolerance to ambient conditions and residual water means that the process is less susceptible to disruptions caused by equipment failure or environmental fluctuations. This robustness ensures a consistent supply of high-purity intermediates, which is critical for maintaining production schedules in the pharmaceutical industry. By reducing the lead time for high-purity chiral aminonitriles through a more reliable process, manufacturers can better respond to market demands and avoid costly delays.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces new challenges, particularly regarding waste management and safety. This method supports the commercial scale-up of complex pharmaceutical intermediates by minimizing the generation of hazardous waste associated with solvent drying and catalyst disposal. The one-pot synthesis capability described in the patent further reduces the number of unit operations, thereby lowering the overall environmental footprint. The use of less toxic cyanating agents and the potential for catalyst recycling align with modern green chemistry principles. This makes the process not only scalable but also compliant with increasingly stringent environmental regulations, ensuring long-term viability for manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric cyanation technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows. The insights provided here aim to clarify the operational parameters and potential benefits for your specific application needs.
Q: What are the advantages of the titanium catalyst system described in CN101848916A?
A: The catalyst system allows for asymmetric cyanation under mild conditions, such as room temperature, with high enantioselectivity up to 98% ee and yields exceeding 99%, reducing the need for energy-intensive cooling.
Q: Can this process be scaled for commercial pharmaceutical intermediate production?
A: Yes, the process supports commercial scale-up of complex pharmaceutical intermediates due to its tolerance to ambient conditions, use of inexpensive reagents, and potential for one-pot synthesis which simplifies workflow.
Q: Does the catalyst require strictly anhydrous conditions?
A: No, the method utilizes controlled water addition for catalyst activation and can tolerate residual water in solvents, significantly lowering the operational complexity compared to traditional moisture-sensitive catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Aminonitriles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic methodologies to maintain a competitive edge in the pharmaceutical supply chain. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the titanium-catalyzed asymmetric cyanation can be successfully translated into industrial reality. We are committed to delivering products with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest standards required for API synthesis. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing you with a partner who understands the nuances of chiral intermediate manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed decisions about your sourcing strategy. Contact us today to discuss how we can support your project with reliable, high-quality chiral intermediates produced using state-of-the-art catalytic methods.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
