Advanced Synthesis of 6-Methoxy-1-Tetralone for High-Purity Steroid Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for critical steroid intermediates, and Patent CN114436790A presents a significant breakthrough in the production of 6-methoxy-1-tetralone. This compound serves as an indispensable foundational raw material for the total synthesis of numerous steroidal drugs, including mifepristone, norethindrone, drospirenone, and levonorgestrel. Historically, the supply chain for this key intermediate has been constrained by hazardous processing conditions and low overall yields, creating bottlenecks for downstream API manufacturers. The disclosed invention introduces a novel four-step synthetic strategy that fundamentally alters the reaction landscape by replacing dangerous high-pressure hydrogenation with a controlled Grignard addition and cyclization sequence. This technical evolution not only enhances the safety profile of the manufacturing process but also delivers a substantial improvement in total mass yield, reaching levels significantly superior to conventional methods. For R&D directors and procurement specialists, understanding this shift is vital for securing a stable supply of high-purity steroid precursors.
The Limitations of Conventional Methods vs. The Novel Approach
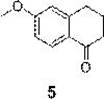
The Limitations of Conventional Methods
Prior to this innovation, the industrial standard for synthesizing 6-methoxy-1-tetralone relied heavily on the hydrogenation and oxidation of 2-methoxynaphthalene, a pathway fraught with significant operational and environmental liabilities. The initial hydrogenation step necessitates high-pressure catalytic equipment, introducing severe safety risks related to hydrogen gas handling and requiring substantial capital expenditure for specialized pressure vessels. Furthermore, the subsequent oxidation step traditionally employs chromium trioxide as the oxidizing agent, which generates heavy metal pollution that is increasingly difficult to manage under modern environmental regulations. The cumulative effect of these harsh conditions is a notoriously low total yield, often reported to be less than 35%, which drastically inflates the cost of goods sold. Additionally, this oxidative process tends to produce a persistent 4-keto group impurity that is chemically similar to the target product, making separation and purification extremely challenging and resource-intensive for quality control laboratories.
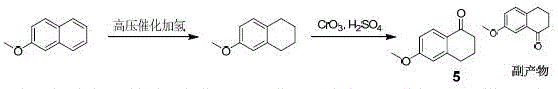
The Novel Approach
In stark contrast to the legacy methodology, the new approach detailed in the patent utilizes 3-bromoanisole as a starting material, leveraging a Grignard reaction followed by a reductive cyclization strategy that circumvents the need for high-pressure infrastructure. By initiating the synthesis with the formation of a 3-methoxyphenyl magnesium bromide solution, the process establishes a highly reactive intermediate that can be precisely coupled with ethyl succinate monoacyl chloride under strictly controlled low-temperature conditions. This sequence avoids the use of toxic chromium reagents entirely, replacing them with more manageable reagents like triethylsilane and thionyl chloride, which simplifies waste stream treatment and reduces environmental compliance costs. The result is a streamlined workflow that not only mitigates safety hazards associated with high-pressure hydrogenation but also achieves a total mass yield of approximately 65%, nearly doubling the efficiency of the traditional route and providing a more economically viable option for large-scale commercial production.
Mechanistic Insights into Grignard Addition and Friedel-Crafts Cyclization
The core of this synthetic advancement lies in the precise control of the Grignard reagent formation and the subsequent intramolecular Friedel-Crafts acylation. The process begins with the activation of metal magnesium using methyl iodide in toluene, followed by the dropwise addition of 3-bromoanisole at temperatures between 60°C and 100°C to ensure complete consumption of the magnesium. Critical to the success of the next step is the rigorous temperature control during the addition of the Grignard solution to ethyl succinate monoacyl chloride, which must be maintained between -45°C and -55°C to prevent side reactions and ensure the formation of the desired ketone intermediate. Following this, a reduction step using triethylsilane and trifluoroacetic acid converts the ketone to a methylene group, setting the stage for the final ring closure. This multi-step cascade demonstrates a sophisticated understanding of organic reactivity, allowing for the construction of the tetralone skeleton with high regioselectivity and minimal byproduct formation.
Impurity control is inherently built into this mechanism through the avoidance of over-oxidation and the use of specific recrystallization solvents in the final stage. Unlike the chromium oxidation method which generates difficult-to-remove 4-keto impurities, this route produces a crude product that can be effectively purified using an isopropanol-n-heptane solvent system. The final cyclization step, mediated by anhydrous aluminum trichloride and thionyl chloride, proceeds under mild thermal conditions, further reducing the risk of thermal degradation or polymerization of the sensitive intermediate. The patent data indicates that the final product can achieve an HPLC purity of 99%, demonstrating that the mechanistic pathway effectively suppresses the formation of structural analogs. For technical teams, this level of purity assurance is critical, as it reduces the burden on downstream purification processes during the synthesis of complex steroid hormones.
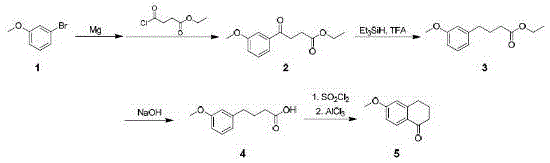
How to Synthesize 6-Methoxy-1-Tetralone Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and thermal management to replicate the high yields reported in the patent documentation. Operators must ensure that the Grignard reagent is fully formed before proceeding to the acylation step, as any unreacted magnesium or halide can compromise the subsequent reduction. The protocol outlines specific mass ratios for key reagents, such as maintaining a triethylsilane to trifluoroacetic acid ratio of approximately 2:1.5 to optimize the reduction efficiency. Detailed standardized synthesis steps see the guide below.
- Prepare 3-methoxyphenyl magnesium bromide from 3-bromoanisole and magnesium in toluene, then react with ethyl succinate monoacyl chloride at -45°C to -55°C.
- Reduce the resulting ketone intermediate using triethylsilane and trifluoroacetic acid in dichloromethane to form the methylene derivative.
- Hydrolyze the ester group using sodium hydroxide in methanol under reflux to obtain the carboxylic acid intermediate.
- Perform intramolecular Friedel-Crafts acylation using thionyl chloride and anhydrous aluminum trichloride to cyclize and form 6-methoxy-1-tetralone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers tangible benefits that extend beyond simple chemical yield improvements. By eliminating the requirement for high-pressure hydrogenation equipment, manufacturers can significantly reduce capital expenditure requirements and lower the barrier to entry for scaling production capacity. The removal of chromium trioxide from the process flow drastically simplifies waste management protocols, leading to substantial cost savings in environmental compliance and hazardous waste disposal. Furthermore, the reliance on common chemical raw materials like 3-bromoanisole and ethyl succinate monoacyl chloride ensures a more resilient supply chain, as these commodities are widely available from multiple global sources, reducing the risk of raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal oxidants and the associated waste treatment procedures leads to a significant reduction in variable production costs. By avoiding the need for specialized high-pressure reactors, facilities can utilize standard glass-lined or stainless steel equipment, which lowers maintenance overhead and extends equipment lifespan. The improved total yield directly translates to lower raw material consumption per kilogram of finished product, enhancing the overall margin structure for the manufacturing operation without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as 3-bromoanisole mitigates the risk of supply disruptions often associated with specialized or regulated reagents. The simplified process flow, which avoids complex high-pressure steps, allows for more flexible production scheduling and faster turnaround times between batches. This operational agility ensures that downstream API manufacturers can rely on a consistent and continuous supply of 6-methoxy-1-tetralone, crucial for maintaining their own production timelines for steroidal drugs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to commercial scale without significant engineering hurdles. The absence of chromium waste streams aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against potential regulatory crackdowns. This eco-friendly profile not only reduces liability but also enhances the corporate sustainability image, which is becoming a key factor in vendor selection for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 6-methoxy-1-tetralone based on the patented methodology. These insights are derived directly from the experimental data and background analysis provided in the intellectual property documentation. Understanding these details helps stakeholders evaluate the feasibility of adopting this supply source for their specific steroid synthesis projects.
Q: How does this new synthesis method improve safety compared to traditional routes?
A: The traditional method requires dangerous high-pressure catalytic hydrogenation and uses chromium trioxide, a severe environmental hazard. This new protocol operates at ambient pressure and eliminates heavy metal oxidants, significantly reducing operational risk and waste treatment complexity.
Q: What is the expected purity and yield of 6-methoxy-1-tetralone using this process?
A: According to the patent data, the total mass yield calculated from the starting material 3-bromoanisole reaches approximately 65%, which is substantially higher than the prior art yield of less than 35%. The final product purity can reach 99% HPLC purity after recrystallization.
Q: Why is 6-methoxy-1-tetralone critical for steroid drug manufacturing?
A: It serves as a fundamental building block for the D-ring construction in various steroidal drugs such as mifepristone, levonorgestrel, and drospirenone. High purity of this intermediate is essential to prevent difficult-to-remove impurities in the final active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methoxy-1-Tetralone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that 6-methoxy-1-tetralone plays in the global steroid hormone market and have invested heavily in mastering this advanced synthetic pathway. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of large-scale API manufacturers. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical intermediate use. Our commitment to process safety and environmental stewardship aligns perfectly with the advantages offered by this chromium-free, ambient pressure synthesis route.
We invite you to collaborate with us to optimize your supply chain for steroid intermediates and leverage the cost efficiencies of this modern manufacturing technique. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your annual volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our production capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
