Advanced Purification Strategies for High-Purity Canagliflozin API Intermediates
The global demand for effective antidiabetic medications has driven intense research into SGLT-2 inhibitors, with Canagliflozin standing out as a critical therapeutic agent. However, the commercial viability of this compound hinges on the ability to control stereoisomeric impurities, specifically the alpha-configuration which can compromise drug safety and efficacy. Patent CN103694230A introduces a groundbreaking purification methodology that addresses this challenge through the formation of eutectic substances with amino acids. This technical insight report analyzes the proprietary data to demonstrate how this novel approach offers a superior alternative to traditional synthesis routes, ensuring high-purity outputs essential for regulatory compliance. By leveraging chiral resolution via amino acid complexation, manufacturers can achieve alpha-configuration impurity levels lower than 0.5%, a benchmark that significantly enhances the therapeutic profile of the final API. This document serves as a strategic guide for R&D and procurement leaders seeking to optimize their supply chains for high-value pharmaceutical intermediates.
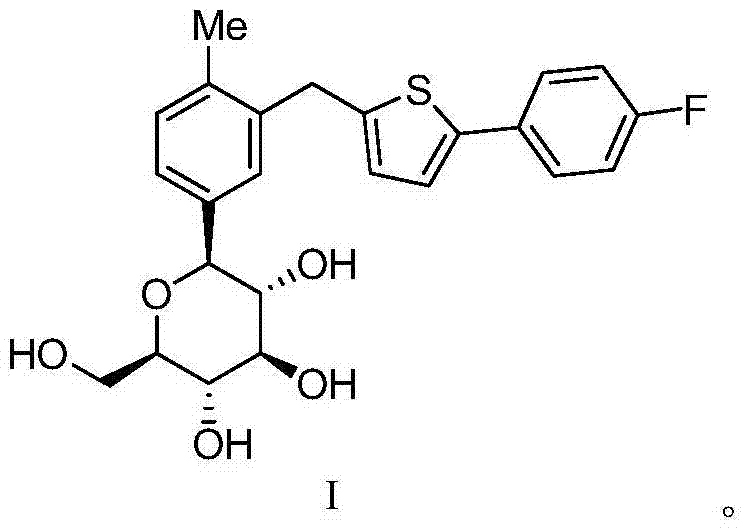
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Canagliflozin often struggle with the inherent difficulty of controlling stereochemistry during the glycosidic bond formation. As illustrated in prior art, conventional processes frequently generate a mixture of alpha and beta configurations, necessitating complex downstream purification steps to isolate the active beta-isomer. Methods such as those disclosed in patent application CN101801371 typically involve acetylation of hydroxyl groups followed by deprotection and recrystallization. While effective to a degree, these multi-step sequences inevitably lead to yield losses and increased consumption of reagents and solvents. Furthermore, the reliance on protection group chemistry adds significant time and cost to the manufacturing process, creating bottlenecks in production schedules. The persistence of alpha-configuration impurities, often remaining above 1% even after rigorous processing, poses a significant risk to quality control standards. These inefficiencies highlight the urgent need for a more streamlined and selective purification technology that can bypass the limitations of classical organic synthesis.
The Novel Approach
The innovation detailed in CN103694230A represents a paradigm shift by utilizing supramolecular chemistry principles rather than covalent modification for purification. Instead of adding and removing protecting groups, this method exploits the specific intermolecular interactions between Canagliflozin and selected amino acids to form a eutectic substance. This eutectic formation acts as a highly selective filter, preferentially incorporating the desired beta-configuration into the crystal lattice while excluding the alpha-impurity. The process involves dissolving crude Canagliflozin with an amino acid such as L-Proline or L-Phenylalanine in a solvent like ethanol, followed by controlled crystallization. This single-step complexation effectively reduces the alpha-configuration impurity to below 0.5% without the need for additional chemical transformations. By eliminating the acetylation and deprotection stages, the novel approach drastically simplifies the workflow, reducing both the environmental footprint and the operational complexity. This strategic simplification not only enhances purity but also aligns perfectly with modern green chemistry initiatives in pharmaceutical manufacturing.
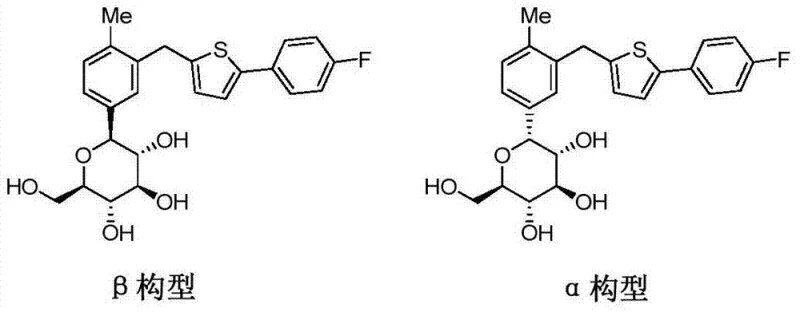
Mechanistic Insights into Amino Acid Eutectic Crystallization
The core mechanism driving this purification success lies in the stereoselective recognition between the chiral centers of Canagliflozin and the amino acid resolver. When crude Canagliflozin is mixed with a chiral amino acid like L-Proline in a suitable solvent, the molecules self-assemble into a specific crystalline arrangement known as a eutectic complex. This assembly is thermodynamically favored for the beta-configuration due to complementary hydrogen bonding and steric fit, whereas the alpha-configuration disrupts this lattice stability. As the solution cools or solvent is removed, the stable eutectic crystals precipitate out, effectively sequestering the target molecule away from the impurity which remains dissolved in the mother liquor. This physical separation is far more efficient than chemical derivatization because it avoids the generation of by-products associated with protection group chemistry. The choice of amino acid is critical, with hydrophobic amino acids such as L-Proline and L-Phenylalanine demonstrating superior resolution efficiency compared to hydrophilic variants. This selectivity ensures that the subsequent decomposition of the complex yields Canagliflozin with exceptional optical purity, meeting the stringent requirements for antidiabetic drug substances.
Impurity control is further enhanced by the reversibility of the eutectic formation, allowing for a clean release of the purified API. Once the eutectic crystals are filtered and washed, they are suspended in water and subjected to pH adjustment using acid or alkali. This step breaks the intermolecular bonds holding the complex together, releasing the free Canagliflozin into the solution while the amino acid remains in the aqueous phase or is otherwise separated. The purified Canagliflozin is then extracted into an organic solvent, concentrated, and dried to obtain the final high-purity product. This decomposition step is crucial as it ensures no residual amino acid contaminates the final API, maintaining the integrity of the impurity profile. The entire mechanism relies on physical phase changes and pH-dependent solubility rather than harsh chemical reactions, minimizing the risk of generating new degradation products. Consequently, the impurity profile is significantly cleaner, with the alpha-configuration consistently maintained below the critical 0.5% threshold defined in the patent data.
How to Synthesize High-Purity Canagliflozin Efficiently
Implementing this purification strategy requires precise control over solvent selection, temperature gradients, and molar ratios to maximize yield and purity. The process begins by dissolving crude Canagliflozin and the chosen amino acid resolver in a solvent system such as ethanol or acetone, often requiring heating to ensure complete solubility. Detailed standard operating procedures regarding specific cooling rates, stirring speeds, and filtration techniques are essential for reproducibility at scale. The following guide outlines the critical operational phases derived from the patent examples to assist technical teams in process validation. For comprehensive step-by-step instructions and parameter optimization, please refer to the standardized protocol section below.
- Dissolve crude Canagliflozin and a selected hydrophobic amino acid, such as L-Proline, in a suitable solvent like ethanol or acetone under heating.
- Cool the solution to induce crystallization of the Canagliflozin-amino acid eutectic complex, then filter to separate the solid complex from the mother liquor.
- Suspend the complex in water, adjust pH to acidic or alkaline conditions to decompose the complex, and extract the purified Canagliflozin into an organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this amino acid-based purification route offers substantial advantages in cost structure and supply chain resilience. By removing the need for acetylation reagents and the associated deprotection steps, the process significantly reduces the bill of materials and waste disposal costs. The reliance on commodity chemicals like ethanol and common amino acids ensures that raw material sourcing is stable and not subject to the volatility of specialized reagent markets. This simplification of the synthetic route translates directly into a more robust supply chain, capable of sustaining continuous production without the bottlenecks typical of multi-step protection strategies. Furthermore, the high yields reported in the patent examples, ranging from 78% to 90%, indicate a highly efficient use of starting materials, minimizing waste and maximizing output per batch. These factors combine to create a compelling economic case for switching to this technology for large-scale API manufacturing.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps removes the need for expensive acetylating agents and the solvents required to handle them. This reduction in chemical consumption directly lowers the variable cost per kilogram of produced Canagliflozin. Additionally, the simplified workflow reduces energy consumption associated with heating and cooling cycles for multiple reaction stages. The use of recyclable solvents like ethanol and ethyl acetate further enhances the economic efficiency by allowing for solvent recovery and reuse. Overall, the process design inherently drives down manufacturing costs through material and operational efficiency.
- Enhanced Supply Chain Reliability: Sourcing amino acids such as L-Proline and L-Phenylalanine is significantly more reliable than sourcing specialized chiral catalysts or protecting group reagents. These amino acids are produced in massive quantities for the food and feed industries, ensuring a stable and diverse supplier base. This availability mitigates the risk of supply disruptions that can halt API production lines. Moreover, the process tolerance for various solvents, including methanol, isopropanol, and acetone, provides flexibility in procurement, allowing supply chain managers to adapt to market fluctuations. This flexibility ensures consistent production schedules and reliable delivery timelines for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The crystallization and filtration unit operations used in this method are standard in the chemical industry, making scale-up from pilot to commercial production straightforward. There are no exotic reaction conditions or hazardous reagents that would require specialized containment or safety measures. The reduction in chemical steps also leads to a lower E-factor, meaning less waste is generated per unit of product. This aligns with increasingly strict environmental regulations and corporate sustainability goals. The ability to produce high-purity material with a smaller environmental footprint enhances the marketability of the API to eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing contexts. The responses cover impurity control, process scalability, and regulatory considerations relevant to API production.
Q: How does the amino acid eutectic method reduce alpha-configuration impurities?
A: The method leverages the specific stereochemical interaction between Canagliflozin and chiral amino acids. By forming a eutectic complex, the process selectively crystallizes the desired beta-configuration while leaving the alpha-configuration impurity in the mother liquor, achieving levels below 0.5%.
Q: What are the advantages of this purification route over conventional acetylation methods?
A: Conventional methods often require additional protection and deprotection steps, such as acetylation, which increase material costs and processing time. The amino acid eutectic approach simplifies the workflow by eliminating these extra reaction steps, thereby reducing solvent consumption and improving overall process efficiency.
Q: Is this purification process suitable for large-scale commercial production?
A: Yes, the process utilizes common solvents like ethanol and readily available amino acids, making it highly scalable. The crystallization and filtration steps are standard unit operations in pharmaceutical manufacturing, ensuring robust supply chain continuity and ease of technology transfer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Canagliflozin Supplier
The technical potential of the amino acid eutectic purification method represents a significant opportunity for optimizing the production of high-value antidiabetic intermediates. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, combining it with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Canagliflozin meets the highest global standards. We understand the critical nature of impurity control in SGLT-2 inhibitors and have the expertise to implement this patent-inspired route with precision and consistency. Partnering with us means gaining access to a supply chain that is both technically sophisticated and commercially robust.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this purification strategy can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this streamlined process for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver high-purity Canagliflozin reliably. Let us collaborate to enhance your product quality while optimizing your manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
